Introduction
Tomato spotted wilt (TSW) caused by tomato spotted wilt virus (TSWV) (Tospovirus Bunyaviridae) is an economically important disease in peanut (Arachis hypogaea L.) produced in the southeastern U.S. The virus is vectored during thrips (Thysanoptera sp.) feeding on peanut foliage. Susceptible peanut varieties show a range of symptoms including mottling of leaves, moderate to severe stunting, mouse-ear shaped leaves, overall yellowing of foliage, and moderate to severe yield loss (Culbreath, et al., 2000). Many weedy plants and other crops serve as hosts to TSWV making disease predictability and control difficult. Peanut planting is historically initiated when soil temperatures average >18.3 C at a 10 cm depth for 3 consecutive days with a favorable forecast as early as April 10 (Beasley, 2007). Many factors influence the incidence of TSW in peanut including plant population, row patterns, insecticide use, cultivar, thrips populations, and planting date. Research has shown that planting after May 10 and before June 1 helps to reduce incidence of TSW by avoiding high thrips populations (Brown, et al., 2005) and is a major factor contributing to the TSWV risk index (Kemerait et al., 2011). Traditionally, growers would start planting early to spread out the planting and harvest seasons. Planting early has several benefits including avoiding potential tropical storms and the risk of incurring cool temperatures in early fall that could reduce peanut maturity at harvest severely affecting yield, grade, and crop value. The tight window of planting required to avoid the majority of thrips feeding forces a greater workload on growers during planting and harvest. Later planting also puts peanut buying points under more duress due to the compressed harvest season. Previous research in the southeastern U.S. indicates variability in TSW incidence among locations and years (Tillman et al., 2007; Culbreath et al., 2010). Because of the unpredictability of TSWV pressure, Tillman et al. (2007) concludes to plant the least susceptible cultivars during the recommended plantings dates, while Culbreath et al. (2010) finds that cultivars showing higher TSW tolerance may be planted earlier than the recommended window. Breeding programs are focused on improving phenotypic tolerance to TSW for agronomic purposes. New cultivars continue to show improved tolerance to TSW (Holbrook et al., 2008) and may allow growers to resume the historical mid-April initiation of planting date with less regard to the current planting window to avoid thrips feeding. The objectives of this study were to evaluate peanut cultivars Georgia Green (Branch, 1996), Georgia-03L (Branch, 2004), AT 3085RO, Flavor Runner 458 and the advanced breeding line EXP 271516 to their phenotypic tolerance to TSW in the field over a 40-day planting window. Evaluations were based on peanut foliar condition, pod yield, and grade in 2008 and 2009 in Dawson, Georgia and Headland, Alabama.
Materials & Methods
Replicated field experiments were conducted near Dawson, Georgia in 2008 [Tifton (Fine-loamy, kaolinitic, thermic Plinthic Kandiudults; 2–5% slope] and 2009 [Tifton (Fine-loamy, kaolinitic, thermic Plinthic Kandiudults; 0–2% slope)] and duplicated near Headland, Alabama [Dothan (Fine-loamy, kaolinitic, themic plinthic kandiudults; 0–2% slope)] in each year.
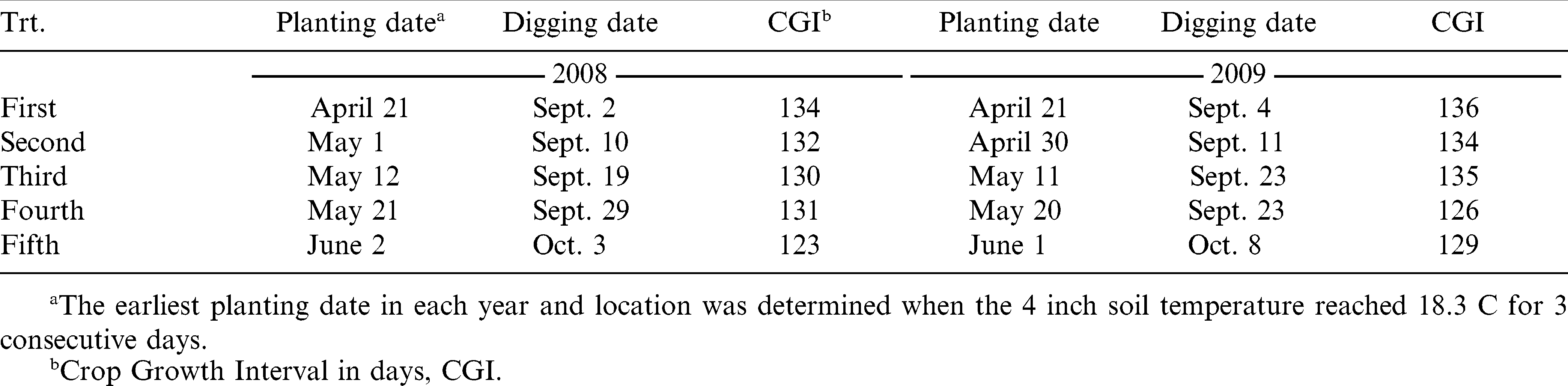
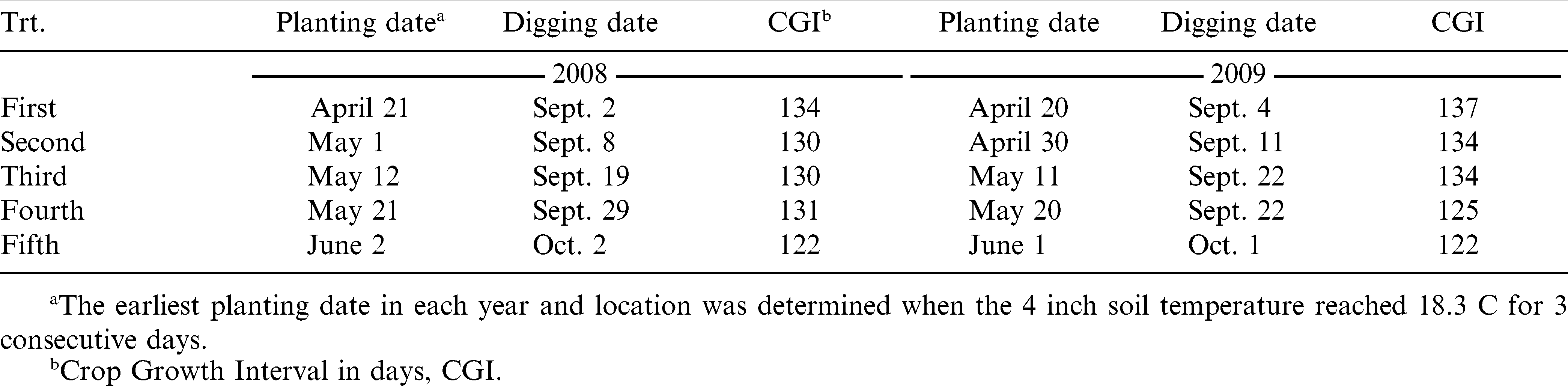
Four peanut cultivars and one advanced breeding line were selected for comparisons. Cultivar Georgia Green had been the accepted agronomic standard and was planted on a majority of acres in the Southeast prior to 2008, because of its tolerance to TSW. The more recently released cultivars Georgia-03L, AT 3085RO, and the experimental line EXP 271516 are considered to have greater field tolerance to TSW compared to Georgia Green. Cultivar Flavor Runner 458 was included in these experiments as a known TSW susceptible entry (Brown et al., 2005). Peanuts were planted at five planting dates each year starting in April. The earliest planting date in each year and location was determined when the 10-cm soil temperature reached 18.3 C for 3 consecutive days after April 10. Once the initial planting date was determined, planting was repeated in 10-day intervals four times. Planting date is considered a fixed term in this study to establish the first two planting dates before the recommended planting window and the latest one after the window (Tables 1 and 2).
