Introduction
In the last several years, Salmonella contamination of peanuts and peanut-containing products has resulted in outbreaks of salmonellosis and several product recalls. An outbreak related to peanut butter and subsequently roasted peanuts occurred in 1996 (Scheil, et al. 1998) and in 2001, in-shell peanuts were associated with an outbreak in Australia (Kirk, et al., 2004). In 2006 an outbreak associated with peanut butter occurred in the U.S. and in 2006–2007, peanut butter was associated with an outbreak that resulted in over 600 confirmed cases but no deaths (Centers for Disease Control, 2007). In 2008–2009 in the U.S., peanut butter and peanut butter-containing products containing Salmonella were associated with an outbreak with over 525 confirmed cases and 8 alleged deaths (Centers for Disease Control, 2009). These occurrences demonstrate the critical necessity to adequately reduce Salmonella at every stage of peanut handling, roasting, and post roast processing to control the ingress, spread, and growth of Salmonella in overall processing and packaging environments. In response to these Salmonella outbreaks, the U.S. Food and Drug Administration (FDA) provided a guidance document addressing the risk of Salmonella in foods that contain a peanut-derived product as an ingredient (Food and Drug Administration, 2009). In that document FDA recommended that peanuts and peanuts as ingredients should be subjected to a validated process to adequately reduce the presence of Salmonella. Dry and oil roasting of peanuts are the processing steps that should result in adequate reduction of Salmonella that may potentially be present on raw peanuts.
Studies on the effect of temperature and time protocols on Salmonella in peanut butter have been reported (Ma, et al., 2009; Burnett, et al., 2000; Shachar and Yaron 2006) and generally indicate that an acceptable reduction of the organism in contaminated peanut butter is not possible with current commercial practices. Although peanut butter is the most consumed roasted peanut product, dry and oil roasted peanuts are also consumed in whole, half, or chopped nut forms and elimination of Salmonella on these products is also of food safety concern.
Because of increasing concerns for food safety, processors and manufacturers are being required to provide validation documentation to include microbial challenge studies and/or time and temperature determinations in peanut roasters. Time/temperature determinations are only of value in validation studies if the relationship of time and temperature to reduction of Salmonella or a suitable surrogate are available. Data on the length of time and temperatures required for a 4 or 5 log CFU/g reduction of Salmonella on specific whole commodities is somewhat lacking, although some information is available for almonds (Almond Board of California, 2007a and 2007b). Based on these two studies, the almond industry has provided limited data on the time/temperature protocols resulting in a 4 log CFU/g reduction of S.enteritidis Phage Type 30 (SE PT 30) identified in the 2001 almond outbreak. Data on specific thermal process reduction of Salmonella in peanuts are not currently publically available.
Roaster temperature evaluation is easily accomplished by passing thermocouples through the roaster which are positioned to provide coverage across the roaster belt and throughout the bed of peanuts (unpublished). Interpretation of roaster temperature data relative to Salmonella reduction is currently inappropriate because time and temperature information on reductions of Salmonella in dry and oil roasting of peanuts is unavailable. Information on Salmonella reductions in peanuts resulting from various combinations of time and temperature are needed. Additionally, information on Salmonella reduction in commercial peanut roaster operations which include not only time and temperature, but also, bed depth and airflow are also needed.
Prevalence of Salmonella on raw, grade samples of peanuts encompassing all growing areas over three crop years (2008–2010) was estimated at 2.33% (Calhoun, et al., 2010). The concentration levels of Salmonella in positive samples, as determined by a most-probable-number (MPN) assay, were <0.03 to 2.4 MPN/g. (Calhoun, et al., 2010). In comparison, the isolation frequency for Salmonella ranged from 0.6–1.5% over four years (2001–2004) in raw almonds sampled from throughout California (Danyluk, et al., 2006a and 2006b). When detected, levels were 1.2 to 2.9 MPN/100 g. On a more detailed level, an outbreak of salmonellosis associated with consumption of raw almonds in 2000 to 2001 resulted in an isolation frequency of 84% in fifty 22.7-kg boxes of recalled almonds and MPN of 8.5 ± 1.3 MPN/100 g (Danyluk, et al., 2006a and 2006b). Bansal et al. (2010) found the prevalence of Salmonella on kernels and inshell almonds to be 1.6 and 0.9%, respectively, in 2006, and 0.83 and 2.2%, respectively, in 2007. When detected, levels were 1.4 to 15.5 MPN/100 g (average 2.3 MPN/100 g) or 1.4 to 18.3 MPN/100 g (average 2.1 MPN/100 g) using two different methods of MPN determination.
