Introduction
Peanut (Arachis hypogaea L.) is an important crop in North Carolina (Brown, 2011). Several biotic and abiotic stresses occur during the peanut growing season and timing of their occurrence can overlap. Peanut growers utilize a wide range of agrochemicals to manage these stresses (Gascho and Davis, 1995; Lynch and Mack, 1995; Sherwood et al., 1995; Wilcut et al., 1995). Among different agrochemicals used in peanut, insecticides are often applied in the seed furrow at planting to control tobacco thrips and include organophosphate and carbamate groups (Brandenburg, 2011; Minton and Morgan, 1974; Riley et al., 1997). Tobacco thrips can affect peanut growth and subsequent yield in North Carolina if insecticides are not used (Carley et al., 2009; Drake et al., 2009; Herbert et al., 2007). Acephate, aldicarb, and phorate can be used to control tobacco thrips in peanut when applied in the seed furrow at planting (Brandenburg, 2011). Acephate can also be applied to peanut foliage to control tobacco thrips (Brandenburg, 2011).
Paraquat can be applied within 28 days after peanut emergence to control small annual broadleaf weeds, grasses, and sedges (Wilcut et al., 1995). Bentazon applied with paraquat reduces injury caused by paraquat and in some cases can either increase or decrease weed control (Wilcut et al., 1995). Injury caused by paraquat generally does not reduce peanut yield under weed-free conditions (Blenk et al., 1991; Carley et al., 2009; Drake et al., 2009).
Removal of aldicarb from the commercial market required growers to rely on postemergence applications of insecticides to control tobacco thrips. Although phorate controls tobacco thrips adequately in most instances, control is often less consistent than control by aldicarb (Herbert et al., 2007). Importance of paraquat in a comprehensive weed management program has increased due to prevalence of herbicide-resistant weeds (Jordan et al., 2007). Applying chloroacetamide herbicides with paraquat can increase weed control (Askew et al., 1999; Bailey et al., 1999; Grey et al., 2002; Grichar and Colburn, 1996) although peanut injury often increases (Jordan et al., 2003). However, interactions of acephate, chloroacetamide herbicides, and paraquat are not well defined.
Although interactions of paraquat and acephate have been evaluated in some instances with respect to weed control and peanut injury, compatibility of these pesticides in spray solution prior to application has not been documented. Lack of information on physical compatibility of different agrochemicals in the spray tank can lead to the formation of precipitates or suspended solids which may adversely affect application and consequently affect pest control (Houghton, 1982).
Defining interactions of paraquat and acephate with respect to visible injury caused by paraquat and damage from tobacco thrips feeding will be of value for growers formulating pest management strategies in peanut. These findings will be especially important if greater use of paraquat occurs due to herbicide-resistant weeds and if acephate is applied more frequently in absence of aldicarb availability. Therefore, objectives of this research were to define interactions of acephate and paraquat applied alone or in combination with chloroacetamide herbicides with respect to tobacco thrips damage and peanut injury and to determine changes in spray solution characteristics when these pesticides are co-applied.
Material and Methods
Interactions of Acephate, Paraquat, and Chloroacetamide Herbicides
Experiments were conducted in North Carolina during 2009 and 2010 at the Peanut Belt Research Station near Lewiston-Woodville on a Norfolk sandy loam soil (fine-loamy, kaolinitic, thermic Typic Kandiudults). The peanut cultivar Phillips (Isleib et al., 2006) was planted in rows spaced 91 cm apart in conventionally prepared seed beds with a final in-row population of 13 plants/m. Plot size was one row by 3.5 m long with a non-treated row separating each plot. Peanut was not irrigated.