Peanuts were planted in single rows on 0.9 m centers with a cone planter applying 0.84 kg ai/ha aldicarb (Bayer CropScience, Research Triangle Park, North Carolina) in furrow. Each planting date at each location was arranged in a randomized complete block design in a separate block (Gomez and Gomez, 1984). Germplasm entries were replicated four times in each planting date block. All germplasm entries are considered to have similar maturity requirements, thus digging date was determined separately by location and planting date according to the hull scrape method (Williams and Drexler, 1981) of Georgia Green in each respective planting date block. Border rows were planted to Georgia Green to use for maturity determination. Plots size was 6.1 m by 2 rows in 2008 and 6.1 m by 4 rows in 2009 to ensure that yield samples were large enough for further testing. Agronomic management inputs and irrigation were applied according to University of Georgia (Beasley et al., 2007) and Auburn University best management practices for peanut, respectively by location.
All plots in each planting date were visually rated for TSW symptoms to foliage according to the 1 to 10 method described by Tillman et al. (2007) once between July 22–25 and again immediately prior to digging. These ratings were converted to a 1 to 100 scale to be expressed as a percentage for presentation. After peanut digging and inverting, the roots and pods of 12.2 m linear row were visually rated for TSW symptoms. Disease hits were recorded per foot of row when yield loss from TSW was evident. Peanuts were threshed with a stationary harvester (Kingaroy