Introduction
Among the soilborne diseases of peanut (Arachis hypogaea L.), Sclerotinia blight, caused by Sclerotinia minor Jagger and Cylindrocladium black rot (CBR), caused by Cylindrocladium parasiticum Crous, Wingfield & Alfenas, are important in some peanut production regions of the USA. Individually, each of these diseases can cause annual yield losses of 5–10% (Phipps, 2006), but losses may go up to 50% or more in problem fields (Melouk and Backman, 1995). These two diseases are of great concern to growers as they are difficult or expensive to control. For example, in 2004, about 45% of NC peanut acreage was fumigated for CBR at a cost of approximately $75/ha (Brandenburg et al., 2005). Similarly, chemical sprays to control Sclerotinia blight are very expensive and cost about $100/ha for each application (Hollowell et al., 2008). Moreover, the residue from the chemical sprays is a food safety and environmental issue. In addition to these concerns, seed quality is affected by the two diseases and results in further losses to the growers. Consequently, the most logical and economical option is to provide the growers with high-yielding cultivars which have resistance to both diseases. However, combining resistances has been difficult and cultivars have only been released with resistance to one or the other disease. For example, in the Virginia-Carolina area, a moderately resistant CBR cultivar, NC 12C (Isleib et al., 1997) was found to be susceptible to Sclerotinia blight. Since growers employ resistant cultivars as an important component of managing these two soilborne diseases along with crop rotation and soil fumigation, cultivars with resistance to both these diseases are desired.
Numerous plant introductions within A. hypogaea have been identified as sources of disease resistances (Dong et al., 2008; Holbrook and Dong, 2005; USDA, 2007). Use of these sources led to the release of CBR and Sclerotinia blight resistant lines (Branch and Brenneman, 2012; Chamberlin and Melouk, 2009). However, peanut breeders are constantly looking for new sources of multiple disease resistances. Wild Arachis species from section Arachis, which cross hybridize with A. hypogaea, have been promoted as sources of resistance to many peanut pests and pathogens (Stalker and Moss, 1987; Subrahmanyam et al., 1985; Stalker and Simpson, 1995; Holbrook, 2001). Developing cultivars with resistance to both CBR and Sclerotinia blight has been a long term goal of the breeding program at N.C. State Univ. The specific objective of this study was to identify new sources of resistance to CBR and Sclerotinia blight among the cross-compatible Arachis species. Greenhouse protocols were used to screen a selected list of Arachis species in 2010 to meet this objective.
Materials and Methods
Plant Material
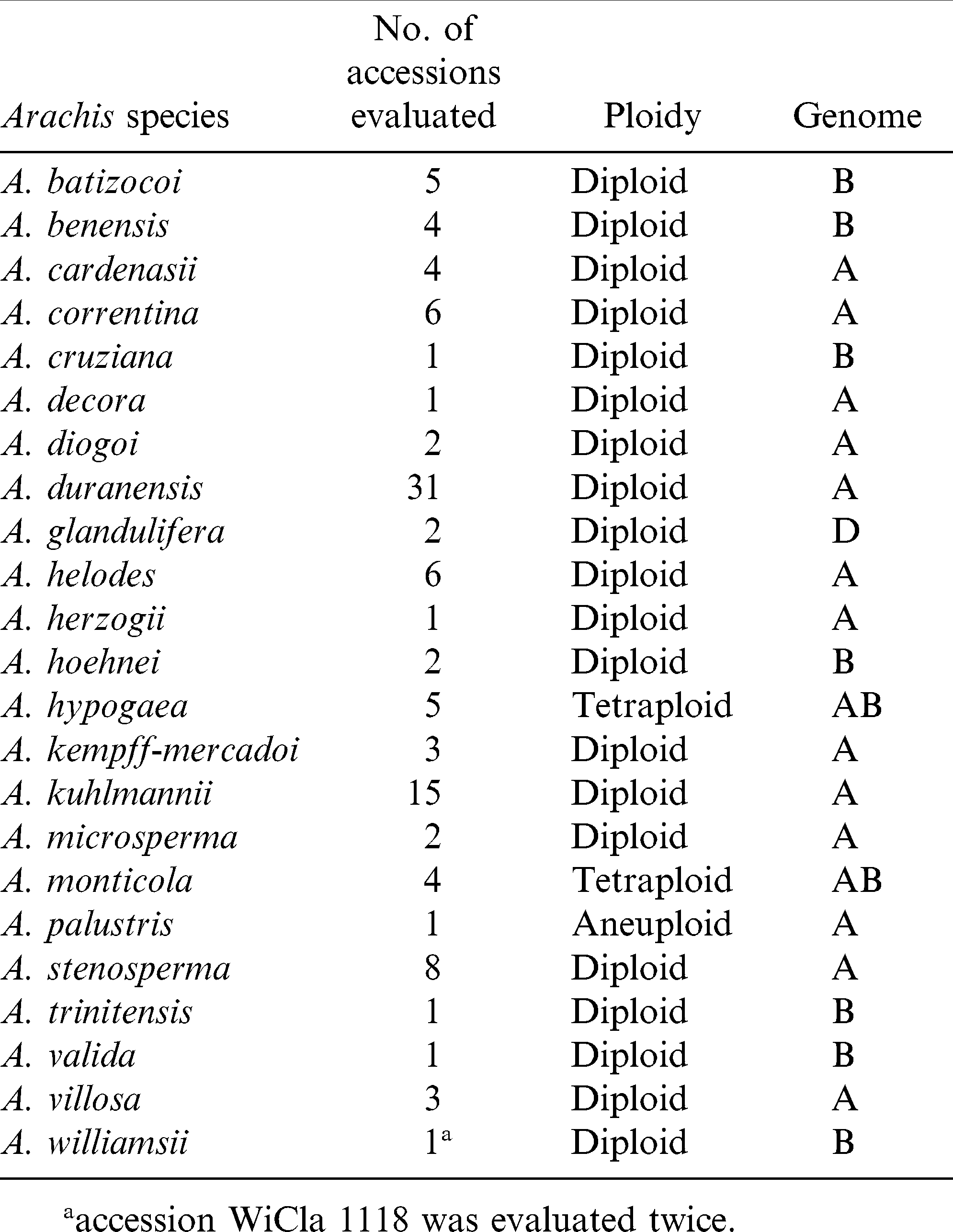