Treatments included two levels of acephate (Orthene97®, Amvac Chemical Corp., Los Angeles, CA 90023) (0 and 1.1 kg ai/ha); three levels of contact herbicides including no contact herbicide, paraquat (Gramoxone Inteon Herbicide, Syngenta Crop Protection, Inc. Greensboro, NC 27419) at 0.14 kg ai/ha, and paraquat plus bentazon (Basagran Herbicide, BASF Corp., Research Triangle Park, NC 27709) at 0.14 kg/ha + 0.28 kg ai/ha; and four levels of chloroacetamide herbicides including no chloroacetamide herbicide, alachlor (Intrro Preemergence Herbicide, Monsanto Co., St. Louis, MO 63167) at 3.4 kg ai/ha, dimethenamid-P (Outlook Herbicide, BASF Corp., Research Triangle Park, NC 27709) at 0.84 kg ai/ha, and S-metolachlor (Dual Magnum Herbicide, Syngenta Crop Protection, Inc. Greensboro, NC 27419) at 1.1 kg ai/ha. Treatments were applied 7 to 10 days after peanut emergence. Nonionic surfactant (Induce®, Helena Chemical Corp., Collierville, TN 38017) at 0.125% (v/v) was applied with all treatments containing paraquat. No insecticide was applied in the seed furrow at planting to promote tobacco thrips feeding in peanut. Treatments were applied in 140 L/ha aqueous solution using a CO2-pressurized backpack sprayer with flat-fan nozzles (TeeJet TP8002 flat-fan spray nozzles, Spraying Systems Co., Wheaton, IL 60189) at 275 kPa.
The experimental design was a randomized complete block with four replications. An ordinal scale of 0 to 5 where 0 = no damage, 1 = noticeable feeding but no stunting, 2 = noticeable feeding and 25% stunting, 3 = noticeable feeding with blackened terminals and 50% stunting, 4 = severe feeding and 75% stunting, and 5 = severe feeding and 90% stunting was used to visibly assess damage from tobacco thrips feeding 10 days after postemergence treatment (DAT). Visible estimates of percent injury relative to symptoms associated with paraquat (Senseman, 2007) were recorded 10 DAT using a scale of 0 to 100% where 0 = no foliar necrosis and 100 = plant death. Peanut was not harvested.
Tobacco Thrips Control with Aldicarb and Foliar Pesticide Treatments
Experiments were conducted in North Carolina during 2011 at the Peanut Belt Research Station near Lewiston-Woodville and the Upper Coastal Plain Research Station located near Rocky Mount in two separate fields at each location. Soils were a Norfolk sandy loam described previously at Lewiston-Woodville and an Adcock fine sandy loam soil (fine-loamy, siliceous, subactive, thermic Aquic Paleudults) at Rocky Mount. The peanut cultivar Phillips was planted as described previously in plots with two rows spaced 91 cm apart by 9 m in length. Peanut was not irrigated.
Treatments included no aldicarb or aldicarb (Temik 15G insecticide, Bayer CropScience, Research Triangle Park, NC 27709) at 1.1 kg ai/ha applied alone in the seed furrow at planting following seed drop but before slit closure. These treatments also received acephate postemergence, paraquat postemergence, acephate plus paraquat postemergence, or no additional pesticides to establish a 2 by 4 factorial treatment structure. Acephate and paraquat were applied at the rates described previously. Nonionic surfactant at 0.125% (v/v) was applied with all treatments containing paraquat.
The experimental design was a randomized complete block with four replications. Visible estimates of tobacco thrips damage from feeding were determined 10 and 14 DAT using the ordinal scale described previously. Visible estimates of percent injury relative to symptoms associated with paraquat were recorded 10 and 14 DAT using the injury scale described previously. The number of days from peanut emergence to canopy closure was determined, and peanut was dug and vines inverted when approximately 65% of pods were in the brown and black category based on pod mesocarp color for the treatment of aldicarb with no other postemergence treatment (Williams and Drexler, 1981).
Physical Compatibility of Insecticides with Other Agrochemicals
Laboratory experiments were conducted to compare physical compatibility of acephate combinations used in the field experiments during 2009 and 2010. Unlike experiments in the field where a municipal water source was used, deionized water at pH 6.27 was used in the laboratory experiments. Pesticides were mixed in the following order: dry flowables (acephate), emulsifiable concentrates (alachlor and S-metolachlor), and soluble liquids (bentazon, dimethenamid-P, nonionic surfactant, and paraquat). Solutions were prepared in a final volume of 80 ml in sterilized plastic specimen cups (Specimen cup120 ml-53 ST ORG CAP, Fisher Scientific, Fairlawn NJ 07410) of 120 ml capacity at ratios reflecting spray volume and pesticide ratios used in the field. After mixing, solution was vortexed (Vortex Genie 2™, Fisher Scientific, Fairlawn, NJ 07410) immediately and evaluated visually for precipitates followed by determining pH using a portable pH meter (Oakton portable pH meter, Fisher Scientific, Fairlawn, NJ 07410). Solutions were allowed to sit for 6 h after mixing, evaluated for precipitates, vortexed, and re-evaluated for precipitates followed by pH determination. Similarly, mixtures were allowed to sit for 24 and 72 h after the initial solution preparation and subjected to the same procedure and observations. Presence or absence of precipitates were determined visually and described as Yes or No, respectively. Visible deposition on the bottom of the specimen cup or presence of suspended solids in the solution was considered precipitates. Temporary precipitates went back into solution on vortexing while permanent precipitates did not go into solution after vortexing. Experiments were conducted as a completely randomized design with two replications and the experiment was repeated.