In response to the peanut butter contamination incident of 2006–2007 (CDC, 2007), the American Peanut Council contracted with Deibel Laboratories (Gainesville, FL, 32606) to conduct studies to examine the effect of various time and temperature protocols on the reduction of Salmonella on peanuts. The objective of that work, reported herein, was to examine various time and temperature parameters for reduction (4 log CFU/g) of Salmonella using either dry or oil roasting. Ancillary to this objective, determination of appropriate hold temperature of inoculated peanuts was also investigated. In addition, parallel experiments were performed to establish that the previously investigated (Almond Board California, 2007b) surrogate, Enterococcus faecium, had similar or greater heat resistance as Salmonella and would thus be a suitable non-pathogenic surrogate for peanut processing plant validation studies. This organism was tested and approved for use in validation of a 4 log CFU/g reduction in almond processing (Almond Board California, 2007b). A minimum 4 log CFU/g reduction level in this peanut study was selected based on the mandated 4 log CFU/g reduction of Salmonella previously set for almonds.
Materials and Methods
Sample Materials
Peanuts used in the study were unblanched, runner-type; unblanched virginia-type and blanched virginia-type obtained from a commercial sheller. Varieties were not identified. Runner-type peanuts are the most commonly grown and most commonly used to manufacture peanut butter. Virginia-type peanuts are large peanuts commonly found in cocktail nuts or roasted in the shell. Although all types of peanuts may be used in peanut butter the range of sizes utilized in the study is representative of all market types to include spanish and valencia market types. Unblanched and blanched refer to the presence or absence, respectively, of the peanut seed coat. Peanut seed coat removal (blanching) is accomplished by gentle abrasion after low heat application. An intact seed coat serves as a barrier to air and/or moisture into the seed lumen (area between the cotyledons). Removal of the seed coat may result in increased surface area during inoculation due to increased access to the lumen and cotyledon internal faces.
Peanuts used in the study contained 48.5 ± 1.0% oil as determined by NMR (mq Minispec, Bruker Optics, Houston, TX) and moisture content for nonblanced peanuts was 7.0 ± 0.5% while blanched peanuts contained 6.5 ± 0.5% moisture. After-roast moisture content for all samples ranged from 1–2% as is common in roasted peanuts. All moisture contents were calculated from original and oven dried (130 C for 3 hr) weight of each sample.
Salmonella enterica servovars used in the study were S. Enteritidis PT 30, ATCC 1045 (almond outbreak organism used for Almond Board of California (ABC) validation studies); S. Tennessee (2006/2007 peanut outbreak strain); S. Typhimurium TM-1 (Reference strain employed for cooking regulations, USDA Appendix A); S. Newport C2:e,h:1,2 (Deibel Labs culture collection); S. Cubana G2:229 (Deibel Labs culture collection); S. Redba (Deibel Labs culture collection); and S. Bredeney (Deibel Labs culture collection).
Enterococcus cultures used in the study were E. faecium ATCC 8459 (NRRL B-2354 used in Almond Board of California studies) and E. faecium ATCC 35667. Throughout the manuscript, references to Salmonella and Salmonella inoculums or E. faecium inoculums in regard to testing in this study refer to the mixture of the Salmonella servovars and the E. faecium cultures, respectively, as described above. Appropriate sterile conditions were utilized in culture, inoculation, and enumeration procedures.