Twenty three Arachis species including A. hypogaea, consisting of a total of 110 genotypes were tested in the greenhouses at N. C. State Univ. in the spring of 2010. The Arachis species with the number of accessions of each were listed in Table 1. Seeds from two different sources of the same accession of A. williamsii (WiCla 1118), A. benensis (Wi 860), and A. duranensis (ScBo 15101) were duplicated in this study. The A. hypogaea lines used as checks included three cultivars and a Sclerotinia blight resistant germplasm line, N96076L (Isleib et al., 2006). The cultivars included two susceptible checks, Gregory (Isleib et al., 1999), and NC 12C (Isleib et al., 1997); and VA 98R (Mozingo et al., 2000) served as a moderately resistant check. Among the cultivars, VA 98R was also duplicated from a different seed source.
Sclerotinia blight and CBR assays were performed in separate experiments under similar greenhouse conditions (23–27C). All experiments were conducted as incomplete block designs with four replications for Sclerotinia blight and six replications for CBR. The inoculation protocols and data collection were exactly as described in Hollowell et al. (2008).
Greenhouse Evaluation of Sclerotinia Blight Reaction
Plants were grown in 10-cm clay pots containing a planting medium of two parts (v∶v) steamed commercial topsoil to one part MetroMix 200 (Sun Gro Horticulture, Bellevue, WA) for 8 wk prior to inoculation. The inoculum was prepared by growing S. minor isolate P13, which was originally isolated from a diseased peanut plant in Chowan County, NC, on potato dextrose agar (PDA) for 2 d. A plug of PDA colonized by S. minor with mycelium side down was placed in a 00 BEEM embedding capsule (Ted Pella, Inc., Redding, CA) with the cap removed and was gently pushed onto a freshly cut petiole on the primary branch of the plant. Inoculated plants were placed on a moisture-retaining mat on the greenhouse bench and were misted daily for 1 min every 2 hr between 6 AM and 8 PM. The bench area was enclosed on the sides and top with plastic sheeting over a PVC frame. After 48 h of incubation, the top cover was removed, but misting continued. Lesion length (mm) was measured 4, 5, 6, and 7 d after inoculation with a digital caliper (Empire Level MFG. Corp., Mukwonago, WI), and area under the disease progress curve (AUDPC) was calculated (Shaner and Finney, 1977).
Greenhouse Evaluation of CBR Reaction
Seeds were planted in plastic cone-tainers, 3.81 cm dia and 20.96 cm in length (Stuewe & Sons, Inc, Corvallis, OR), with a cotton ball placed in the bottom to serve as a wick for water, then filled with a planting medium of two parts (v∶v) steamed commercial topsoil, and one part MetroMix 200. The medium was artificially infested with 25 microsclerotia of C. parasiticum per g of medium at the time it was mixed. Four isolates of C. parasiticum originally obtained from diseased peanut grown in North Carolina were maintained in culture on PDA. Inoculum was prepared by transferring cultures to PDA and incubating at room temperature for 6 wk in the dark. Microsclerotia from cultures of each of the four isolates were recovered using a blender and water, sieved separately, later mixed in a water suspension and quantified (Black and Beute, 1984; Phipps et al., 1976).