Statistical Analysis
Data for peanut damage, visible peanut injury, and solution pH from field and physical compatibility studies were subjected to ANOVA using the PROC MIXED procedure of SAS (Statistical Analysis Systems®, version 9.2, SAS Institute Inc., Cary, NC 27513) appropriate for the factorial arrangement of treatments using expected mean squares to test fixed and random effects. Pesticide treatments were considered as fixed effects while experiments and replications were considered as random effects. Data for peanut damage from thrips feeding and peanut injury from paraquat were log and arcsine transformed, respectively, using Box-Cox procedure. However, transformation of data did not change data interpretation, hence, data were presented without transformation (Box et al., 1978). Means for significant main effects and interactions were separated using Tukey's pair-wise comparison test at P ≤ 0.05.
Results and Discussion
Interactions of Acephate, Paraquat, and Chloroacetamide Herbicides
Damage from tobacco thrips feeding 10 DAT was affected by interaction of acephate X chloroacetamide herbicide (p = 0.0028). When pooled over contact herbicides and years, damage from tobacco thrips feeding was no more than 1.3 on a scale of 0 to 5 when acephate was applied irrespective of chloroacetamide herbicide (Table 1). In absence of acephate, more damage from tobacco thrips feeding was noted when alachlor was applied compared with no chloroacetamide herbicide. Damage was similar for all chloroacetamide herbicides in absence of acephate. When acephate was included there was no difference in damage from tobacco thrips when comparing among chloroacetamide herbicides.
Interactions of acephate by contact herbicide by chloroacetamide herbicide (p = 0.0075) and acephate by contact herbicide (p = 0.0011) were significant for peanut injury associated with paraquat symptomology during 2009 and 2010, respectively. In the absence of acephate, co-application of paraquat with alachlor, dimethenamid-P, and S-metolachlor increased injury compared to peanut treated with paraquat alone during 2009 (Table 2). Peanut injury associated with paraquat did not differ when bentazon was included in absence of acephate or when acephate was applied irrespective of chloroacetamide herbicide treatment (Table 2). Jordan et al. (2003) reported that the chloroacetamide herbicides S-metolachlor applied with paraquat increased peanut injury compared with paraquat alone. The mechanism of increased injury when paraquat was applied with chloroacetamide herbicides compared with paraquat alone is not known. However, it is speculated that emulsifiers in the formulated product containing S-metolachlor may increase injury from paraquat through increased absorption of paraquat in peanut. Although chloroacetamide herbicides did not affect injury in 2010, less injury from paraquat was noted when acephate was applied with paraquat compared with paraquat alone (Table 3). However, when bentazon was included with paraquat, acephate did not reduce injury. When acephate was not included, our results were similar to previous research (Wehtje et al., 1986, 1992; Wilcut et al., 1989) indicating that bentazon reduces peanut injury by paraquat compared with paraquat alone. In contrast, benefits of bentazon in reducing injury by paraquat were reduced or eliminated when acephate was included in the mixture. These results are not substantiated or refuted in the peer-reviewed literature.
Tobacco Thrips Control with Aldicarb and Foliar Pesticide Treatments
Results from the experiment with acephate, chloroacetamide herbicides, and paraquat were unclear with respect to whether or not reduction in paraquat injury by acephate was associated with interactions in the spray solution, physiological response on the leaf surface, or the plant's ability to compensate for injury through more expansive growth in absence of tobacco thrips feeding. Therefore, the experiment comparing paraquat injury and tobacco thrips damage was conducted to address this question. In theory, applying combinations of acephate, bentazon, and paraquat in presence and absence of tobacco thrips feeding could allow a greater distinction among peanut response to these interactions.
