Introduction
Peanut (Arachis hypogaea L.) has several unique features that contribute to challenging weed management. Peanut cultivars grown in the United States require a fairly long growing season (140 to 160 d), depending on cultivar and geographical region (Henning et al., 1982; Wilcut et al., 1995). Peanut also has a prostrate growth habit, a relatively shallow canopy, and is slow to shade inter-rows allowing weeds to be more competitive (Walker et al., 1989; Wilcut et al., 1995). Consequently, soil-applied herbicides may not provide season-long control and mid-to-late season weed pressure can occur. Additionally, peanut fruit develops underground on pegs originating from branches that grow along the soil surface. This prostrate growth habit and pattern of fruit development restricts cultivation to an early season control option (Brecke and Colvin, 1991; Wilcut et al., 1995). With conventional row spacing (91 to 102 cm), complete ground cover may not be attained until 8 to 10 wk after planting. In some areas of the United States peanut growing region, complete canopy closure may never occur.
Paraquat is one of the most frequently used postemergence (POST) herbicides in Southeastern United States peanut production systems but is seldom used in the Southwestern production region because crop injury may occur, reducing yield and grade characteristics (Knauft et al., 1990; Wilcut and Swann, 1990). The addition of bentazon to paraquat is a common practice to reduce peanut injury, although it may be either antagonistic or synergistic on weed control depending on the weed species and herbicide rate (Wehtje et al., 1992) and often does not improve peanut yield despite the reduction in crop injury (Wehtje et al., 1986, 1992; Wilcut et al., 1989). Due to the low price of paraquat, growers in the southwest continue to express an interest in its use (authors' personal observations). However, concerns have been expressed about peanut cultivar response to paraquat applications. Evaluations of cultivar response to herbicide treatments containing paraquat have been studied (Knauft et al., 1990; Wehtje et al., 1991; Wilcut and Swann, 1990).
Flumioxazin is a N-phenyl phthalamide soil-applied herbicide that received a federal label in the United States for use in peanut in 2001 (Grichar et al., 2004). Flumioxazin inhibits the enzyme protoporphyrinogen oxidase (Anderson et al., 1994; Senseman, 2007; Yoshida et al., 1991). In Georgia, flumioxazin applied preemergence (PRE) was shown to control morningglory spp. (Ipomoea spp.), prickly sida (Sida spinosa L.), and Florida beggarweed (Desmodium tortuosum (Sw.) DC) (Wilcut, 1997) while in Texas, pitted morningglory [Ipomoea lacunosa L.] were controlled greater than 75% (Grichar and Colburn, 1996). Flumioxazin has been reported to cause peanut injury especially when the application is delayed until peanut emergence (Johnson et al., 2006; Jordan et al., 2009; Tredaway-Ducar et al., 2009). When applied soon after peanut planting (1 to 2 d), Grichar et al. (2004) reported that flumioxazin plus metolachlor combinations, under cool, wet conditions resulted in peanut stunting which was evident throughout the growing season. They attributed this to increased uptake of flumioxazin and metolachlor with the heavy rainfall and the slowed metabolism of these herbicides as a result of cool temperatures (Yoshida et al., 1991). Askew et al. (1999) reported that flumioxazin at 0.07 and 0.11 kg ai/ha injured peanut 45 and 62%, respectively, when evaluated 2 wks after peanut planting. Peanut stunting of greater than 60% was followed by as much as 35% leaflet discoloration, which was characterized as necrotic spots on foliage. Scott et al. (2001) reported that flumioxazin treated peanuts were injured 10% when evaluated 3 wks after planting. However, injury was transient and was not appartent 6 wk after planting. Flumioxazin enters plants mainly by shoot and root uptake, and plant injury can be avoided via rapid metabolism (Yoshida et al., 1991; Anderson et al., 1994).
Smellmelon is becoming more of a problem in south Texas peanut production fields and has become a problem in several crops along the Texas Gulf coast (author's personal observation). The range of smellmelon stretches from Georgia to the southern part of California and as for north as Arkansas (SWSS, 1999). Smellmelon can be a problem at peanut harvest as the melon can become broken apart when run through the combine and increase drying time because of the high moisture content of the melon itself (author's personal observation). In IMI-tolerant corn (Zea mays L.), Thompson et al. (2005) reported that imazapic at 0.07 and 0.14 kg ai/ha applied either PRE, early POST, or late POST controlled smellmelon greater than 90%. Tingle and Chandler (2004) reported that smellmelon control was at least 93% with low-, medium-, and high-input herbicide systems. In cotton (Gossypium hirsutum L.), glyphosate systems have provided effective smellmelon control (Livingston et al., 2004; Livingston, 2006). Tingle et al. (2000) reported when smellmelon was allowed to compete with cotton for at least 6 wks, yield was reduced 7% compared to the weed-free check but when smellmelon was allowed to compete for 10 to 12 wks, cotton yield was reduced 22 and 27%, respectively.
