Introduction
Root-knot nematodes (RKN) [Meloidogyne arenaria (Neal) Chitwood race 1] are major problems in US peanut (Arachis hypogaea L.) production (Kokalis-Burelle and Rodriquez-Kabana, 1997). Consequently, new and improved RKN-resistant cultivars are needed to minimize damage, reduce chemical control costs, and increase yield, grade, and dollar value returns.
‘COAN’ (Simpson and Starr, 2001) was the first peanut cultivar developed with very high RKN-resistance. The RKN-resistance was introgressed into the cultigen as a single dominant gene, Rma, from a wild species cross [A. batizocoi Krapov. & W. C. Gregory × (A. cardenasii Krapov. & W. C. Gregory × A. diogoi Hoehne, formerly A. chacoense Krapov. & W. C. Gregory)] (Simpson et al., 1993). ‘NemaTAM’ (Simpson et al., 2003) was the second peanut cultivar released with further backcrosses to the recurrent parent ‘Florunner’ (Norden et al., 1969) cultivar. Unfortunately, Florunner is highly susceptible to tomato spotted wilt caused by Tomato spotted wilt virus (TSWV), and TSWV is also a major disease problem in the southeast U.S. So, the combination of TSWV and RKN-resistance was incorporated into the release of ‘Tifguard’ (Holbrook et al., 2008) cultivar which used COAN as the RKN-resistant donor parent.
Since these earlier cultivar releases, molecular markers were identified for marker assisted selections (MAS) and used to develop ‘Tifguard Hi-O/L’ peanut (Chu et al., 2011). Sequence characterized amplified region (SCAR) marker 197/909 is a new nematode resistance dominant marker (Chu et al., 2007); whereas, codominant simple sequence repeat (SSR) marker GM565 (Nagy et al., 2010) can be used to identify the heterozygotes for nematode resistance. The objective of the present study was to determine the accuracy of these two new molecular markers versus actual field test results for RKN-resistant screening.
Materials and Methods
During 2011 and 2012, field tests were conducted at the Rigdon Farm (latitude: 31.516° N and longitude: 83.545° W) and in 2012 at the Blackshank Farm (31.500° N and 83.545° W, respectively) near the Coastal Plain Experiment Station, Tifton, GA. Each year 15 genotypes (12 advanced Georgia breeding lines and 3 check cultivars) were evaluated at the Rigdon Farm location for RKN-resistance. The advanced breeding lines result from the same three-way cross combination [‘Georgia-02C’ (Branch, 2003) × (‘Georgia-01R’ (Branch, 2002) × COAN)], and the check cultivars were the RKN-resistant Tifguard and the susceptible check cultivars ‘Georgia-07W’ (Branch and Brenneman, 2008) and ‘Georgia Greener’ (Branch, 2007). Both test sites have a long history of high RKN populations and continuous peanut production. The soil type at both locations was a Tifton loamy sand (fine-loamy, kaolinitic, thermic Plinthic Kandiudults).
Planting dates at the Rigdon Farm were May 6, 2011 and May 11, 2012, and the seeding rates were six seed per 30.5-cm of row. Recommended cultural practices with irrigation were used throughout each growing season, except that no nematicides were applied with activity against RKN. A randomized complete block design was used each year with three replications. Plots consisted of two rows 7.62 m long × 1.83 m wide (0.91 m spacing between rows). The severity of nematode galling on roots and pods was rated after digging and inverting the plants each year. The percent damage was visually estimated from 0 to 100% with 0% representing no galls and 100% representing galling on all pods and roots. Leaf tissue samples from young unfolded terminal leaflets were taken from each plot early during the growing season and sent to ESTA Lab in Longmont, CO for molecular nematode screening using two markers (SCAR 197/909 and SSR-GM565). Individual plots were harvested near optimum maturity according to visual above-ground disease pressure in conjunction with the hull-scrape maturity method from adjoining border plots (Williams and Drexler, 1981). Plots were mechanically harvested, and pods were dried with forced warm air to 10% moisture content, before weighing for pod yield.