Engineering Works, Kingaroy, Australia) after the vines were sufficiently field dried. Yield samples were dried to a kernel moisture content of <10%, and yield calculations are shown corrected to 7% moisture. A uniform portion of the sample was cut out with a riffle divider to be graded.
Data were analyzed in SAS (version 9.1) with PROC GLM under the general linear model. The variety*location*year, planting date*year, planting date*location, and variety*location interactions were significant so each planting date is reported separately by year and location. Treatment effect F tests were carried out against their specific error source. Means were separated using Fisher's Protected LSD at p≤0.05.
Results and Discussion
Foliar TSW Ratings at Mid-Season
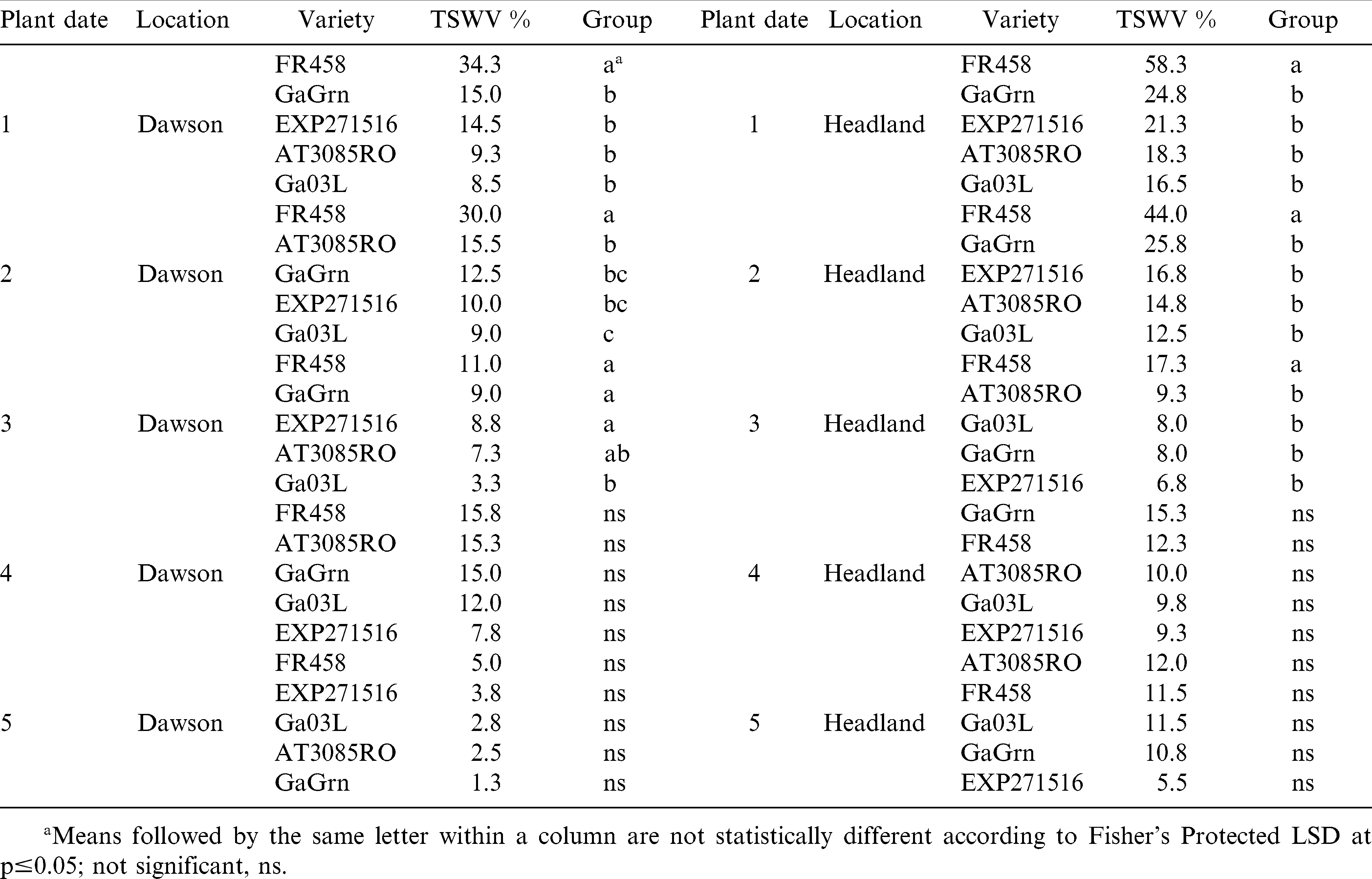
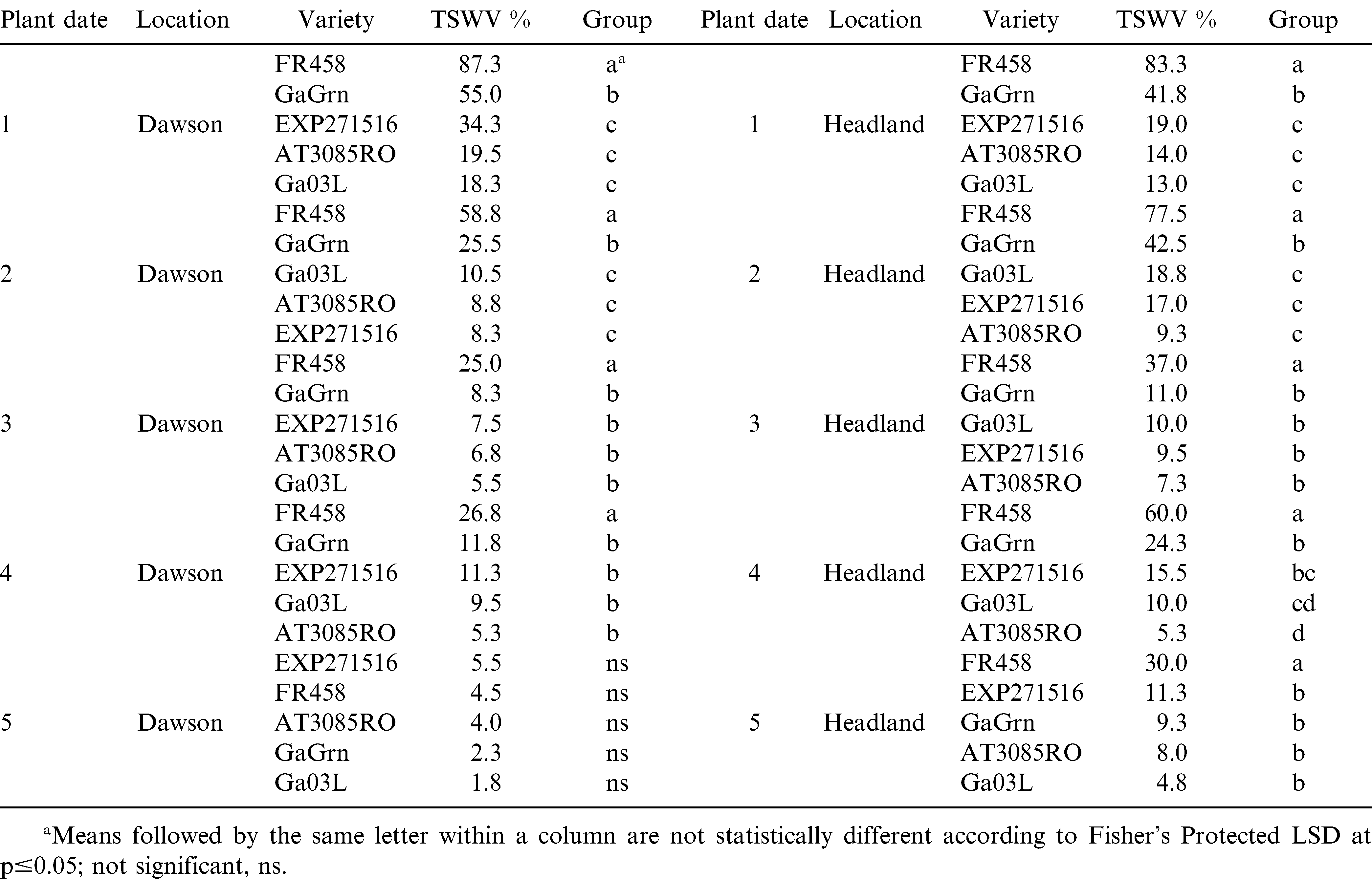
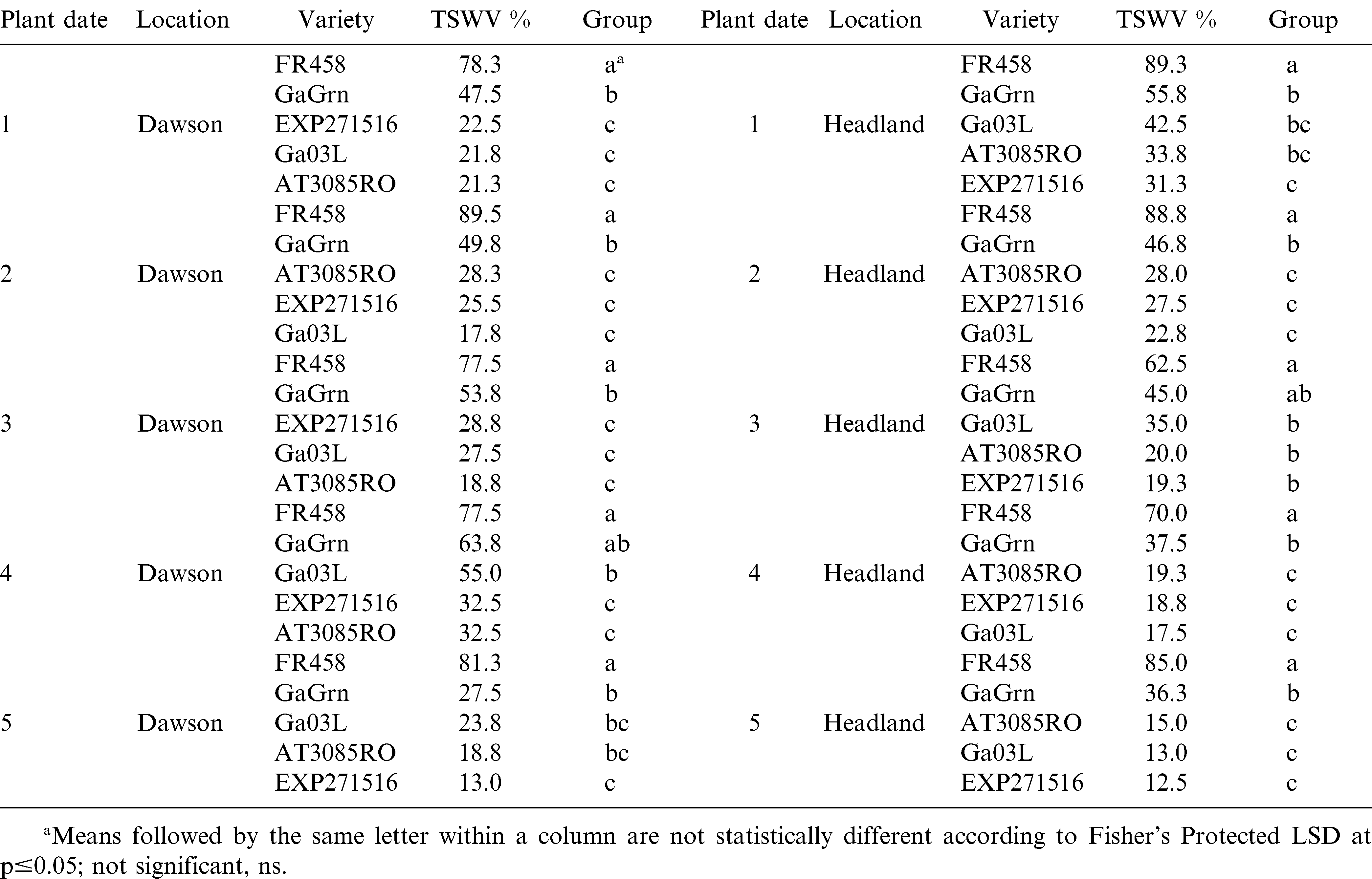
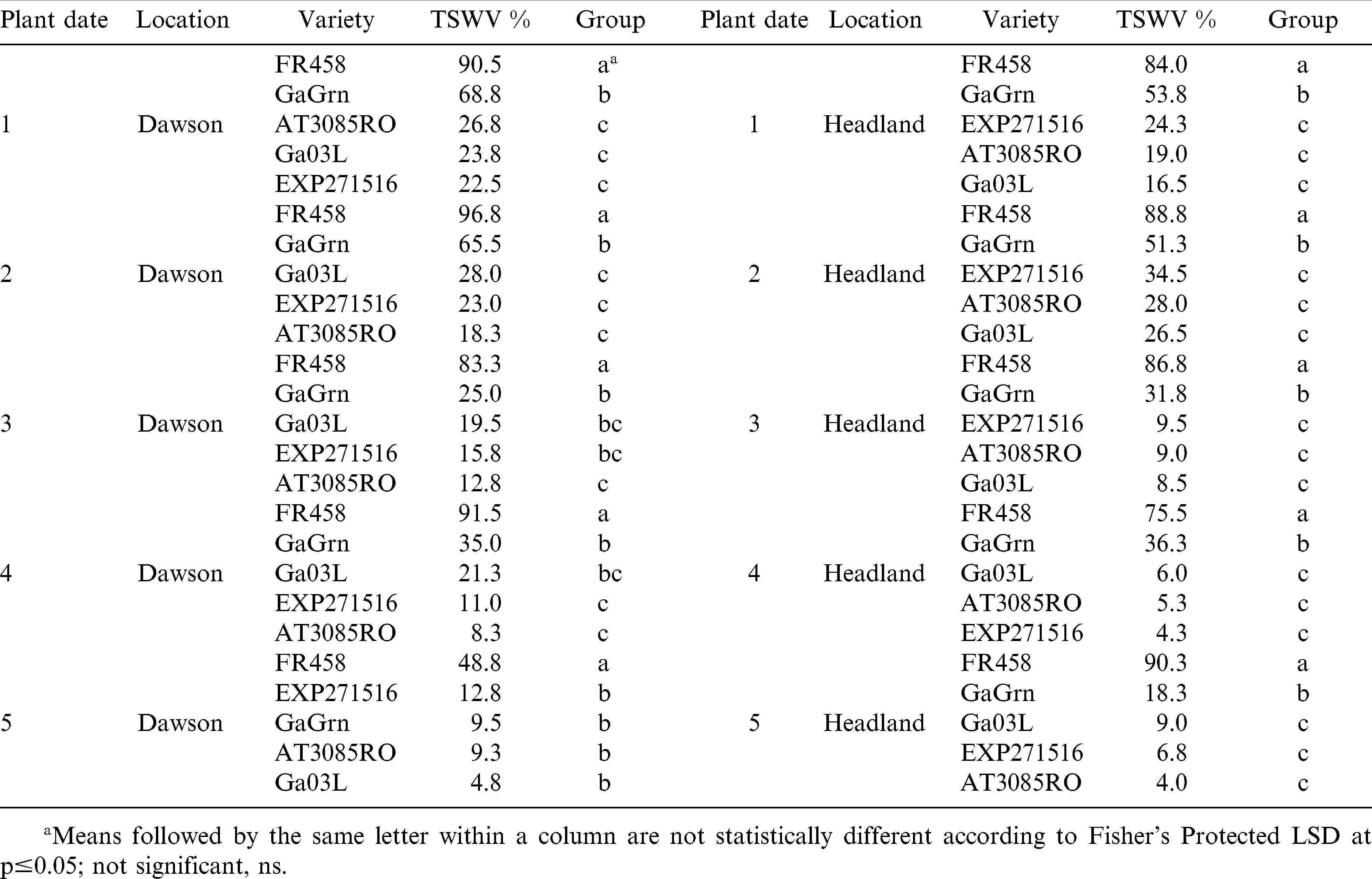
In 2008, foliar ratings recorded at mid-season (July 22 Dawson; July 25 Headland) for TSW were significant among genotypes for the first 3 planting dates at both locations (Table 3). Symptom ratings ranged between 1.3 and 15.8% for TSW tolerant genotypes across all planting dates at Dawson. In planting dates 1 and 2 at Dawson, Flavor Runner 458 showed 30–34% TSW, which was significantly higher than ratings for the other genotypes. The TSW ratings were much higher at Headland at mid-season resulting in a clear separation between Flavor Runner 458 and the tolerant genotypes in the first 3 planting dates. Ratings for TSW were considerably higher at mid-season (July 24 Dawson and Headland) in 2009 (Table 