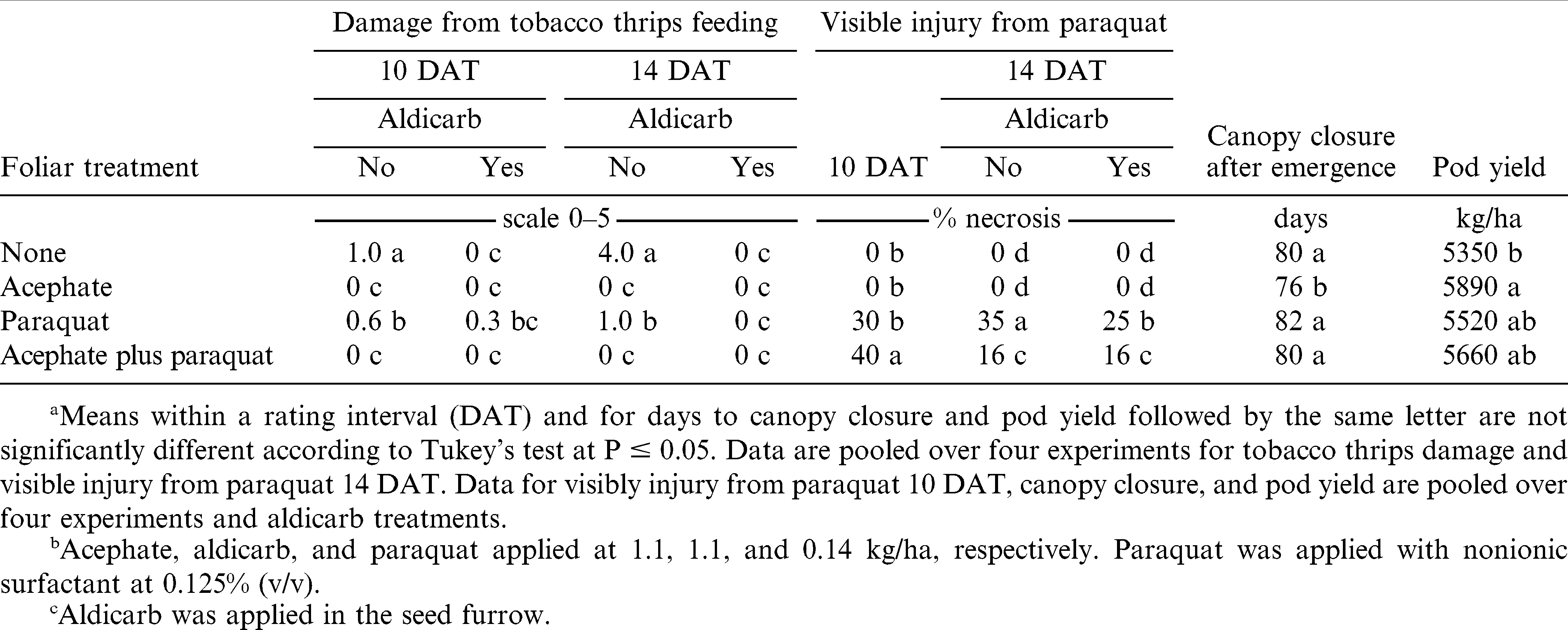
Peanut damage caused by tobacco thrips at 10 and 14 DAT was affected by the interaction of aldicarb (no aldicarb or aldicarb) X foliar pesticide (no pesticide, acephate, paraquat, or acephate plus paraquat) (p = 0.0379 at 10 DAT; p ≤ 0.0001 at 14 DAT). Damage from tobacco thrips feeding did not exceed a value of 1.0 at 10 DAT regardless of foliar pesticide treatment but ranged from 0 to 4.0 by 14 DAT (Table 4). Surprisingly, less visible damage from tobacco thrips feeding was noted when paraquat was applied alone compared with the no-insecticide control 10 and 14 DAT. Acephate and aldicarb were both equally effective in preventing visible damage from tobacco thrips 10 and 14 DAT (Table 4). Previous research (Drake et al., 2009; Funderburk et al., 1998) indicates that peanut damage by tobacco thrips decreases when aldicarb is applied in the seed furrow at planting or when acephate is applied to peanut foliage (Brandenburg, 2011; Herbert et al., 2007).