Planting date at the Blackshank Farm was April 27, 2012 at a seeding rate of six seed per 30.5-cm of row. Recommended cultural practices with irrigation were used throughout the growing season, except that no nematicides were applied with activity against RKN. A randomized complete block design was used with ten genotypes and five replications. Plots consisted of two rows 7.62 m long × 1.83 m wide (0.91 m spacing between rows). The severity of nematode galling on roots and pods was rated after digging and inverting the plants each year. The percent damage was visually estimated from 0 to 100%, with 0% representing no galls and 100% representing galling on all pods and roots. All plots were dug at the same time, Oct. 8. Plots were mechanically harvested, and pods were dried with forced warm air to 10% moisture content, before weighing for pod yield.
Reciprocal crosses were made in the fall of 2011 between nematode resistant parents (GA 082522 × Tifguard) and (GA 082524 × Tifguard). The F1 hybrids were grown in 2012, and F2 segregating populations were grown in the greenhouse during the winter months of 2012 to 2013. Leaf samples were taken from young immature leaflets and sent to ESTA Lab for RKN molecular marker screening using both SCAR 197/909 and SSR-GM565.
Data for all three field experiments were subjected to analysis of variance. LSD T-test was used for mean separation at the P≤0.05 probability level. Genetic analysis using molecular markers for RKN resistance was based upon individual plants, and segregation data was analyzed by chi-square program to test goodness-of-fit of observed vs. expected genetic ratios.
Results and Discussion
The 15 peanut genotypes used in this study were not present in all three field test experiments. Consequently, each test was analyzed separately, and the results from these field tests versus molecular markers will be discussed individually.
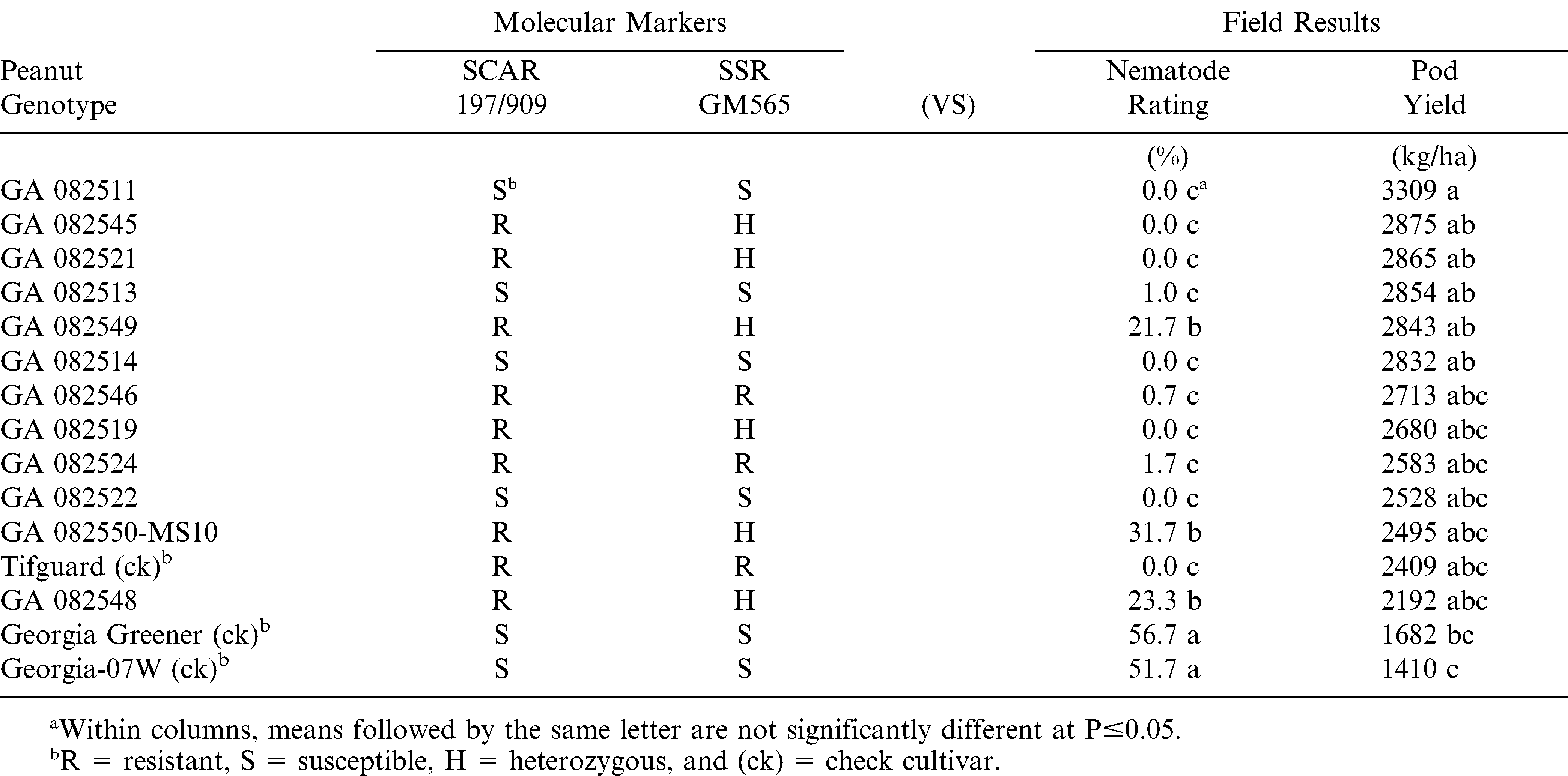
During 2011 at the Rigdon Farm, all three check cultivars were classified correctly by the two RKN molecular markers (SCAR 197/909 and SSR-GM565) with Tifguard being resistant and Georgia-07W and Georgia Greener each being susceptible (Table 1). Three of the 12 advanced Georgia breeding lines (GA 082548, GA 082549, and GA 082550-MS10) had intermediate RKN-resistance in the field. The SCAR 197/909 marker classified these three breeding