4). Scores for Flavor Runner 458 were as high as 87% and were significantly higher than other genotypes in 9 of 10 comparisons. Mid-season ratings in 2009 also revealed a clear difference in the susceptibility of Georgia Green compared to the more TSW tolerant genotypes in planting dates 1 and 2 in Dawson and planting dates 1, 2, and 4 in Headland. These results indicate that mid-season TSW symptoms can vary significantly over planting dates, locations, and years. The more tolerant genotypes have fewer TSW symptoms at mid-season compared to susceptible genotypes and later planting dates reduce symptomology to the point of absence in some cases.
Foliar TSW Ratings prior to Digging
At maturity in 2008, visual ratings for TSW ranged between 62 and 89% for the susceptible Flavor Runner 458 which showed significantly more TSW symptoms than other genotypes in 8 of 10 comparisons (Table 5). Ratings for Georgia Green were greater than those for the more tolerant genotypes in planting dates 1, 2, and 3 at Dawson and planting dates 2, 4, and 5 at Headland. Similar results were observed by Culbreath et al. (2008) between cultivars Georgia Green to Georgia-03L. The visual ratings for foliar TSW symptoms in 2008 were more strongly affected by genotype than planting date. The level of TSW symptoms did not drop off significantly over planting dates for the susceptible Flavor Runner 458 and high disease symptoms were maintained in the recommended planting date window (dates 3 and 4) for Georgia Green. Among the 3 newer and more TSW tolerant genotypes, Georgia-03L had higher incidence of TSW in planting date 4 of 2008 at Dawson.