Main effect of aldicarb and foliar pesticide treatment were significant (p ≤ 0.0001) 10 DAT for peanut injury reflecting paraquat symptoms. The interaction of aldicarb X foliar pesticide treatment was also significant (p ≤ 0.0001) 14 DAT. When pooled over foliar pesticide treatments at 10 DAT, paraquat injury was 16% (data not presented in tables). When pooled over aldicarb treatments and experiments, injury by acephate plus paraquat exceeded that of paraquat alone at 10 DAT (Table 4). However, by 14 DAT, less injury from paraquat was noted when paraquat was applied either with acephate or following aldicarb applied to the seed furrow compared with paraquat alone (Table 4). The reason for increased injury when paraquat and acephate were co-applied is not clear. Adverse interactions of carbamate insecticides and herbicides have been reported previously (Hatzios and Penner, 1985) but not with this particular combination. Fewer symptoms associated with paraquat by 14 DAT may have been associated with more rapid growth of foliage in absence of tobacco thrips feeding and less total foliar necrosis on a percentage basis than would have been observed with paraquat symptoms on plants with less growth during the 5-day period after the first visible rating was recorded.
Main effects aldicarb and foliar pesticide treatment were significant for the number of days from peanut emergence to row closure (p ≤ 0.0001 for both main effects) and pod yield (p = 0.0033 and 0.0169 for these respective main effects). When pooled over foliar pesticide treatments, fewer days from emergence to row closure were required when aldicarb was applied (77 vs. 82 days, data not shown in tables), and pod yield was higher following aldicarb (5760 kg/ha vs. 5430 kg/ha, data not shown in tables). These differences most likely reflect a reduction in tobacco thrips feeding resulting in more rapid and sustained peanut growth when aldicarb was applied. Peanut yield is often higher when aldicarb is applied and tobacco thrips damage is minimized (Carley et al., 2009; Drake et al., 2009; Funderburk et al., 1998; Herbert et al., 2007). When pooled over aldicarb treatments, peanut treated with acephate took fewer days to close the canopy when paraquat was not included (Table 4). However, no difference in days to row closure were noted when paraquat was applied alone or with acephate compared with the no-foliar pesticide control. Pod yield following acephate exceeded that of the no-pesticide control while yield following paraquat alone or with acephate was intermediate between these treatments (Table 4). Consistent with research by Herbert et al. (2007), these data indicate that both aldicarb and acephate contribute to yield increases over peanut not treated with insecticide in presence of tobacco thrips and the damage they cause. Drake et al. (2009) reported that paraquat injury generally does not affect pod yield unless paraquat is applied when significant damage from tobacco thrips feeding is present. Also, manufacturers of paraquat indicate that paraquat should not be applied if tobacco thrips damage is severe (Anonymous, 2011). In contrast, peanut yield reductions caused by damage from tobacco thrips in absence of insecticide are not affected by paraquat treatment (Carley et al., 2009; Drake et al., 2009).
Physical Compatibility of Insecticides with Other Agrochemicals
The interaction of acephate by contact herbicide by chloroacetamide herbicide was significant for solution pH (p = ≤ 0.0001). When compared to carrier pH (6.27), all combinations reduced solution pH except the combination of alachlor with paraquat plus bentazon. When compared with mixtures without acephate, solution pH decreased when acephate was included irrespective of chloroacetamide or contact herbicides. Regardless of chloroacetamide herbicide in solution, paraquat plus bentazon decreased solution pH to approximately 5.0. When compared with solutions containing paraquat with or without acephate, including bentazon increased solution pH.
In most of instances, solutions either with or without acephate combinations formed temporary precipitates at 0, 6, 24, and 72 h sampling times (data not shown in tables). Combinations of acephate with dimethenamid-P formed temporary precipitates, however, in the absence of acephate this combination did not produce precipitates across sampling intervals (data not shown in tables). Solutions containing alachlor with acephate formed permanent precipitates across sampling times but the addition of paraquat or paraquat plus bentazon to these combinations changed the permanent precipitates to temporary precipitates (data not shown in tables).
Practical application of these physical observations is currently unknown. However, very little information on changes in pH and observations on precipitate formation are found in the peer-reviewed literature. These results may at some point contribute to a greater understanding of issues related to agrochemical compatibility.