lines as resistant; instead of, the SSR-GM565 marker classified the same three breeding lines as heterozygous (Table 1). In 2012 at the Rigdon Farm (Table 2), GA 082548 and GA 082549 were not significantly different (P≤0.05) from the RKN-resistant check Tifguard for gall rating and pod yield. Also in 2012 at the Blackshank Farm (Table 3), GA 082550-MS10 was again not significantly different from Tifguard for gall rating and pod yield.
However in 2011 at the Rigdon Farm, four of the 12 advanced Georgia breeding lines (GA 082511, GA 082513, GA 082514, and GA 082522) from the same cross combination had RKN-resistance in the field, but both molecular markers classified the four breeding lines as susceptible (Table 1). Thus, 26.7% among these 15 genotypes were misclassified by both molecular markers as false negatives.
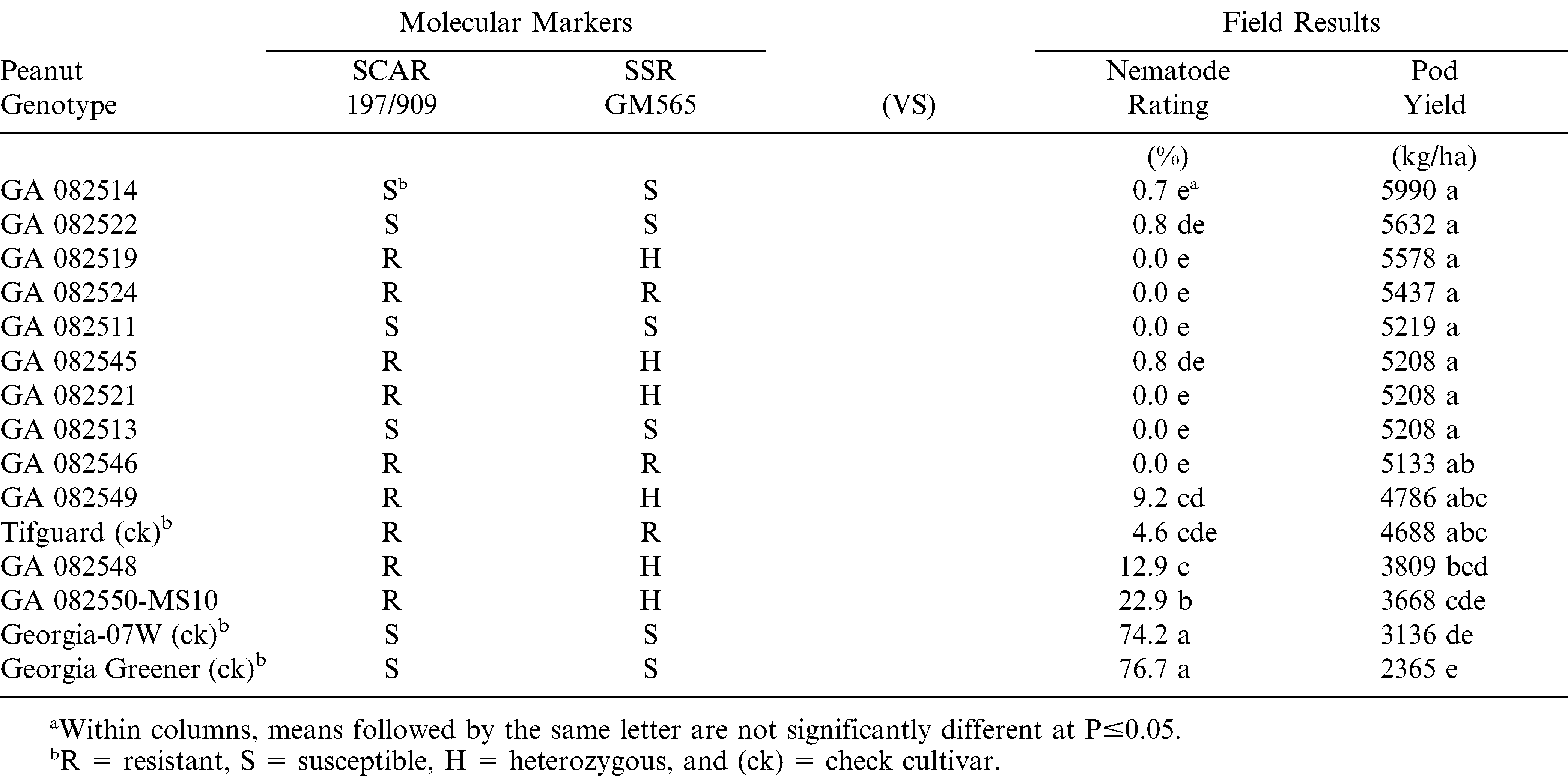
Likewise in 2012 at the same Rigdon Farm field test location, the same results were found (Table 2). All three check cultivars were classified correctly by both markers, but the same four RKN-resistant breeding lines (GA 082511, GA 082513, GA 082514, and GA 082522) were again misclassified as susceptible by both molecular markers.