In 2009, TSW incidence in planting dates 1 through 4 was significantly stronger in Dawson than that of planting date 5 (Table 6). Regardless of disease pressure, Flavor Runner 458 received significantly higher ratings than other genotypes in all 10 comparisons. In plating dates prior to the recommended planting window at Dawson, Georgia Green had higher ratings than the more TSW tolerant genotypes. In all five planting dates at Headland, Georgia Green was rated better than Flavor Runner 458 but significantly more infected than the 3 advanced genotypes indicating improved TSW tolerance. These results conclude that genotypes with advanced tolerance to TSW help reduce TSW incidence compared to the susceptible check. Planting later, in most cases, reduces TSW incidence in the tolerant genotypes.
Visual Ratings of Roots and Pods prior to Digging
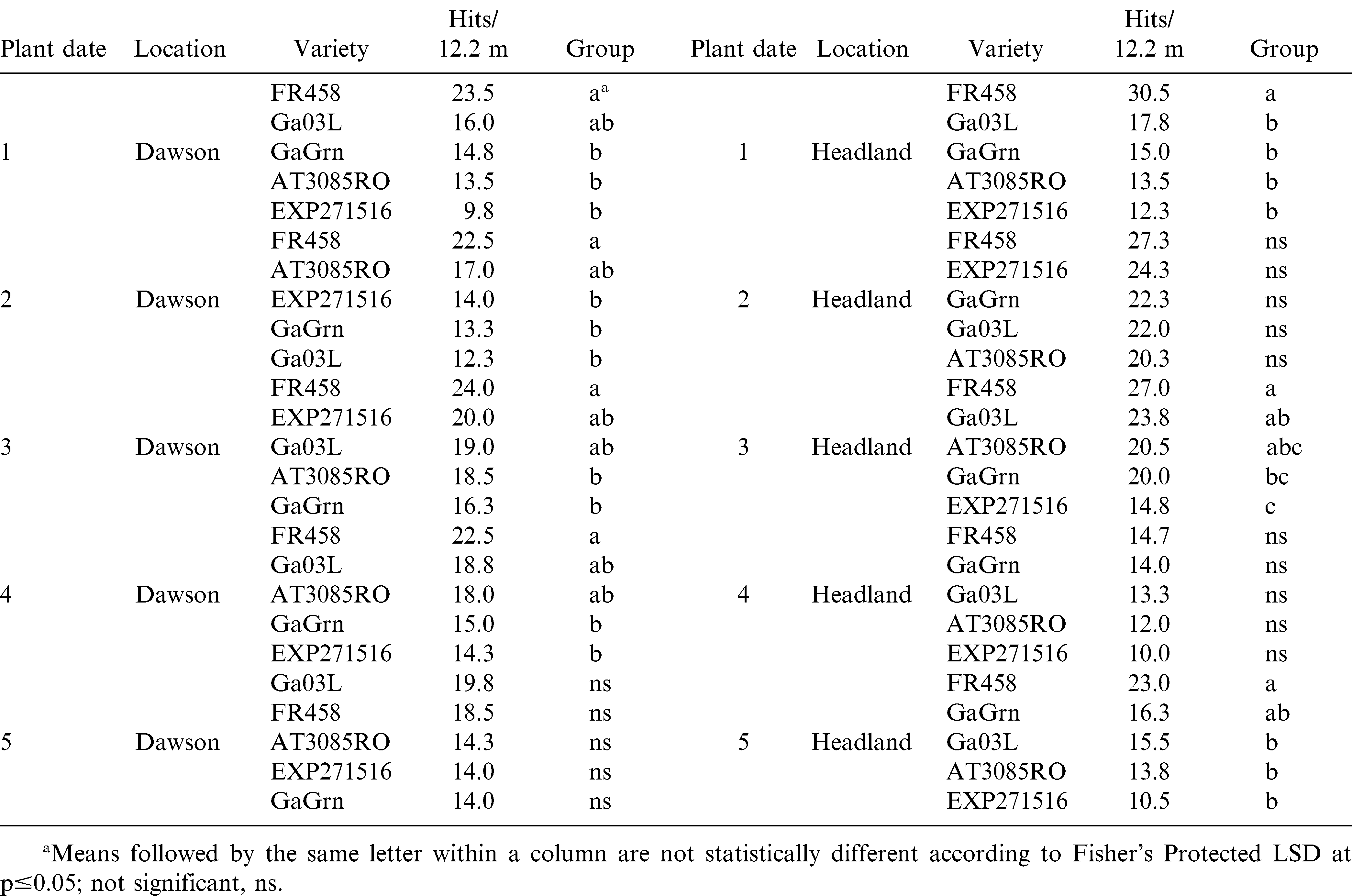
In 2008, Flavor Runner 458 had a strong trend for higher TSW symptoms on visual pod load, however was only significantly different than all other genotypes in planting date 1 at Headland (Table 7). Other significant differences between genotype for visual pod load rating in 2008 were not consistent between planting date or location.
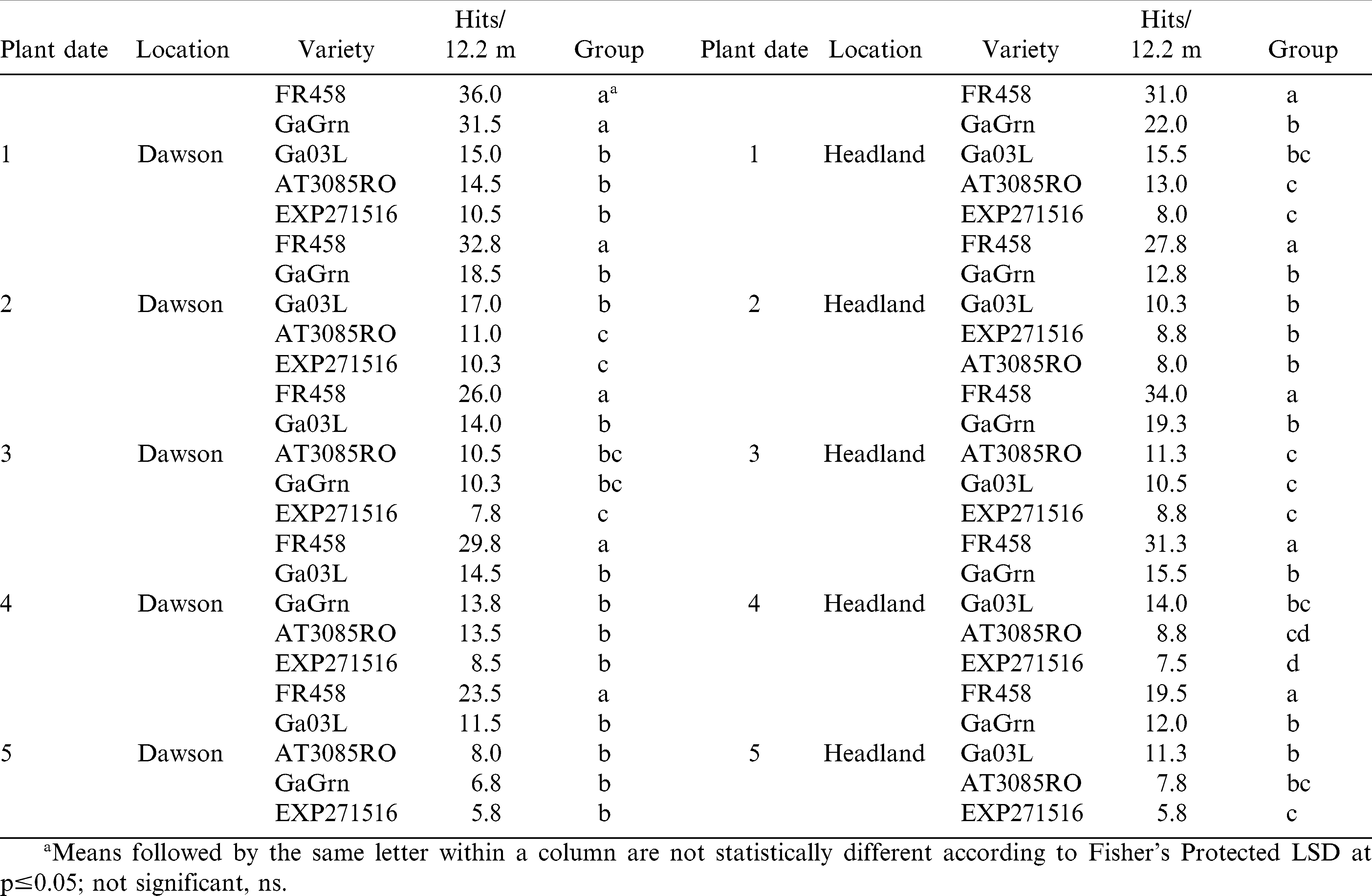