Collectively, results in the field during 2009 and 2010 demonstrated that chloroacetamide herbicides were more injurious when acephate was not applied and damage from tobacco thrips was present compared with co-application with acephate. Acephate and aldicarb contributed to tobacco thrips control independently and protected pod yield, although the benefit from acephate was marginalized when paraquat was included. Co-application of paraquat with alachlor and S-metolachlor increased injury in the absence of acephate compared with co-application with acephate. Paraquat injury was lower when paraquat was applied with bentazon in absence of acephate compared with paraquat alone. When acephate was applied, there was no difference in injury with paraquat alone or paraquat plus bentazon. The combination of paraquat without acephate was more injurious than this paraquat applied with acephate. However, the value of acephate in reducing paraquat injury most likely was associated with ability of peanut to grow more rapidly in presence of acephate, and less damage from tobacco thrips most likely was associated with the use of a visible scale for evaluation. Bentazon reduces injury from paraquat by affecting physiological process associated with electron transport in treated leaves (Wehtje et al., 1992). Bentazon was not included in the second experiment where pesticides were applied to foliage in presence or absence of damage from tobacco thrips feeding created by aldicarb treatment. While these results provide some insight into possible interactions of acephate and paraquat and subsequent injury and damage when co-applied, the replacement of bentazon with acephate as a “safener” is not established in this research. These data suggest that reductions in tobacco thrips feeding by acephate contribute to a perception of less injury from paraquat, and recommendations of using bentazon to minimize peanut injury from paraquat should not be changed. Paraquat and chloroacetamide herbicides did not adversely affect tobacco thrips control by acephate suggesting that these pesticides can be co-applied to maximize weed control without adversely affecting tobacco thrips control.
Acknowledgements
This research was supported financially by the North Carolina Peanut Growers Association. The authors would like to appreciate Dewayne Johnson, Gary Little, and staff at the Peanut Belt Research Station and Upper Coastal Plain Research Station for technical assistance.
Literature Cited
Anonymous 2011 Gramoxone Inteon herbicide label Syngenta Crop Protection , Greensboro, NC .
Askew S.D Wilcut J.W and Cranmer J.R 1999 Weed management in peanut (Arachis hypogaea) with flumioxazin preemergence Weed Technol. 13 : 594 – 598 .
Bailey W.A Wilcut J.W Jordan D.L Swann C.W and Langston V.B 1999 Response of peanut (Arachis hypogaea) and selected weeds to diclosulam Weed Technol. 13 : 771 – 776 .
Blenk E.S Linker H.M and Coble H.D 1991 Interaction of tobacco thrips (Frankliniella fusca), paraquat, and mechanical defoliation on peanut (Arachis hypogaea) growth, quality, and yield Proc. Am. Peanut Res. And Ed. Soc. 22 : 76 .
Box G.E.P Hunter W.G and Hunter J.S 1978 Statistics for experimenters: An introduction to design, data analysis, and model building New York : J. Wiley .
Brandenburg R.L 2011 Peanut insect management . pp. 88 – 100 In: 2011 Peanut Information North Carolina Coop. Ext. Ser. Publication AG-331 .
Brown A.B 2011 . pp. 7 – 9 In: 2011 Peanut Information North Carolina State University Coop. Ext. Ser. Publication AG-331 .
Carley D.S Jordan D.L Brandenburg R.L and Dharmasri L.C 2009 Factors influencing response of Virginia market type peanut (Arachis hypogaea) to paraquat under weed-free conditions Peanut Sci. 36:180 : 189 .
Drake W.L Jordan D.L Brandenburg R.L Lassiter B.R Johnson P.D and Royals B.M 2009 Peanut cultivar response to damage from tobacco thrips and paraquat Agron. J. 101 : 1388 – 1393 .
Funderburk J.E Gorbet D.W Tearre I.D and Stavisky J 1998 Thrips injury can reduce peanut yield and quality under conditions of multiple stress Agron. J. 90 : 563 – 566 .
Gascho G.J and Davis J.G 1995 Soil fertility and plant nutrition . pp. 383 – 418 In: Pattee H.E and Stalker H.T (eds.) Advances in Peanut Science Am. Peanut Res. And Education Soc. , Stillwater, OK .
Grey T.L Bridges D.C Eastin E.F and MacDonald G.E 2002 Influence of flumioxazin rate and herbicide combinations on weed control in peanut (Arachis hypogaea L.) Peanut Sci. 29 : 24 – 29 .
Grichar J.E and Colburn A.E 1996 Flumioxazin for weed control in Texas peanut (Arachis hypogaea L.) Peanut Sci. 23 : 30 – 36 .