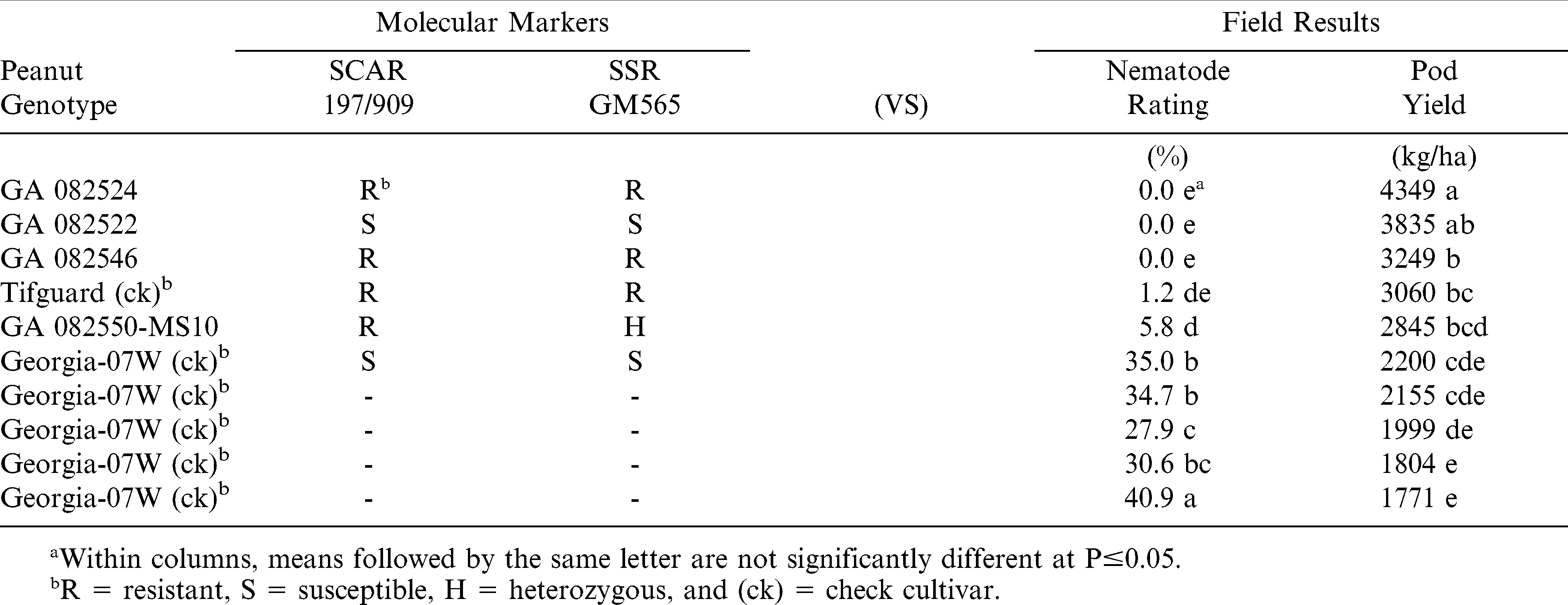
In 2012 at the Blackshank Farm, similar results were also observed (Table 3). This field test location involved fewer genotypes but more replications. The resistant check cultivar Tifguard and several entries of the susceptible check cultivar Georgia-07W were again in agreement with both markers. However, GA 082522 again showed RKN-resistant in the field test but was misclassified as susceptible by both markers.
Only five of the 12 advanced Georgia breeding lines (GA 082519, GA 082521, GA 082524, GA 082545, and GA 082546) were classified as resistant by at least one marker and were found to have very low nematode gall rating and relatively high pod yields (Tables 1 and 2). Two of these breeding lines (GA 082524 and GA 283546) were consistently and correctly classified as RKN-resistant each year by both molecular markers in all three field tests (Tables 1– 3).
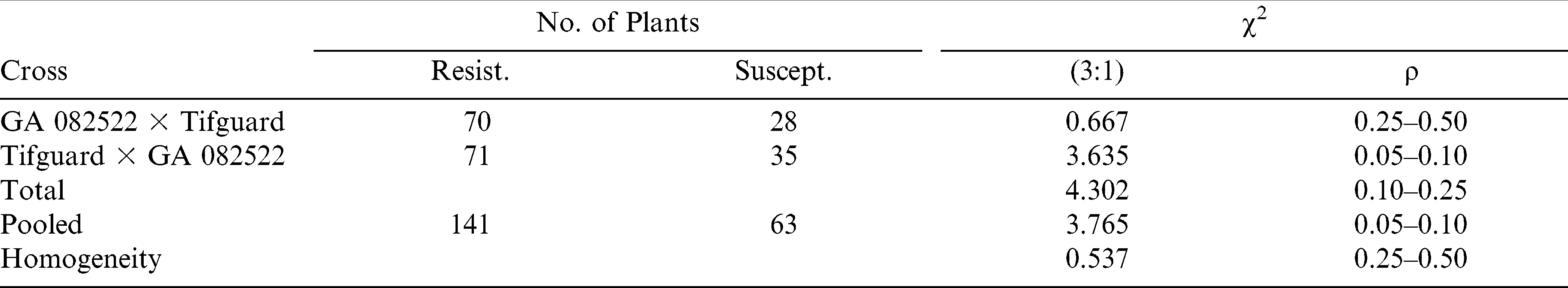
These findings suggest genetic differences among the MAS resistant and MAS susceptible genotypes. Reciprocal cross combinations between MAS resistant Tifguard × MAS susceptible GA 082522 and between MAS resistant Tifguard × MAS resistant GA 082546 resulted in all F1 hybrid plants testing resistant by the SCAR 197/909 marker; whereas, F1 plants were classified as heterozygous as expected with the co-dominant SSR-GM565 marker. So, no maternal or cytoplasmic effects were observed among these reciprocal crosses in the F1 generation.