Some growers in the southwest have questioned the feasibility of applying flumioxazin and paraquat at planting or soon after peanut emergence to control weeds that are not normally controlled with either herbicide alone and response of peanut cultivars to these herbicides and application timing. The product label states that PRE applications of flumioxazin must be made within 2 d after planting and prior to peanut emergence. Application after the peanut have begun to crack, or are emerged, will result in severe crop injury (Anonymous, 2009). Although off-label with respect to flumioxazin, the objective of this research was to evaluate the effect of flumioxazin and paraquat alone and in combination applied PRE or after peanut cracking for smellmelon (Cucumis melo L. var. Dudaim Naud.) control and peanut response.
Materials and Methods
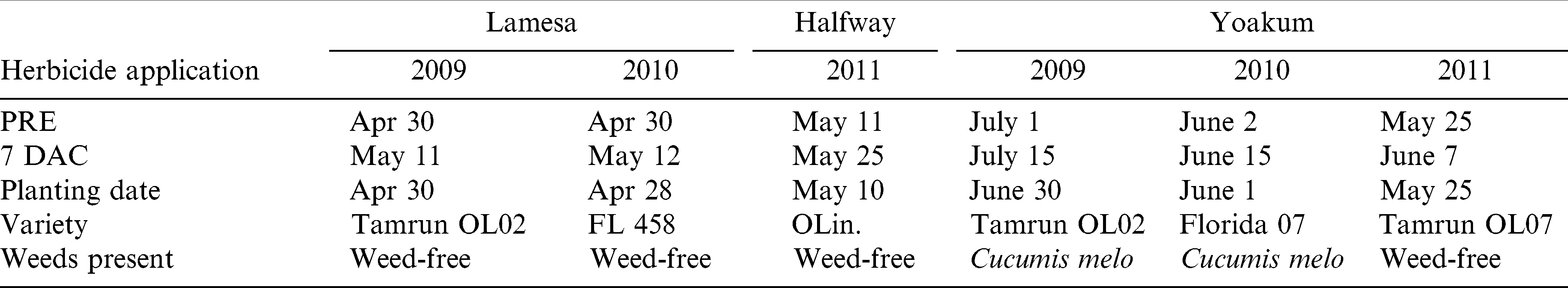
Smellmelon control and peanut tolerance studies were conducted in south Texas near Yoakum and in the Texas High Plains near Lamesa and Halfway with runner and Spanish market-types during the 2009 through 2011 growing seasons. The studies in south Texas in 2009 and 2010 were conducted in areas with high populations of smellmelon while the study in 2011 and at the High Plains locations in 2009 and 2010 were conducted under weed-free conditions. The soils at Yoakum were a Denhawken sandy clay loam (fine, smectitic, hyperthermic, Vertic Haplustepts, 1.6% organic matter, pH 7.6); at Lamesa the soils were a Amarillo fine sandy loam (fine-loamy, mixed, superactive, thermic Aridic Paleustalf, 0.4% organic matter, pH 7.8); and at Halfway the soils were a Acuff sandy clay loam (fine-loamy, mixed, superactive thermic Aridic Paleustolls) with < 0.8% organic matter and pH 7.7. Planting date, application dates, and other variables for each study are given in Table 1.
Treatments consisted of a factorial arrangement of two application timings (PRE and 7 days after peanut cracking [DAC]) and 8 herbicide treatments. Herbicide treatments included flumioxazin alone at 0.07 and 0.11 kg ai/ha, paraquat alone at 0.14 and 0.28 kg ai/ha, flumioxazin at 0.07 kg/ha plus paraquat at 0.14 or 0.28 kg/ha, and flumioxazin at 0.11 kg/ha plus paraquat at 0.14 or 0.28 kg/ha. An untreated check was also included for comparison.