Inoculum Procedure
All media utilized were obtained from DB Diagnostics (Sparks, MD 21152). All cultures were grown in Tryptic Soy Broth at 35 C for 24 hrs. The broth cultures (1.0 ml /plate) were then spread over 15mm × 150mm plates of Tryptic Soy Agar (TSA) Broth and incubated for 24 hrs at 35 C. After incubation, 5–6 ml of 0.1% peptone was added to each plate and the bacterial lawn loosened with a sterile spreader. Sterile pipettes were used to collect the loosened cells from five plates and the collections were pooled into a 25 ml inoculum preparation. Each 25 ml preparation was used to inoculate a 400g batch of peanuts.
Five 400g peanut batches of each peanut type were prepared with either mixed Salmonella inoculum or mixed E. faecium inoculum. Separate 400g batches of each peanut type were weighed into 30.3cm × 30.5cm Cryovac (Duncan, SC 29334) bags and 25 ml mixed inoculum was added. The bags were closed and mixed by hand by repeated inversion for 1 minute and the inoculated peanuts were poured out onto filter paper over a metal rack in a sterile plastic tub. The inoculated peanuts in tubs were loosely covered with sterile cheesecloth and allowed to dry for 24 hrs at room temperature (about 24 C). Prior to the roasting tests, six 25 g samples of each batch were plated onto appropriate agars to confirm that the inoculation level was at least 7 log CFU/g. The agars employed were TSA and Bismuth Sulfite Agar (BSA) for Salmonella and Dextrose Tryptone/Yeast Extract Agar (DTYE) and Kenner Fecal agar supplemented with Triphenyltetrazoliumchloride (TTC) agar (KF) for E. faecium. Moisture content of peanuts did not increase as a result of the exterior application of 25 ml of inoculum onto 400 g of peanuts. Visual observation indicated that water from the inoculum application of 1 min was quickly evaporated or was absorbed by the filter paper onto which the inoculated peanuts were poured.
The same protocol was utilized to conduct preliminary evaluations of Salmonella and E. faecium survival after various holding temperatures before roasting. Unblanched, virginia-type peanuts were inoculated with Salmonella (7.84 log CFU/g) and E. faecium (7.81 log CFU/g) and held at 4.4, 10.0, 15.5 and 21.1 C for 24 hrs. Peanuts from all hold temperatures were subsequently oil roasted for 1.5 min at 140 C and dry roasted for 10 min at 149 C before enumeration.
Ovens and Thermocouple Devices
For dry roasting experiments, a Fisher Scientific (Waltham, MA 02454) Isotemp 851F convection oven was utilized and for oil roasting, an Oster (Fort Lauderdale, FL 33301) commercial oil roaster was modified with a circulating device. All temperature measurements (5 sec intervals) were recorded with a Multichannel Occurrent Logger Evaluation unit (Model Xpert-Ready Gold, ECD Corp., Milwaukie, OR).
Roasting Temperatures and Times
For each experiment, five or more minutes after the oven had reached the set temperature or oil temperature had stabilized to the set temperature, 2.5 cm diameter × 7.5 cm tall, cylindrical, aluminum wire baskets containing 25 g of each type of inoculated peanuts were placed in the oven or oil roaster. Inoculated samples were held at 4.4 C prior to roasting and exposure time to a given set temperature was initiated based on preliminary studies to determine the length of time for internal temperatures to “come up” to the oven set point. Preliminary determinations of “come up” times were conducted in triplicate for all times and temperatures by sealing a thermocouple tip into each respective peanut type and placing the sealed peanut in the center of the basket prior to placing the basket into the oven or oil. The approximate time for the peanuts to reach the oil temperature in the circulating bath was one (1) min for all three peanut types. The approximate “come-up” times for the respective peanut types were 3.5 min for the blanched virginia-type, 4.0 min for the unblanched runner-type and 5.0 min for the unblanched virginia-type. During all tests, “come-up” times were allowed before predetermined roasting times were initiated. Dry roasting temperatures evaluated for Salmonella and E. faecium reduction on peanuts were 129, 138, 146, 154, and 163 C with times ranging up to 60 min at the lower temperatures and as low as 10 min for the higher temperatures. Oil roasting temperatures were 120, 130, 140, 150, and 160 C with times ranging up to 5 min for the lower temperature and as low as 0.5 min for the highest temperature. Each dry and oil roasting temperature and time protocol was evaluated in triplicate for each peanut type. After each heat treatment, the wire baskets were removed and the peanuts were immediately placed into sterile Cryovac bags in an ice bath to halt thermal lethality.