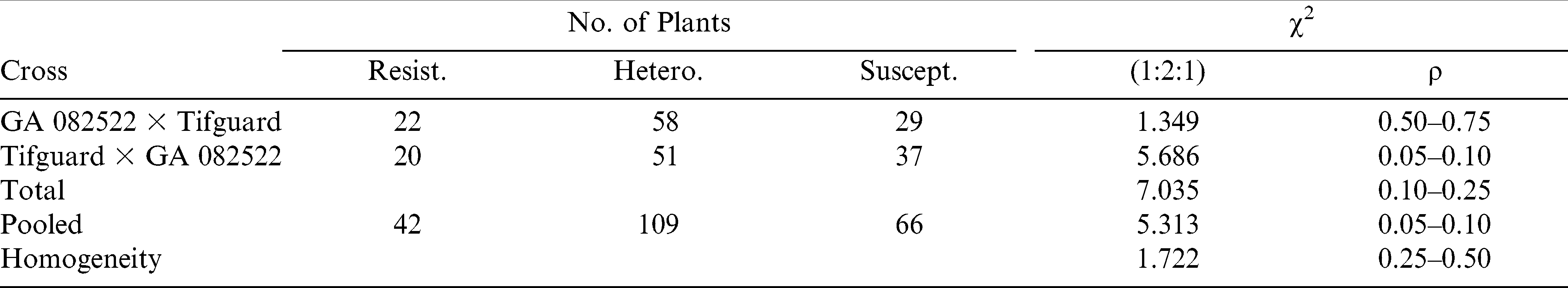
The F2 segregation from the reciprocal cross MAS resistant Tifguard × MAS susceptible GA 082522 fit a 3∶1 resistant to susceptible ratio using the SCAR 197/909 marker (Table 4). Likewise, the F2 segregation from the same reciprocal cross fit a 1∶2∶1 ratio using the SSR-GM 565 marker (Table 5). Total, combined, and homogeneity chi-squared values also agreed with the one-gene genetic ratio of either 3∶1 vs 1∶2∶1 depending upon the molecular marker utilized. These results suggest one-gene difference among these two MAS parental genotypes (Tifguard vs GA 082522).
No F2 segregation was observed in the reciprocal cross of MAS resistant Tifguard × MAS resistant GA 082546. This would be expected since both parental MAS resistant genotypes were classified correctly by these two molecular markers.
Conclusions
The one-gene difference found in the MAS resistant × MAS susceptible reciprocal cross combinations suggest that genetic recombination may have occurred between the two molecular markers (SCAR 197/909 and SSR-GM565) and the RKN-resistant gene, Rma. This lack of linkage could explain the misclassification among four of the 15 genotypes that were found in the three field tests conducted during this 2-yr study (Tables 1 to 3).
Another possibility may also be the identification of a second unlinked nematode resistant gene. Garcia et al., (1996) first proposed two dominant genes, Mae and Mag, conditioning peanut root-knot nematode resistance from a F2 population derived from the interspecific cross of 4× (A. hypogaea X A. cardenasii) = GA 6 and PI 261942. Yet, another possibility may be attributed to random chromosome segregation involving an autotetraploid introgressed large segment with the RKN-resistant gene(s) from the original 3-way [BB × (AA × AA) genome] cross combination as described by Strickberger (1968).