Cone-tainers in racks were placed in plastic trays, and plants were grown for 8 wk in the greenhouse. Water in each tray was maintained at a height of approximately 10 cm (approx. 3 cm from the bottom of the tubes) to provide adequate soil moisture. The root system of any plant that died before data collection was washed and plated on PDA to determine whether C. parasiticum was present in the decaying roots. At 60d, surviving plants were removed from the cone-tainers, and the roots were washed and rated for degree of decay on a 0–5 proportional scale (0 = no lesions or decay, 1 = few lesions on secondary roots and / or a few small lesions on taproot, 3 = many lesions on secondary roots and many lesions on the taproot and with several secondary roots missing, 5 = completely decayed roots with most secondary roots and part of taproot missing, with 2 and 4 = intermediate levels of severity, respectively (Black and Beute, 1984; Rowe and Beute, 1975). Any plant that died prior to the end of the experiment and was confirmed to have been infected with C. parasiticum received a score of 5. Any plant that died early, but did not harbor the fungus was considered a missing value.
Data Analysis
Lesion lengths, AUDPC values, and root rot scores from the greenhouse experiments were subjected to analysis of variance for the incomplete block design using the general linear models procedure (PROC GLM) of SAS v8.0 statistical software (SAS Institute, Cary, NC). If the effects of blocks within reps were not significant at P<0.05, then blocks were dropped from the model and the data were analyzed as if they came from a randomized complete block experiment. The AUDPC values were log transformed to stabilize the error variance among the means and adjusted means were computed using the final model. For the duplicated species accessions, contrasts of means were constructed to determine if the duplicated sample means are different from each other.
Results and Discussion
Reaction of Arachis Species to Sclerotinia Blight
Petioles of plants inoculated with S. minor in the greenhouse exhibited water-soaked symptoms after 24 hr, and lesions were found on stems by 3 to 4 d after inoculation. In some cases, the petiole of an inoculated plant became water soaked, but no lesion developed. Occasionally, petioles and capsules fell off due to the weakening of petioles after attack by the fungus. Usually the fungus had already invaded the stem, and lesions developed as observed on other plants. Otherwise, data for the plant was recorded as missing (Hollowell et al., 2008).