Plot size was two rows (97 cm apart) by 9.5 m. at Yoakum and four rows (101 cm apart) by 7.3 m. at Lamesa and Halfway and the two middle rows per plot were sprayed and the other rows were left untreated to serve as buffers. All PRE applications of herbicide treatments including paraquat and all 7 DAC treatments included a non-ionic surfactant, Induce® [non-ionic surfactant, a blend of alkylarylpolyoxyalkane ether, free fatty acids, and isopropyl (90%), and water and formulation acids (10%); Helena Chemical Co., 225 Schilling Boulevard, Suite 300, Collierville, TN 38017] in south Texas or R-11® (90% akylphenol ethoxylate, butyl alcohol dimethylpolysiloxane; Wilbur-Ellis Company, P.O. Box 16458, Fresno, CA 93755) in the High Plains area at the rate of 0.25% v/v. Herbicides were applied in water using a CO2- pressurized backpack sprayer with TeeJet® 11002 DG (Spraying Systems Company, P.O. Box 7900, North Avenue, Wheaton, IL 60188) nozzles calibrated to deliver 190 L/ha at 180 kPa at Yoakum and TurboTee® 110015 nozzles calibrated to deliver 140 L/ha at 207 kPa at the Lubbock location.
Runner market-type peanut varieties ‘Tamrun OL02’ (Simpson et al., 2006) in 2009, ‘Florida 07’ (Gorbet and Tillman, 2009) in 2010 and ‘Tamrun OL07 (Baring et al., 2006) in 2011 were planted at Yoakum while at the High Plains locations, Tamrun OL02 was planted in 2009 and FlavorRunner 458 (Beasley and Baldwin 2009) was planted in 2010 at Lamesa. At the Halfway location the Spanish peanut OLin (Simpson et al., 2003) was planted.
Supplemental irrigation was supplied as needed at both locations. Traditional production practices were used to maintain peanut growth, development, and yield. In the weed-free studies, all plots received a dinitroaniline herbicide applied preplant incorporated and were cultivated and hand-weeded throughout the growing season to maintain weed-free conditions. Clethodim at 0.18 kg ai/ha + crop oil concentrate (COC) was applied POST to control annual grass escapes at the south Texas location. No insecticides were needed at any location in any year.