Genotype response for rating of TSW symptoms on pod load was more consistent in 2009 (Table 8). Visual yield loss was greater for Flavor Runner 458 compared to genotypes other than Georgia Green in all 10 comparisons. Ratings for Georgia Green were similar to the susceptible genotype in planting date 1 at Dawson. Among the 3 genotypes showing more foliar tolerance to TSW than Georgia Green, the advanced genotype, EXP 271516, consistently tended to maintain pod load over all planting dates at both locations and significantly protected visual pod load better than Georgia-03L in planting dates 2 and 3 at Dawson and planting dates 4 and 5 in Headland in 2009.
Yield and Grade
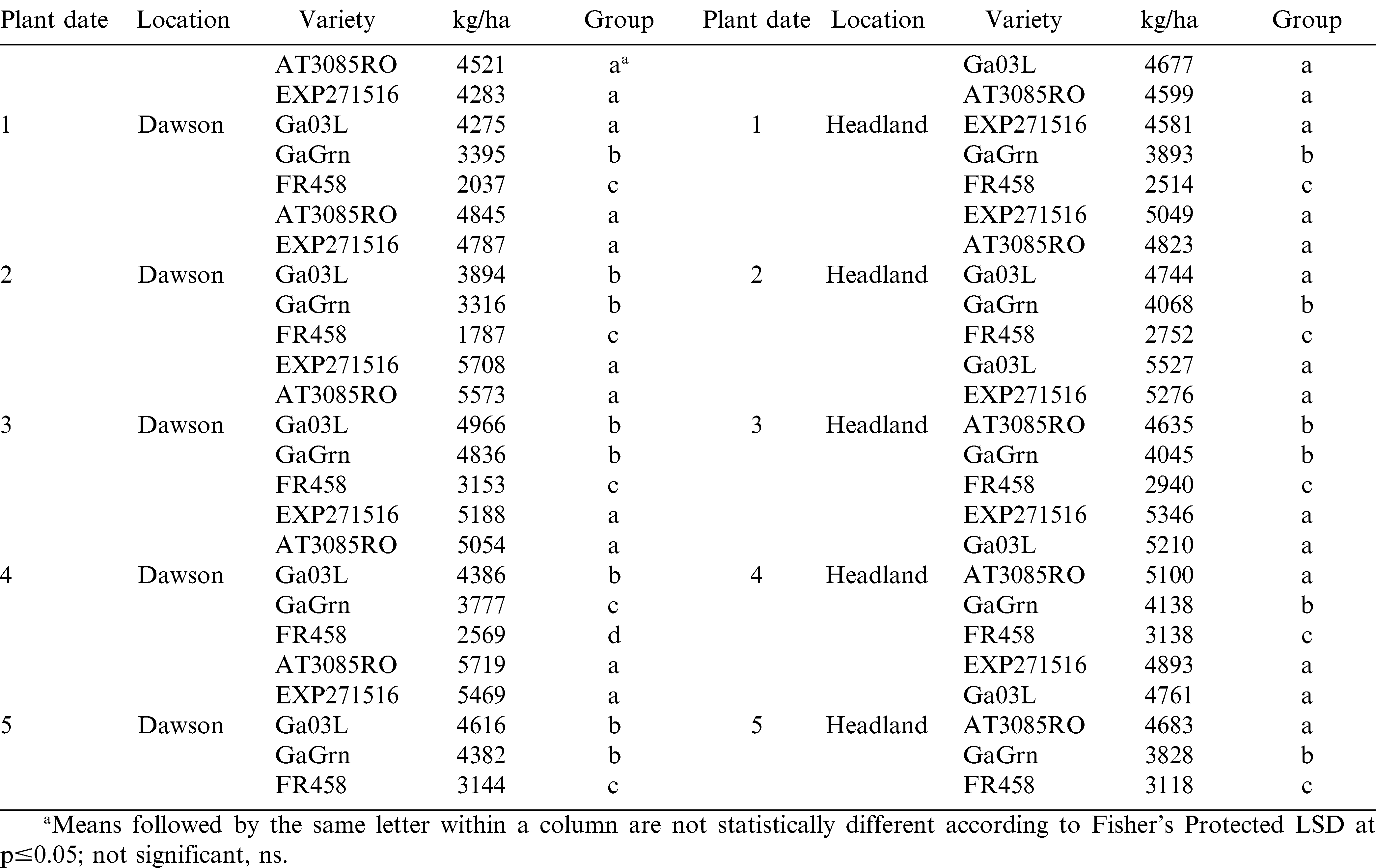
In 2008, planting dates 3, 4, and 5 provided the highest yields in both Dawson and Headland compared to planting dates 1 and 2 (Table 9). Although yield was higher for the more TSW tolerant genotypes compared to Flavor Runner 458, planting the more tolerant genotypes earlier than the recommended window significantly reduced their respective yield in 2008. In Headland, Georgia-03L produced an average of 730 kg/ha more than AT 3085RO in planting dates 1 through 4 and EXP 271516 also yielded 780 kg/ha more than AT 3085RO in planting date 4 at Headland. Yield produced by Georgia Green was often statistically similar to some of the more TSW tolerant genotypes. However, the trend was for Georgia Green to have intermediate yield potential over planting dates between the TSW susceptible genotype and the genotypes with greater TSW tolerance.