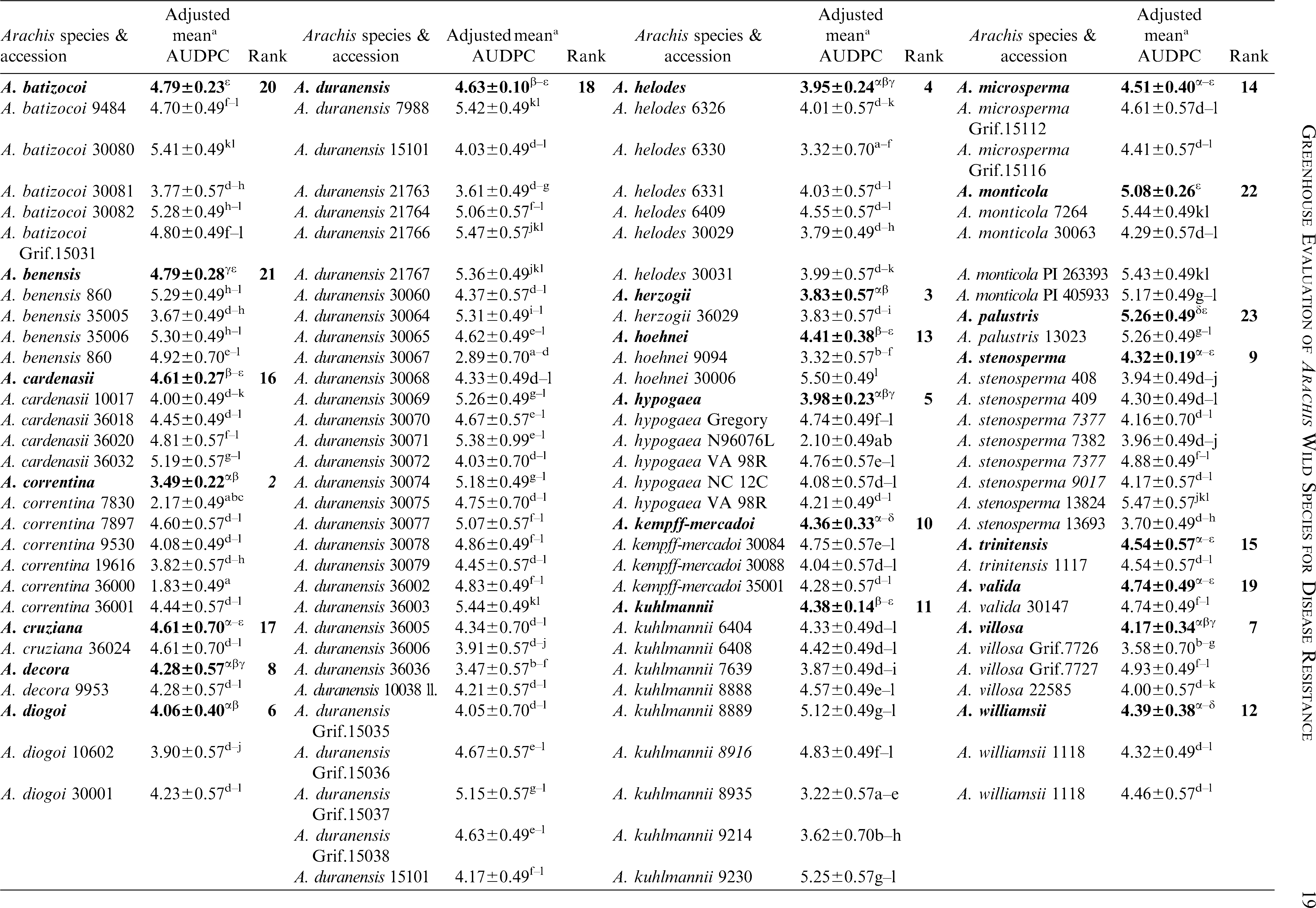
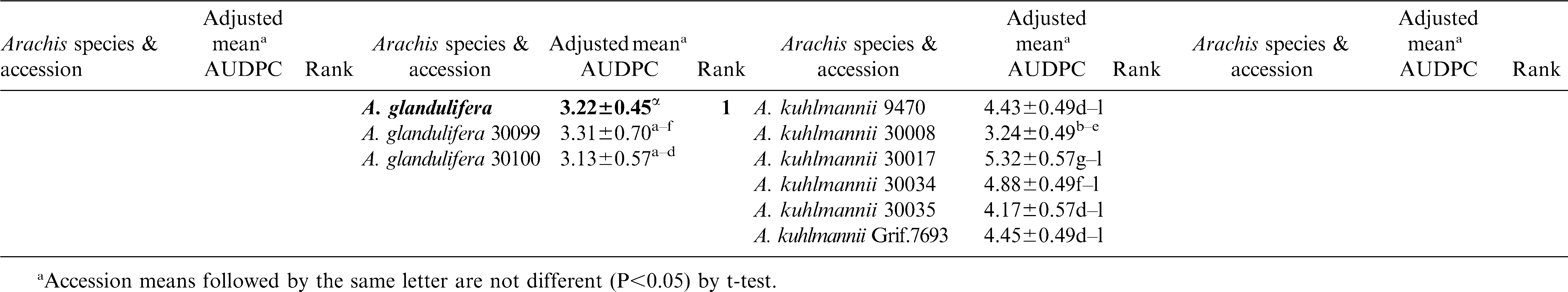
Significant variation among the 23 Arachis species and within accessions of species were observed (P<0.05) for lesion length (Table 2). Replications within blocks did not have significant effects (P<0.05), and as such, blocks were dropped from the analysis. As a group, A. glandulifera exhibited the lowest mean AUDPC of 3.22 ± 0.45 and ranked as the most resistant among the 23 species. The two accessions of this species, KGSSc 30099 and KGSSc 30100, had mean AUDPC values significantly lower than the checks (Table 2). Arachis glandulifera is the sole D genome species in the genus (Stalker, 1991), but it is cross compatible with A. hypogaea (AABB genomes) and thus offers the possibility that resistance may be transferred. Additionally, A. correntina (A genome) with a mean AUDPC value of 3.49 ± 0.22 (mean of six accessions) had the highest levels of resistance as a species and significantly lower mean value than the cultivars. Two different accessions of this species, KSBVn 36000 and KRiP 7830 had the lowest mean AUDPC values among the diploid species accessions (Table 2) and were the most resistant of all the diploid species accessions evaluated in this study. Although as a group A. batizocoi was susceptible, one of its accessions, KGBSPSc 30081, had mean AUDPC of 3.77 ± 0.57 which was significantly lower than the cultivars as a group. Additionally, A. herzogii and A. helodes ranked higher than A. hypogaea based on the mean AUDPC values, although they are not significantly different from the latter (Table 2). Among the A. hypogaea lines, the partially resistant Sclerotinia blight germplasm line, N96076L (Isleib et al., 2006) exhibited the lowest mean AUDPC value (2.10 ± 0.49) as expected. In this study, A. duranensis (A genome) contained the highest number of accessions (31) with a large amount of variation for Sclerotinia blight infection. One of the accessions, KGBSPSc 30067, exhibited the least mean AUDPC value of 2.89 ± 0.70 within this group. However, many of the other accessions of this species were not significantly different from A. hypogaea. For those species with duplicated samples of the same accession, for example A. williamsii accession WiCla 1118 (Tables 2), the mean AUDPC values were not significantly different. Overall, limited useful genetic variability for Sclerotinia blight resistance was observed among the wild species accessions, but some individual accessions of A. correntina (KSBVn 36000 and KRiP 7830) have promising levels of Sclerotinia blight resistance.