Enumeration of Treated Peanuts
After cooling, the treated samples were enumerated immediately by transferring the 25 g portion into 50 ml of sterile buffer in a heavy duty Cryovac bag, stomaching for 2 min, sitting stationary for 2 min, shaking and diluting, and spread plating in 0.1 ml aliquots onto the respective plating medium described previously. Duplicates from each of the three replications were incubated at 35 C for 48 hrs and counted. Mean organism counts were converted to log CFU/g. Limit of detection of the enumeration methods with an initial dilution of 25/75 or 1/3 (25 g peanuts and 50 g buffer) and spread plating 0.1 ml was the log of 0.0333 or 1.48. Log CFU/g reductions were calculated by subtracting the log CFU/g of the mean of survivor counts from the log CFU/g of organisms on the inoculated peanuts. Preliminary experiments to compare the Stomacher method and a surface wash method were conducted.
Statistical Analysis
Data were analyzed with Statistical Analysis System (version 8.2) software (SAS Institute, Cary, NC). Analysis of variance with the general linear models (GLM) procedure and Duncan's multiple range tests were used to identify the significant differences among sample means (p < 0.05).
Results and Discussion
Inoculated Peanut Hold Temperature
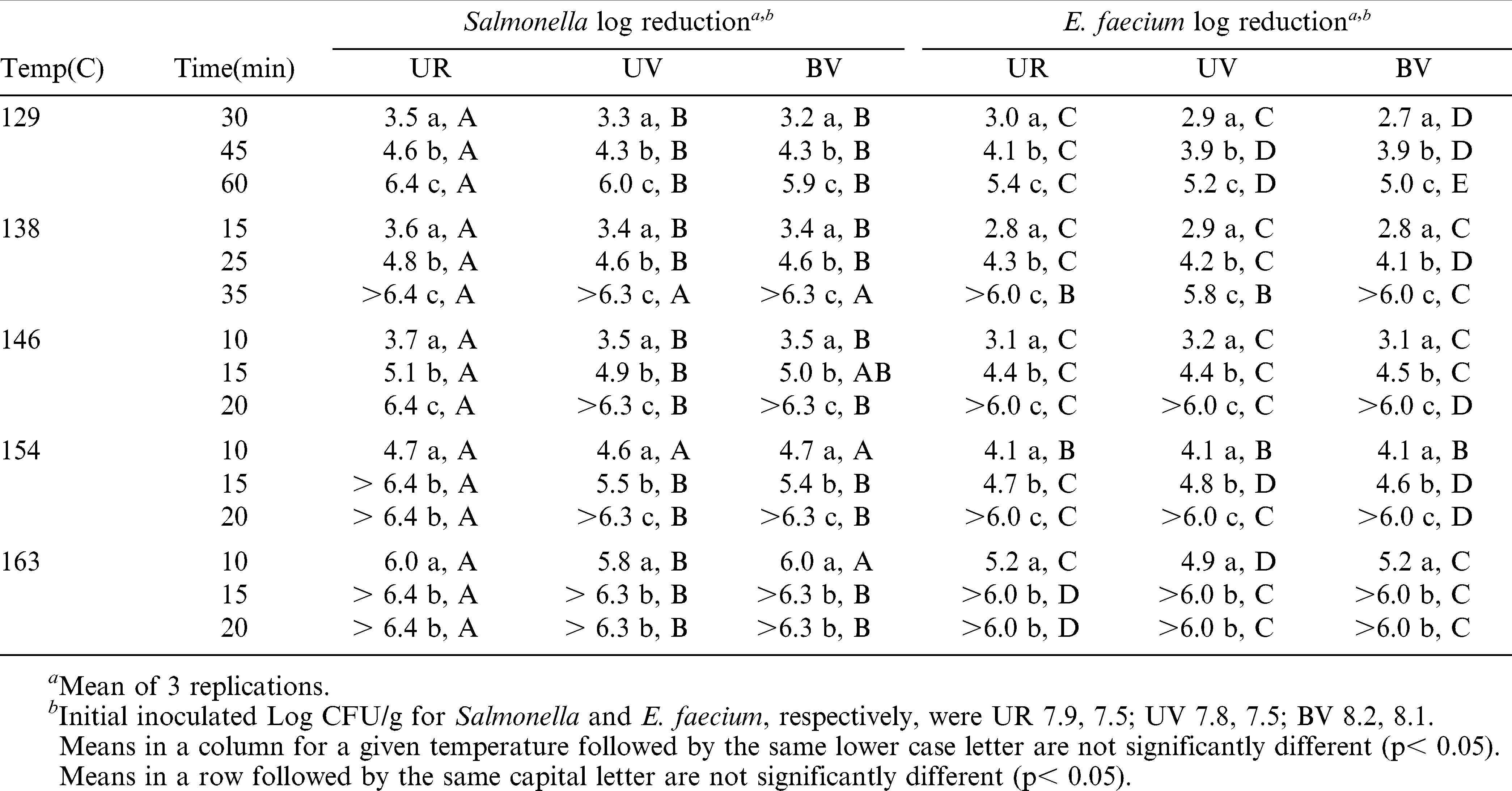
The effect of inoculated peanut temperature before roasting could be a factor in accurate determination of the thermal reduction of the mixed inoculums of both Salmonella and E. faecium . When inoculated, raw peanuts were held at 4.4, 10.0, 15.5 and 21.1 C for 24 hr before oil roasting, peanuts from all hold temperatures roasted for 1.5 min at 140 C resulted in a post roast mean of <1.48 log CFU/g (limit of detection) of Salmonella for all temperatures and a range of 2.36–2.81 log CFU/g of E. faecium over all hold temperatures (Table 1). Similarly, dry roasting of peanuts from each hold temperature for 10 min at 149 C resulted in a log CFU/g of Salmonella range of 3.41–3.90 and log CFU/g of E. faecium range of 3.70–3.96 (Table 2). Generally, significantly greater survival of both organisms was observed at the lowest or lower hold temperatures and subsequently all tests were conducted with inoculated peanuts held at a temperature of 4.4 C before roasting.
Dry Roast