In 2009, Flavor Runner 458 had the lowest yield of all genotypes over planting dates and locations averaging 1250 kg/ha below Georgia green and 2190 kg/ha below the more tolerant genotypes (Table 10). Planting dates 3, 4, and 5 at Dawson had significantly higher yields by 855 kg/ha compared to planting dates 1 and 2, while planting dates 3 and 4 tended to be higher than the remaining planting dates in Headland by 335 kg/ha. Genotypes AT 3085RO and EXP 271516 yielded 825 kg/ha more than Georgia-03L in planting dates 2 through 5 at Dawson. In Headland, Georgia-03L and EXP 271516 had 680 kg/ha higher yield compared to AT 3085RO in planting date 3.
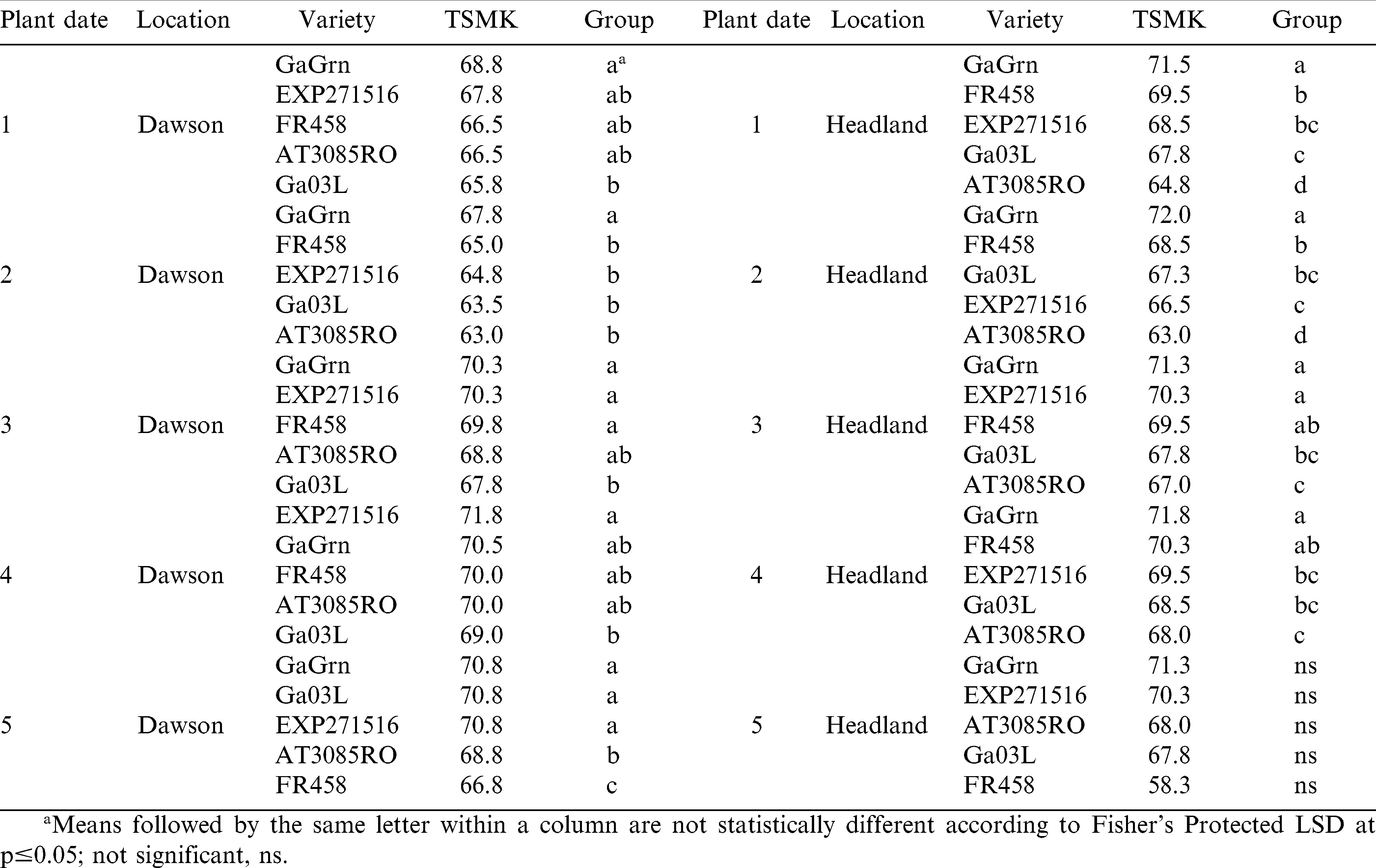
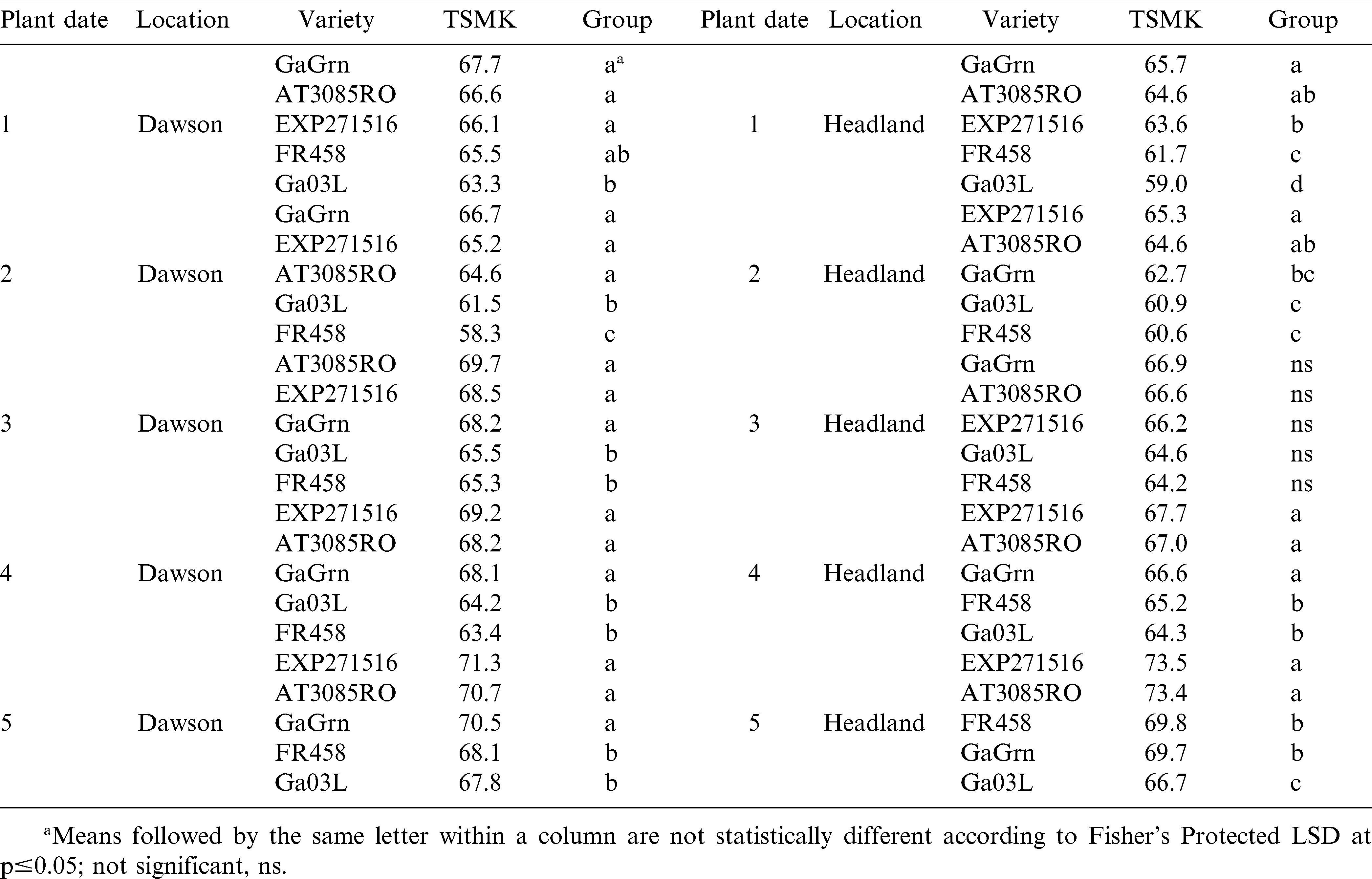
In 2008, Georgia Green had significantly higher grades than the other genotypes in planting date 2 (3.7 TSMK) at Dawson and in planting dates 1 and 2 in Headland (Table 11). In 2009, peanut grades were lower overall than in 2008 with Flavor Runner 458 in planting date 2 at Dawson and Georgia-03L in planting date 1 in Headland having abnormally low grades (Table 12). Peanut grade was affected more by peanut genotype than planting date.
Summary
The difference in magnitude of TSW incidence in several more tolerant genotypes compared to the susceptible entry in all planting dates is evidence of achieving greater genetic tolerance to the disease. The variability of ratings within a genotype between planting dates across site years is evidence of variable disease pressure. In this study, the overall trend was the later the planting date (planting dates 3, 4, and 5), the more likely that TSW foliar disease symptoms in tolerant genotypes would be reduced. The tolerance of Georgia-03L, AT 3085RO, and EXP 271516 was consistently higher than that of Georgia Green. Regardless of visual TSW disease tolerance and planting date, genetic yield potential has been improved through breeding efforts and following the recommended planting date window consistently improves yield over the earliest planting dates. The results of this research indicate that although greater TSW tolerance and yield potential does exists in the newer genotypes (Georgia-03L, AT 3085RO, and EXP 271516) (Culbreath et al., 2010), risk is best managed by planting the most TSW tolerant genotypes during the recommended planting dates as previously shown by Tillman et al. (2007).
Acknowledgements
Appreciation is given to Bobby Tennille, Sam Hilton, Larry Powell, Sonia Chestnut, Jason Blankenship, Bobby Hagler, Corey Collins, Jesse Bolton, Larry Wells, and Brian Gamble for technical input, plot management, data collection, and cooperation involved in conducting this research.