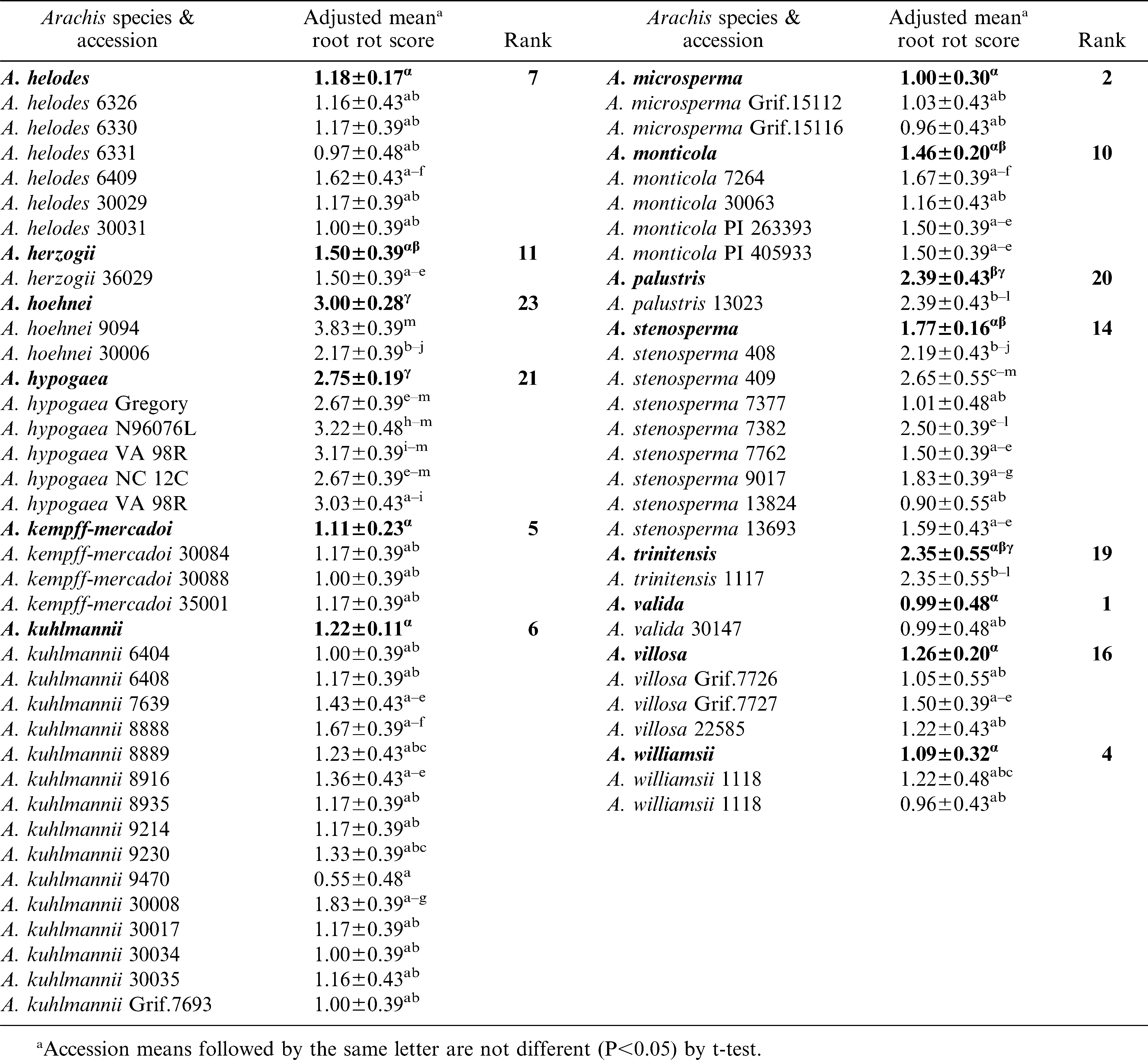
Reaction of Arachis Species to CBR
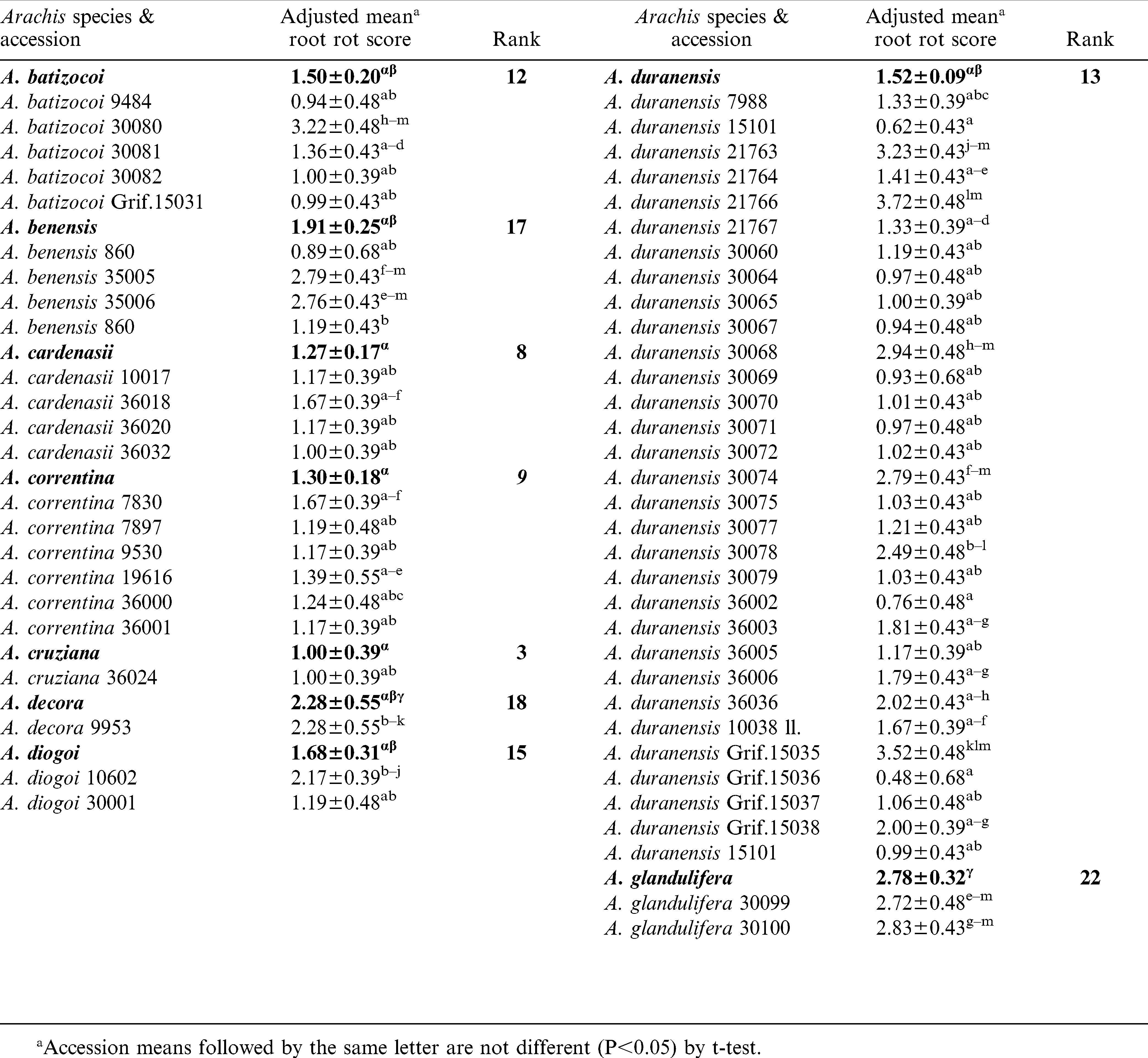
Significant variation (P<0.05) among and within species accessions was observed for CBR root decay (Table 3). The mean root rot scores suggested that A. valida, A. microsperma, and A. cruziana as the most resistant species. The next best group of species included, A. williamsii, A. helodes and A. kempff-mercadoi. Arachis williamsii accession, WiCla 1118, which was duplicated, had similar levels of root rot that did not differ significantly. The root rot scores of A. hypogaea checks were higher than most entries tested in this study. It is also interesting to note that N96076L which was resistant to Sclerotinia blight but susceptible to CBR. Individual accessions of several other species such as, A. batizocoi (K 9484, KGBSPSc 30081 and KGBSPSc 30082), A. kuhlmannii (Grif.7693, VSGr 6404), and A. stenosperma (VSMGeSv 7377), and A. correntina (KSBVn 36000) also exhibited low levels of mean root rot (Table 3). Additionally, several A. duranensis accessions also had low mean root rot including the A genome donor accession, KGBSPSc 30067.