The oven “come-up” times for the various peanut types varied from 3.5–5 min because of the hold temperature of 4.4 C and in relation to moisture content and size of the peanuts. The blanched virginia-type peanuts contained the lowest moisture content due to the heat applied during blanching and had the shortest come up time (ca.3.5 min). The unblanched runners “come up” time was ca. 4.0 min and the unbalanced virginia-type was ca. 5 min. Lower moisture content in the blanched sample resulted in shorter come up time because there was less water to be removed during roasting and thus a shorter time of evaporative cooling. Intact seed coats (nonblanched) may also retard moisture removal and increase equilibration (“come up”) time. The difference between ca 3.5–5 min is relatively small compared to potential differences that occur in commercial dry roasters in which heat is applied to peanuts for a total time of 35–45 min (personal communication) although few peanuts enter a commercial roaster at 4.4 C.
In separate testing with medium runner peanuts of the same moisture content, peanuts at room temperature (ca. 25 C) varied little from oven temperatures from beginning to end of a 15 min oven roast at 165 C. (data not presented) The use of 4.4 C as a starting temperature was based on survival of microorganisms and resulted in a much slower set point temperature response as indicated from 3.5–5 min “come up” times.
At each dry roast combination of temperature and time in the study, Salmonella and E. faecium reductions were 2.7 log CFU/g or greater (Table 3). For each temperature setting, increasing time resulted in highly significant (P< 0.001) differences in final log CFU/g reductions. At 129 C, 30–45 min were required to achieve a 4 log CFU/g reduction of Salmonella and E. faecium. At 138 C, between 15–25 min of exposure were required for a 4 log CFU/g reduction of both organisms. At 146 C, 10 minutes exposure resulted in a 3.5–3.7 log CFU/g reduction of Salmonella and 3.1–3.2 log CFU/g reduction of E. faecium. After 15 min the reductions for both organisms were 4.4 log CFU/g or greater. Temperatures of 154 and 163 C provided a 4.1 log CFU/g reduction or higher of both organisms. Salmonella reductions at 163 C for 10 min were 5.8 log CFU/g or higher. This reduction suggests that exposure to 163 C for a time period somewhat less than 10 min should provide a 4 log CFU/g reduction. In both dry and oil roasting (Tables 3 and 4) reductions of E. faecium were less than those of Salmonella. The use of E. faecium as a surrogate organism in commercial roasting evaluations should provide a positive margin of error in estimating reduction of Salmonella.