Peanut stunting was evaluated approximately 60 d after peanut planting with the runner peanuts cultivars in south Texas and the High Plains area, while the Spanish cultivars were rated prior to peanut digging. Peanut stunting were based on a scale of 0 (no peanut stunting) to 100 (peanut death). Peanut yield was determined by inverting the pods based on maturity of untreated control plots, air-drying in the field for 6 to 10 d, and harvesting individual plots with a combine. Yield samples were cleaned and adjusted to 10% moisture. For grades, a 200-g pod sample from each plot was obtained and grades determined following procedures described by the Federal-State Inspection Service (USDA, 1993).
Data for percentage of peanut injury and stunting were transformed to the arcsine square root prior to analysis; however, nontransformed means are presented because arscine transformation did not affect interpretation of the data. Data were subjected to ANOVA and analyzed using SAS PROC MIXED (SAS 2002). Treatment means were separated using Fisher's Protected LSD at P ≤ 0.05. The untreated check was used for peanut yield and grade calculation comparison and a visual comparison for peanut injury and was only included in yield and grade analysis.
Results and Discussion
Smellmelon control
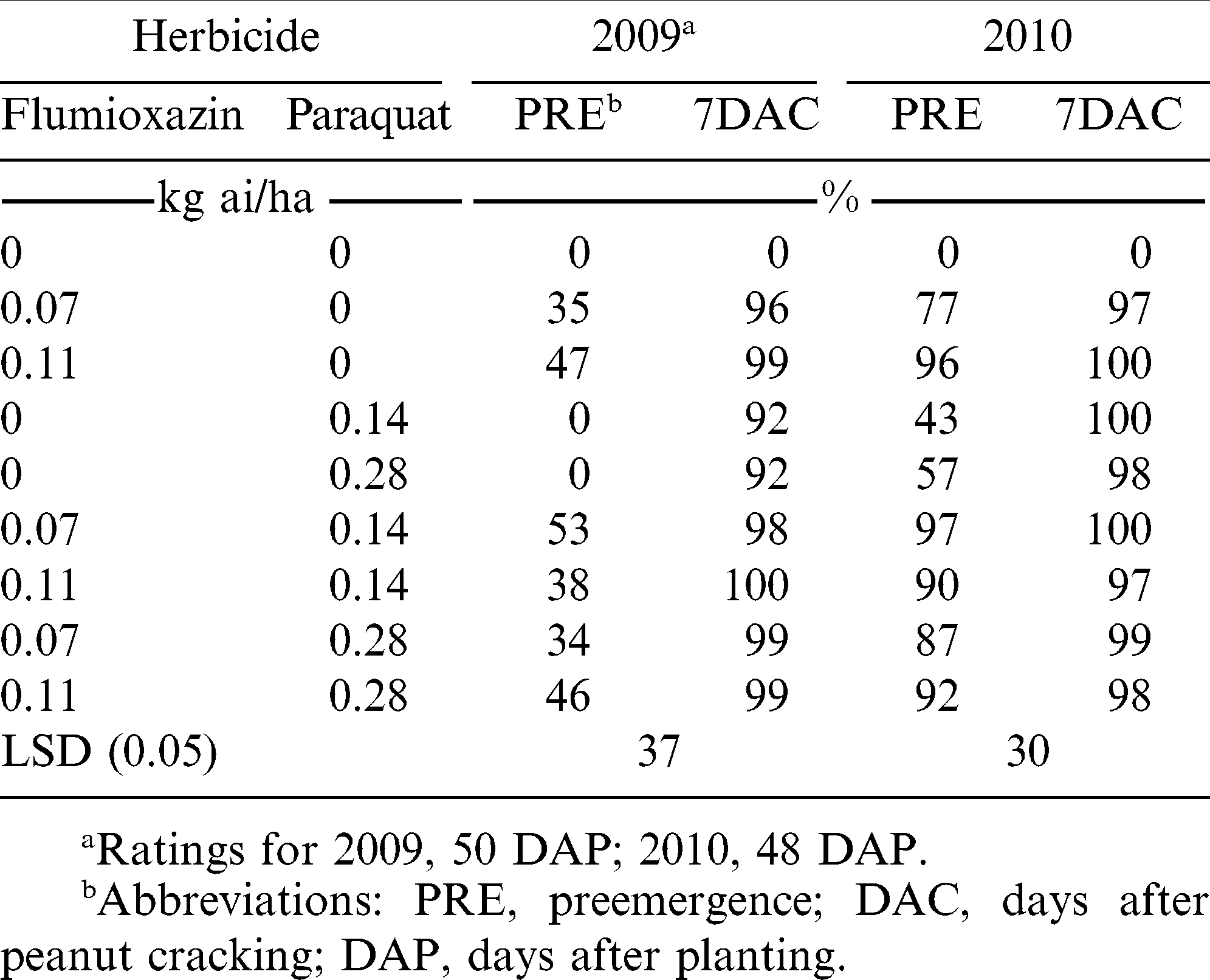
There was a treatment by year interaction for smellmelon control; therefore, that data were presented separately by year. In 2009, PRE applications of flumioxazin with/without paraquat failed to control smellmelon ( ≤ 53%) while flumioxazin or paraquat alone or in combination controlled smellmelon at least 92% when applied 7 DAC (Table 2). In 2010, flumioxazin alone applied PRE at 0.07 kg/ha controlled smellmelon 77% while flumioxazin at 0.11 kg/ha controlled 96% smellmelon. The addition of paraquat to flumioxazin PRE controlled smellmelon 87 to 97% while all flumioxazin or paraquat treatments alone or in combination applied 7 DAC controlled smellmelon at least 97% (Table 2). Effective control of smellmelon may be attributed to the amount of flumioxazin absorbed. Price et al. (2004) reported that the total 14C-flumioxazin absorbed by ivyleaf morningglory (Ipomoea hederacea Jacq) was 57% of applied while sicklepod (Senna obtusifolia L.) absorbed 46% when evaluated 72 h after application and that a majority of this remained in the roots.
Early-season peanut injury
Since different peanut cultivars were used in each year of the study no attempt was made to combined peanut injury data over years. No peanut injury was observed with any PRE applications either in south Texas or the Texas High Plains (data not shown).