Regardless of the reason, greater than a fourth of the RKN-resistant peanut genotypes would have been missed by using only these two molecular markers in a MAS breeding program. Consequently, the need for molecular markers more tighter-linked with the RKN-resistant gene(s) is now apparent for future marker assisted selection.
Literature Cited
Branch, W.D. 2002 Registration of ‘Georgia-01R’ peanut Crop Sci. 42 : 1750 – 1751 .
Branch, W.D. 2003 Registration of ‘Georgia-02C’ peanut Crop Sci. 43 : 1883 – 1884 .
Branch, W.D. 2007 Registration of ‘Georgia Greener’ peanut J. Plant Reg. 1 : 121 .
Branch, W.D. and Brenneman, T.B. 2008 Registration of ‘Georgia-07W peanut J. Plant Reg. 2 : 88 – 91 .
Chu, Y. Holbrook, C.C. Timper, P. and Ozias-Akins, P. 2007 Development of a PCR- based molecular marker to select for nematode resistance in peanut Crop Sci. 47 : 841 – 847 .
Chu, Y. Wu, C.L. Holbrook, C.C. Tillman, B.L. Person, G. and Ozias-Akins, P. 2011 Marker-assisted selection to pyramid nematode resistance and the high oleic trait in peanut The Plant Genome. 4 : 110 – 117 .
Garcia, G.M. Stalker, H.T. Shroeder, E. and Kochert, G. 1996 Identification of RAPD, SCAR, and RFLP markers tightly linked to nematode resistance genes introgressed from Arachis cardenasii into Arachis hypogaea Genome 39 : 836 – 845 .
Holbrook, C.C. Timper, P. Culbreath, A.K. and Kvien, C.K. 2008 Registration of ‘Tifguard’ peanut J. Plant Reg. 2 : 92 – 94 .
Kokalis-Burelle, N. and Rodriguez-Kabana, R. 1997 Root-knot nematodes In: Compendium of Plant Diseases 2nd Ed Amer. Phytopath. Soc. Press , pp. 45 – 48 .
Nagy, E.D. Chu, Y. Guo, Y. Khanal, S. Tang, S. Li, Y. Dong, W.B. Timper, P. Taylor, C. Ozias-Akins, P. Holbrook, C.C. Beilinson, V. Nielsen, N.C. Stalker, H.T. and Knapp, S.J. 2010 Recombination is suppressed in an alien introgression in peanut harboring Rma, a dominant root-knot nematode resistance gene Mol. Breeding 26 : 357 – 370 .
Norden, A.J. Lipscomb, R.W.. and Carver, W.A.. 1969 Registration of Florunner peanuts (Reg. No. 2) Crop Sci. 9 : 850 .
Simpson, C.E. and Starr, J.L. 2001 Registration of ‘COAN’ peanut Crop Sci. 41 : 918 .
Simpson, C.E. Starr, J.L. Church, , G.T. Burow, , M.D. and Paterson, , A.H. 2003 Registration of ‘NemaTAM’ peanut Crop Sci. 43 : 1561 .
Simpson, , C.E. Nelson, , S.C. Starr, , J.L. Woodard, , K.E. and Smith, , O.D. 1993 Registration of TxAG-6 and TxAG-7 peanut germplasm lines Crop Sci. 33 : 1418 .
Strickberger, M.W. 1968 Chap. 21 Chromosome variation in number pp. 449 – 478 In: Genetics. The Macmillan Co. , New York, NY .
Williams, E.J. and Drexler, J.S. 1981 A non-destructive method for determining peanut pod maturity Peanut Sci. 8 : 134 – 141 .
Notes
- Professor, University of Georgia, Dept. of Crop and Soil Sciences, Coastal Plain Experiment Station, 2360 Rainwater Road, Tifton, GA 31793-5766
- Professor, University of Georgia, Department of Plant Pathology, Coastal Plain Experiment Station, 2360 Rainwater Road, Tifton, GA 31793-5766
- Division Manager, Eurofins STA Laboratories, Inc. Genetic Services, 1821 Vista View Dr., Longmont, CO. 80504 * Corresponding author email: wdbranch@uga.edu
Author Affiliations