The market type and blanched condition of peanuts did not appear to affect the temperature/time required to achieve a given log CFU/g reduction of Salmonella or E. faecium inoculated onto the peanuts.
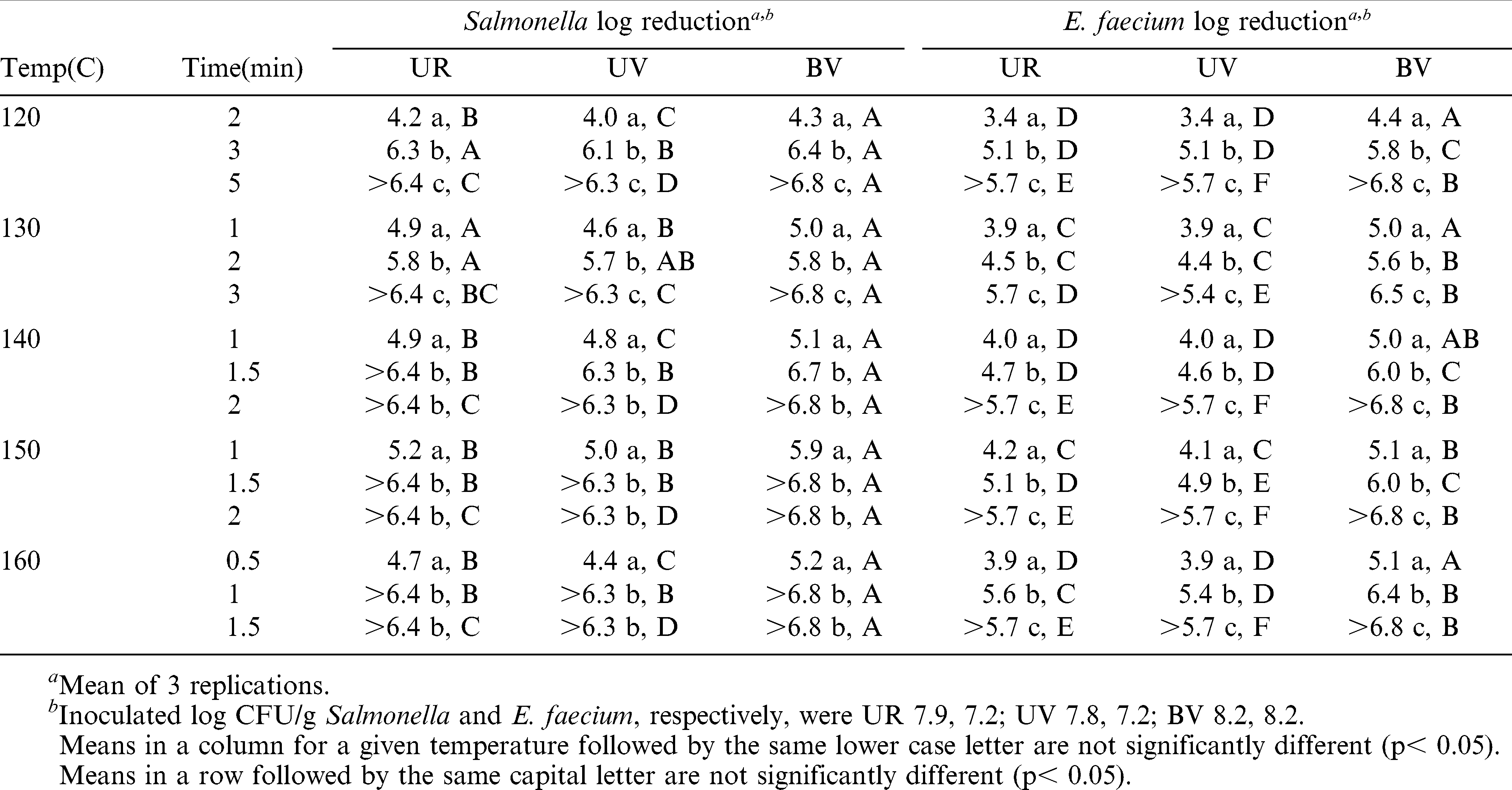
Oil Roast
Because of the thermal conductivity of the oil medium (ca. 0.17) compared to air (ca 0.025), and a rapid release of water vapor (thermal conductivity ca 0.16) at the peanut surface during the initial heating period, oil roast processing is more effective in reducing microorganisms than air roasting (The Engineering Toolbox, 2013). For the same reasons, (as well as an initial temperature of 4.4 C) the “come up” time in oil roasting was much less than for dry roasting. The come up time for peanuts to reach the oil temperature in the circulating oil bath was approximately one (1) min for all three peanut types. For each temperature setting, adjusting the time of oil roasting resulted in significant differences (p<0.0001) among final log CFU/g reductions (Table 4). All protocols resulted in a 4.0 log CFU/g reduction of Salmonella and most resulted in a 5 log CFU/g or greater reduction. E. faecium reductions were consistently less than those for Salmonella and were greater than 5 log CFU/g at the longest (or next to longest) time for all temperatures.
Oil roasting in these studies was similar to commercial oil roasting in that peanuts were place in oil for a time then removed. Commercial oil roasters use a moving belt to transport peanuts into and out of the oil. Normal oil exposure times in commercial oil roasters are 4–5 min and oil temperatures are in excess of 150 C for the entire process. Salmonella reduction in all peanut types was >6 log CFU/g at 150 C for 1.5 and 2 min and at 160 C for 1 and 1.5 min. Similarly, reduction of Salmonella on inoculated almonds oil roasted at 127 for 30 sec was 3.6 log CFU/g but Salmonella could not be recovered by enrichment of 1g samples after almonds inoculated at 5 log CFU/g were exposed to oil at 127 C for 1.5 min (Du, et al., 2010). Commercial oil roasting times and temperatures that achieve acceptable kernel color and texture resulted in much greater than 5 log CFU/g reductions of Salmonella in both almonds and peanuts.