Greenhouse evaluations are less expensive and less laborious compared to field evaluations and may be more reliable for preliminary screening of germplasm for disease resistances. Additionally, good performance in the greenhouse assay can be used to supplement field results in the selection of lines for disease resistance. Hollowell et al. (2008) reported, in a comparison of multiple-year greenhouse assays with field data, a correlation of r = 0.83 (P<0.0001) for CBR, suggesting that the greenhouse assay is a good predictor of field performance. However, the assay for Sclerotinia blight was less reliable as a predictor of field performance as the correlation was only 0.35 (P<0.0001). Similar greenhouse screening methods were used in the early identification of CBR resistant germplasm in peanut (Pataky et al., 1983).
Developing genetic resistance to these diseases will offer growers the economic stability to manage the crop with less input costs. New cultivars with partial resistance to CBR and Sclerotinia blight are being made available to growers (Branch and Brenneman, 2012; Chamberlin and Melouk, 2009). However, new sources of resistances are needed to develop more cultivars with better levels of resistance to these two diseases. As shown in this study, several diploid wild species have exhibited resistance to Sclerotinia blight and CBR in the greenhouse tests and offer new germplasm to breed for resistance to these two diseases. Overall, in this study, the A. batizocoi accession KGBSPSc 30081 and A. correntina accession KSBVn 36000, appeared to contain resistance to both Sclerotinia blight and CBR and are promising sources to breed for resistance to both these diseases. Some of the CBR resistant species such as A. valida, A. williamsii and A. cruziana are B genome species, and it is likely that amphidiploids can be produced with other CBR resistant A genome species such as A. duranensis accession KGBSPSc 30067. Similarly, the CBR resistant B genome species accessions can be hybridized with Sclerotinia blight resistant A genome species accessions to produce amphidiploids with resistance to both these diseases. Also, the A. duranensis accession KGBSPSc 30067, which exhibited resistance to both diseases in this study, has been reported as the A genome donor (Kochert et al., 1996) to A. hypogaea and offers the possibility that some of the PIs within A. hypogaea may contain higher levels of resistance to both CBR and Sclerotinia blight than has been reported. This observation prompted speculation that an evaluation of A. hypogaea PIs collected in close proximity to the location where KGBSPSc 30067 was found may provide resistant germplasm. In the present study, the partially resistant Sclerotinia blight germpalsm line, N96076L, as expected, exhibited resistance where as the diploid wild species A. cardenasii (GKP 10017) which was involved in the parentage of N96076L was susceptible to Sclerotinia blight. N96076L has several ancestors, and not all of them may have contributed the resistance. Further, discrepancies in the reaction of the same Arachis species or the same accession within a species under different environmental conditions were reported by Subrahmanyam et al. (1985). They indicated that the differences in disease reactions may be due to the interaction of the host, pathogen and the environment and also due to the variation within the pathogen. However, the recent multiple disease resistant cultivar releases, Bailey (Isleib et al., 2011), and Tifguard (Holbrook et al., 2008) trace their resistances to diploid Arachis species indicating the potential benefits of wild species research and warrants their use for peanut improvement.
Acknowledgements
This research was supported by funds from NC Peanut Growers Association, NC Crop Improvement Association and NC Foundation Seed Producers, Inc.
Literature Cited
Black M.C and Beute M.K 1984 Relationships among inoculum density, microsclerotia size, and inoculum efficiency of Cylindrocladium crotalariae causing root rot on peanuts Phytopathology 74 : 1128 – 1132 .
Branch W.D and Brenneman T.B 2012 New sources of CBR resistance among runner-type peanut cultivars Peanut Sci 39 : 38 – 42 .
Brandenburg R.L Jordan D.L Shew B.B Wilcut J.W and Toth S.J (eds.) 2005 2005. Crop Profile for Peanuts in North Carolina N.C. Coop. Ext. Serv., N.C. State Univ. , Raleigh 33 pp. Revised .
Chamberlin K.D and Melouk H.A 2009 Identification of possible new sources of Sclerotinia blight resistance in the U.S. peanut mini core collection using a molecular marker associated with resistance to Sclerotinia minor Jagger ASA-CSSA-SSSA Annual Meeting Abstracts, Nov. 1–5, 2009, Pittsburgh, PA, Abstract No. 56804. Available: http://a-c s.confex.com/crops/2009am/webprogram/Paper56804.html .
Dong W.B Brenneman T.B Holbrook C.C and Culbreath A.K 2008 Evaluation of resistance to Cylindrocladium parasiticum of runner-type peanut in the greenhouse and field Peanut Sci 35 : 139 – 148 .
Holbrook C.C 2001 Status of the Arachis germplasm collection in the United States Peanut Sci 28 : 84 – 89 .