Hatzios K.K and Penner D 1985 Interaction of herbicides with other agricultural chemicals in higher plants Rev. Weed Sci. 1 : 1 – 64 .
Herbert D.A Malone S Aref S Brandenburg R.L Jordan D.L Royals B.M and Johnson P.D 2007 Role of insecticides in reducing thrips injury to plants and incidence of tomato spotted wilt virus in Virginia market-type peanut J. of Economic Entomol. 100 ( 4 ): 1241 – 1247 .
Houghton R.D 1982 Pesticide compatibility: overview from technical services Pesticide Tank Mix Applications: First conference, ASTM STp 764. Wright J.F Lindsay A.D and Sawyer E (eds.) American Society for Testing and Materials. pp. 3 – 10 .
Isleib T.G Rice P.W Mozingo R.W Copeland S.C Graeber J.B Pattee H.E Sanders T.H and Coker D.L 2006 Registration of ‘Phillips’ peanut Crop Sci. 46 : 2308 – 2309 doi:10.2135/cropsci2005.12.0491 .
Jordan D.L Spears J.F and Wilcut J.W 2003 Tolerance of peanut (Arachis hypogaea L.) to herbicides applied postemergence Peanut Sci. 30 : 8 – 13 .
Jordan D Prostko E Dotray P Wilcut J Baughman T Brecke B Chapin J Faircloth J Faircloth W Ferrell J Grey T Grichar J MacDonald G and Medlin C 2007 Managing herbicide-resistant weeds in peanuts in the United States North Carolina Cooperative Extension Service Publication AG-692 , 7 pp.
Lynch R.E and Mack T.P 1995 Biological and biotechnical advances for insect management in peanut In: Advances in Peanut Science , Pattee H.E and Stalker H.T (eds.) , Stillwater: Am. Peanut Res. and Educ. Soc. 95 – 159 .
Minton N.A and Morgan L.W 1974 Evaluation of systemic and nonsystemic pesticides for insect and nematode control in peanuts Peanut Sci. 1 : 91 – 98 .
Riley D.G Douce G.K and McPherson R.M 1997 Summary of losses from insect damage and costs of control in Georgia 1996 Georgia Agri. Exp. Sta., Univ. Georgia Spec. Publ. No. 91 , Athens .
Senseman S.A 2007 Herbicide handbook Weed Science Society of America , Lawrence, KS 458 pp.
Sherwood J.L Beute M.K Dickson D.W Elliot V.J Nelson R.S Opperman C.H and Shew B.B 1995 Biological and biotechnical control in Arachis diseases In: Pattee H.E and Stalker H.T Advances in Peanut Science Raleigh: Am. Peanut Res. and Educ. Soc. pp. 160 – 206 .
Wehtje G McGuire J.A Walker R.B and Patterson M.G 1986 Texas panicum (Panicum texanum) control in peanuts (Arachis hypogaea) with paraquat Weed Sci. 34 : 308 – 311 .
Wehtje G Wilcut J.W and McGuire J.A 1992 Influence of bentazon on the phytotoxicity of paraquat to peanut (Arachis hypogaea) and associated weeds Weed Sci. 40 : 90 – 95 .
Wilcut J.W York A.C Grichar W.J and Wehtje G.R 1995 The biology and management of weeds in peanut (Arachis hypogaea L.) In: Pattee H.E and Stalker H.T (eds.) Advances in Peanut Science Stillwater: Am. Peanut Res. and Educ. Soc. pp. 207 – 224 .
Wilcut J.W Wehtje G Hicks T.V and McGuire J.A 1989 Postemergence weed control systems without dinoseb for peanuts (Arachis hypogaea) Weed Sci. 37 : 385 – 391 .
Williams E.J and Drexler J.S 1981 A non-destructive method for determining peanut pod maturity, pericarp, mesocarp, color, morphology, and classification Peanut Sci. 8 : 134 – 141 .
Notes
- Former Graduate Research Assistant, Professor, and Former Graduate Research Assistant, Department of Crop Science, Box 7620, North Carolina State University, Raleigh, NC 27695; and William Neal Reynolds Professor, Department of Entomology, Box 7613, North Carolina State University, Raleigh, NC 27695 * Corresponding author's E-mail: david_jordan@ncsu.edu
Author Affiliations