The experimental protocols of both dry and oil roasting experiments incorporated the use of an ice bath to rapidly halt thermal lethality in peanuts removed from the roasting environment. This was done to provide more accurate information concerning thermal destruction achieved at the identified target temperatures. In commercial roasting, peanuts do not enter the roaster at very low temperatures and they are cooled more slowly with ambient temperature air after exposure to roasting temperatures. Thus, for a given temperature in these laboratory studies, commercial processing using those temperature settings expose peanuts to that temperature and temperatures decreasing slowly from that temperature for longer times than those use in these laboratory experiments.
Summary and Conclusions
These studies were conducted to provide general information on potential Salmonella and E. faecium reductions at temperatures somewhat below and above temperatures normally encountered in commercial peanut roasting. These data should be referenced by industry only as general guidelines since results obtained from specific manufacturing plant production processes may vary. Equipment, equipment function and raw peanut composition may influence the achieved Salmonella reduction in any particular set of conditions. Commercial processors should verify and monitor bed depth, air flow, and belt speed in commercial roasters to assure that heat is applied as uniformly as possible across and within the peanut bed. Processors should confirm oven temperature in relation to set point, temperature variability within the roaster, and microorganism reduction through an effective microbiological validation testing program for finished roasted peanuts.
Acknowledgements
This study was funded by the American Peanut Council, Alexandria, VA and analytical operations were performed at Deibel Labs, Gainesville, FL. The assistance of Keith Hendrix, USDA, ARS, MQHRU is gratefully acknowledged.
Literature Cited
Almond Board of California (ABC) , 2007a Guidelines for Validation of Dry Roasting Processes Available at http://www.almondboard.com/Handlers/Documents/Dry-Roast-Validation-guidelines.pdf. Accessed April 28, 2014 .