Holbrook C.C and Dong W.B 2005 Development and evaluation of a mini core collection for the U.S. peanut germplasm collection Crop Sci 45 : 1540 – 1544 .
Holbrook C.C Timper P Culbreath A.K and Kvien C.K 2008 Registration of ‘Tifguard’ peanut J. Plant Reg 2 : 92 – 94 .
Hollowell J.E Isleib T.G Tallury S.P Copeland S.C and Shew B.B 2008 Screening of virginia-type peanut breeding lines for resistance to Cylindrocladium black rot and Sclerotinia blight in the greenhouse Peanut Sci 35 : 18 – 24 .
Isleib T.G Milla-Lewis S.R Pattee H.E Copeland S.C Zuleta M.C Shew B.B Hollowell J.E Sanders T.H Dean L.O Hendrix K.W Balota M and Chapin J.W 2011 Registration of ‘Bailey’ peanut J. Plant Reg 5 : 27 – 39 .
Isleib T.G Rice P.W Bailey J.E Mozingo R.W and Pattee H.E 1997 Registration of ‘NC 12C’ peanut Crop Sci 37 : 1976 .
Isleib T.G Rice P.W Mozingo R.W Copeland S.C Graeber J.B Shew B.B Smith D.L Melouk H.A and Stalker H.T 2006 Registration of N96076L peanut germplasm Crop Sci 46 : 2329 – 2330 .
Isleib T.G Rice P.W Mozingo R.W Mozingo R.W and Pattee H.E 1999 Registration of ‘Gregory’ peanut Crop Sci 39 : 1526 .
Kochert G Stalker H.T Gimenes M Galgaro L Lopes C.R and Moore K 1996 RFLP and cytogenetic evidence for the progenitor species of allotetraploid cultivated peanut, Arachis hypogaea, (Leguminosae) Am. J. Bot 83 : 1282 – 1291 .
Melouk H.A and Backman P.A 1995 Management of soilborne fungal pathogens , pp. 75 – 82 In: Melouk H.A and Shokes F.M (eds.) Peanut Health Management APS Press , St. Paul, MN .
Mozingo R.W Coffelt T.A and Isleib T.G 2000 Registration of ‘VA 98R’ peanut Crop Sci 40 : 1202 – 1203 .
Pataky J.K Black M.C Beute M.K and Wynne J.C 1983 Comparative analysis of Cylindrocladium black rot resistance in peanut: greenhouse, microplot, and field testing procedures Phytopathology 73 : 1615 – 1620 .
Phipps P.M Beute M.K and Barker K.R 1976 An elutriation method for quantitative isolation of Cylindrocladium crotalariae microsclerotia from peanut field soil Phytopathology 66 : 1255 – 1259 .
Phipps P.M 2006 Applied research on field crop disease control VA Polytechnic Inst. and State Univ. Info. Ser. No. 482, 152 pp.
Rowe R.C and Beute M.K 1975 Variability in virulence of Cylindrocladium crotalariae isolates on peanut Phytopathology 65 : 422 – 425 .
Shaner G and Finney R.E 1977 The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat Phytopathology 67 : 1051 – 1056 .
Stalker H.T 1991 A new species in section Arachis of peanuts with a D genome Am. J. Bot 78 : 630 – 637 .
Stalker H.T and Moss J.P 1987 Speciation, cytogenetics, and utilization of Arachis species Adv. Agron 41 : 1 – 40 .
Stalker H.T and Simpson C.E 1995 Germplasm resources in Arachis , pp. 14 – 53 In: Pattee H.E and Stalker H.T (eds.) Advances in Peanut Science , Amer. Peanut Res. Educ. Soc., Inc. , Stillwater, OK .
Subrahmanyam P Ghanekar A.M Nolt B.L Reddy D.V.R and McDonald D 1985 Resistance to groundnut diseases in wild Arachis species Proc. of an International Workshop on Cytogenetics of Arachis. 31 Oct–2 Nov.,1983. ICRISAT, Patancheru, A. P. 502 324, India .
Subrahmanyam P Moss J.P McDonald D Subba Rao P.V and Rao V.R 1985 Resistance to leaf spot caused by Cercosporidium personatum in wild Arachis species Plant Dis 69 : 951 – 954 .
USDA, ARS, National Genetic Resources Program. Germplasm Resources Information Network - (GRIN) [Online Database] National Germplasm Resources Laboratory , Beltsville, MD URL: http://www.ars-grin.gov/cgi-bin/npgs/html/index.pl (23 January 2007) .
Notes
- Clemson University Pee Dee Research and Education Center, 2200 Pocket Rd., Florence, SC 29506-9727
- Dept. of Crop Sci., Box 7629, N.C. State Univ., Raleigh, NC 27695-7629, respectively * Corresponding author's Email: stallur@clemson.edu
Author Affiliations