Disclaimer: Mention of trade names, cultivars, or commercial products is solely for the purpose of providing specific information and does not imply recommendation or endorsement by Dow AgroSciences, LLC or the U.S. Department of Agriculture, nor implies approval of a product to the exclusion of others that may be suitable.
Literature Cited
Beasley J.P 2007 Seeding rate, row patterns, and planting date In: Prostko E.P (ed.) 2007 Peanut update. CSS-03-0105. Georgia Coop. Ext. Serv ., Univ. of Georgia , Athens .
Branch W.D 1996 Registration of Georgia Green peanut Crop Sci 36 : 806 .
Branch W.D 2004 Registration of Georgia-03L peanut Crop Sci 44 : 1485 – 1486 .
Brown S.L Culbreath A.K Todd J.W Gorbet D.W Baldwin J.A and Beasley J.P 2005 Development of a method of risk assessment to facilitate integrated management of spotted wilt of peanut Plant Disease 89 : 348 – 356 .
Culbreath A.K Todd J.W Gorbet D.W Brown S.L Baldwin J Pappu H.R and Shokes F.M 2000 Reaction of peanut cultivars to spotted wilt Peanut Sci 27 : 35 – 39 .
Culbreath A.K Tillman B.L Gorbet D.W Holbrook C.C and Nischwitz C 2008 Response of new field resistant peanut cultivars to twin row pattern or in-furrow applications of phorate insecticide for management of spotted wilt Plant Dis 92 : 1307 – 1312 .
Culbreath A.K Tillman B.L Tubbs R.S Beasley J.P Kemerait R.C and Brenneman T.B 2010 Interactive effects of planting date and cultivar on tomato spotted wilt of peanut Plant Disease 97 : 898 – 904 .
Gomez K.A and Gomez A.A 1984 Statistical procedures for agricultural research John Wiley & Sons , New York .
Holbrook C.C Timper P Culbreath A.K and Kvien C.K 2008 Registration of Tifguard peanut J. Plant Registrations 2 : 92 – 94 .
Kemerait R.C Culbreath A.K Beasley J.P Prostko E.P Brenneman T.B Smith N Tubbs R.S Olatinwo R Srinivasan R Boureau M Tillman B.L Rowland D.L Dufault N.S Hagan A.K and Faircloth W.H 2011 The 2011 Version of the Peanut Disease Risk Index In: Beasley J.P (ed.) 2011 Peanut update. CSS-11-0110. Georgia Coop. Ext. Serv ., Univ. of Georgia , Athens .
Prostko E.P (ed.) 2007 2007 Peanut update Ext. Ann. Pub. CSS-03-0105. Univ. Ga. Coop. Ext. Ser ., Athens ).
Tillman B.L Gorbet D.W and Andersen P.C 2007 Influence of planting date on yield and spotted wilt of runner market type peanut Peanut Sci 34 : 79 – 84 .
Williams E.J and Drexler J.S 1981 A non-destructive method for determining peanut pod maturity Peanut Sci 8 : 134 – 141 .
Notes
- Cotton Development Specialist (Former USDA ARS Research Agronomist), Dow AgroSciences LLC, P.O. Box 120, Shellman, Georgia 39886
- Associate Professor, Auburn University, 201 Funchess Hall, Auburn, Alabama 36849
- Research Biochemist, USDA ARS, National Peanut Research Laboratory, 1011 Forrester Drive, SE, Dawson, Georgia 39842
- Peanut Breeder, Auburn University, 201 Funchess Hall, Auburn, Alabama 36849 * Corresponding author (email: rcnuti@dow.com).
Author Affiliations