Almond Board of California (ABC) , 2007b Guidelines for Process Validation Using Enterococcus faecium NRRL B-2354 Available at http://www.almondboard.com/Handlers/Documents/Enterococcus-Validation-Guidelines.pdf. Accessed April 28, 2014 .
Bansal A Jones T.M Abd S.J Danyluk M.D and Harris L.J 2010 Most-probable-number determination of Salmonella levels in naturally contaminated raw almonds using two sample preparation methods J Food Prot. 73 : 1986 – 92 .
Burnett S.L Gehm E.R Weissinger W.R and Beuchat L.R 2000 Survival of Salmonella in peanut butter and peanut butter spread J. Appl. Microbiol. 89 : 472 – 477 .
Calhoun S Post L Warren B Thompson S and Bontempo A.R 2013 Prevalence and concentration of Salmonella on raw shelled peanuts in the United States J Food Prot. 76 : 575 – 579 .
Centers for Disease Control and Prevention [CDC] , 2007 Multistate outbreak of Salmonella serotype Tennessee infections associated with peanut butter—United States, 2006–2007 Morb. Mortal. Wkly. Rep. 56 : 521 – 524 .
Centers for Disease Control and Prevention [CDC] , 2009 Multistate outbreak of Salmonella infections associated with peanut butter and peanut butter-containing products - United States, 2008–2009 Morb. Mortal. Wkly. Rep. 58 : 1 – 6 .
Danyluk M.D Jones T.M Abd S.J Schlitt-Dittrich F Jacobs M and Harris L.J 2006a Prevalence and amounts of Salmonella found on raw California almonds J. Food Prot. 70 : 820 – 827 .
Danyluk M.D Harris L.J and Schaffner D.W 2006b Monte Carlo simulations assessing the risk of Salmonellosis from consumption of almonds J. Food Prot. 69 : 1594 – 1599 .
Du W.-X Abd S.J McCarthy K.L and Harris L.J 2010 Reduction of Salmonella on Inoculated Almonds Exposed to Hot Oil J Food Prot 73 : 1238 – 46 .
Food and Drug Administration [FDA] , 2009 Measures to address the risk for contamination by Salmonella species in food containing a peanut-derived product as an ingredient Available at: http://www.fda.gov/Food/GuidanceComplianceRegulatoryInformation/GuidanceDocuments/ProduceandPlanProducts/ucm115386.htm. Accessed 27 Jan 2012 .
Kirk M.D Little C.L Lem M Fyfe M Genobile D Tan A Threlfall J Paccagenella A Lightfoot D Lyi H McIntyre L Ward L Brown D.J Surnam S and Fisher I.S.T 2004 An outbreak due to peanuts in their shell caused by Salmonella enterica serotypes Stanley and Newport – sharing molecular information to solve international outbreaks Epidemiol. Infect. 132 : 571 – 577 .
Ma L Zhang G Serner-Smidt P Mantripragada V Ezeoke I and Doyle M.P 2009 Thermal inactivation of Salmonella in peanut butter J. Food Prot. 72 : 1596 – 1601 .
Scheil W Cameron S Dalton C Murray C and Wilson D 1998 A south Australian Salmonella Mbandaka outbreak investigation using a database to select controls Aust. N. Z. J. Public Health. 22 : 536 – 539 .
Shachar D and Yaron S 2006 Heat tolerance of Salmonella enterica serovars Agona Enteritidis, and Typhimurium in peanut butter J. Food Prot. 69 : 2687 – 2691 .
The Engineering Toolbox , 2013 Thermal conductivity of some common materials and gases Available at: http://www.engineeringtoolbox.com/thermal-conductivity-d_429.html. Accessed December 2, 2013 .
Notes
- Research Leader and USDA Professor, USDA, ARS, Market Quality and Handling Research Unit, Raleigh, NC, 27695; Consultant, American Peanut Council, Alexandria, VA 22314, respectively * Corresponding author’s E-mail: tim.sanders@ars.usda.gov
Author Affiliations