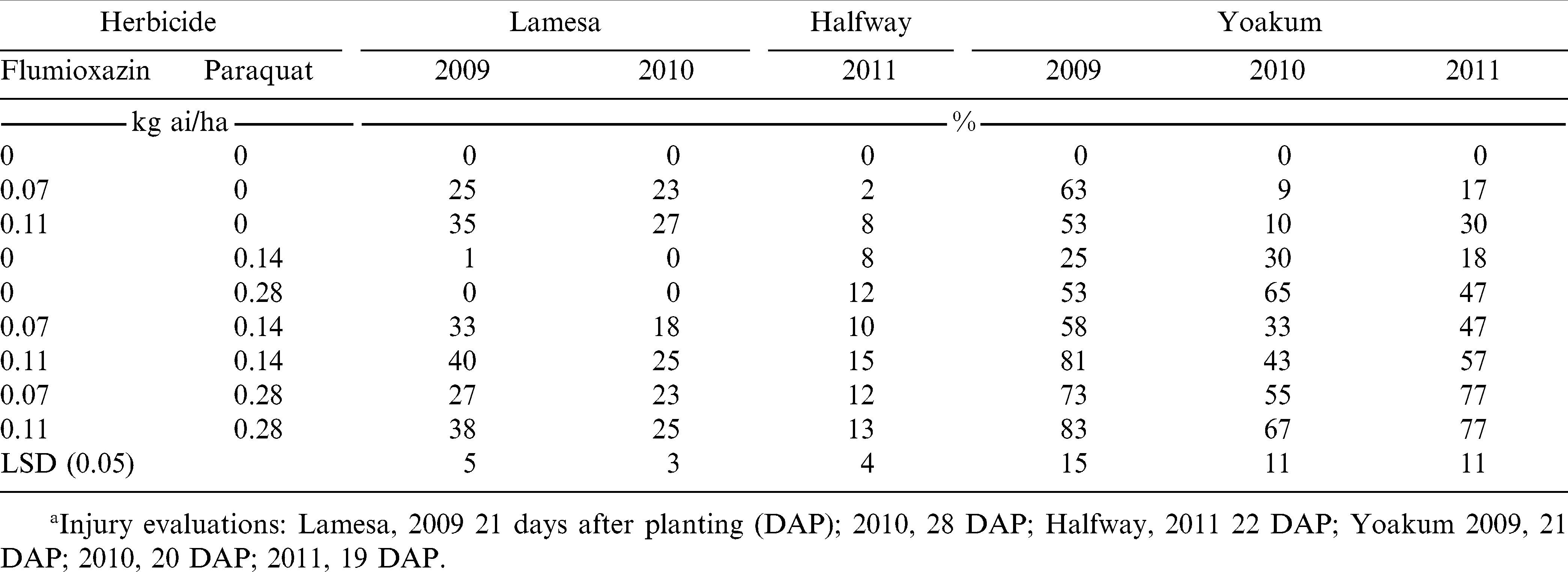
Peanut injury from the 7 DAC treatments consisted of peanut stunting, leaf chlorosis and necrosis, and leaf bronzing that is typical of flumioxazin and paraquat injury (Grichar et al., 2004; Wehtje et al., 1992; Wilcut et al., 1995; Johnson et al., 2006). At Lamesa in 2009, flumioxazin alone applied 7 DAC resulted in 25 to 35% peanut injury depending on rate while combinations of flumioxazin plus paraquat resulted in 27 to 40% injury and this injury increased as the rate of flumioxazin increased regardless of paraquat rate (Table 3). In 2010 at Lamesa, 7 DAC applications of flumioxazin alone or flumioxazin plus paraquat caused at least 18% injury. No injury was observed with paraquat alone. In 2011 at Halfway, peanut injury with flumioxazin at 0.07 kg/ha was no different from the untreated check. Flumioxazin at 0.11 kg/ha plus paraquat at 0.14 kg/ha resulted in 15% peanut injury which was greater than flumioxazin alone, paraquat at 0.14 kg/ha, or a combination of flumioxazin at 0.07 kg/ha plus paraquat at 0.14 kg/ha.
At Yoakum in 2009, flumioxazin or paraquat alone and combinations of flumioxazin plus paraquat resulted in at least 53% peanut injury with the exception of paraquat alone at 0.14 kg/ha which resulted in 25% injury (Table 3). In 2010, peanut injury with flumioxazin alone was no different than the untreated check while paraquat alone at 0.14 kg/ha or flumioxazin at 0.07 kg/ha plus paraquat at 0.14 kg/ha caused 30 and 33% injury which was less than combinations that included paraquat at 0.28 kg/ha. In 2011, all herbicides caused peanut injury which was greater than the untreated check with the greatest injury observed with combinations that included paraquat at 0.28 kg/ha.
Mid-season peanut injury
As observed with the early-season peanut injury, PRE herbicide treatments resulted in no peanut injury in either area (data not shown). Mid-season injury from the 7 DAC treatments consisted of peanut stunting and peanut leaflet necrosis and chlorosis.
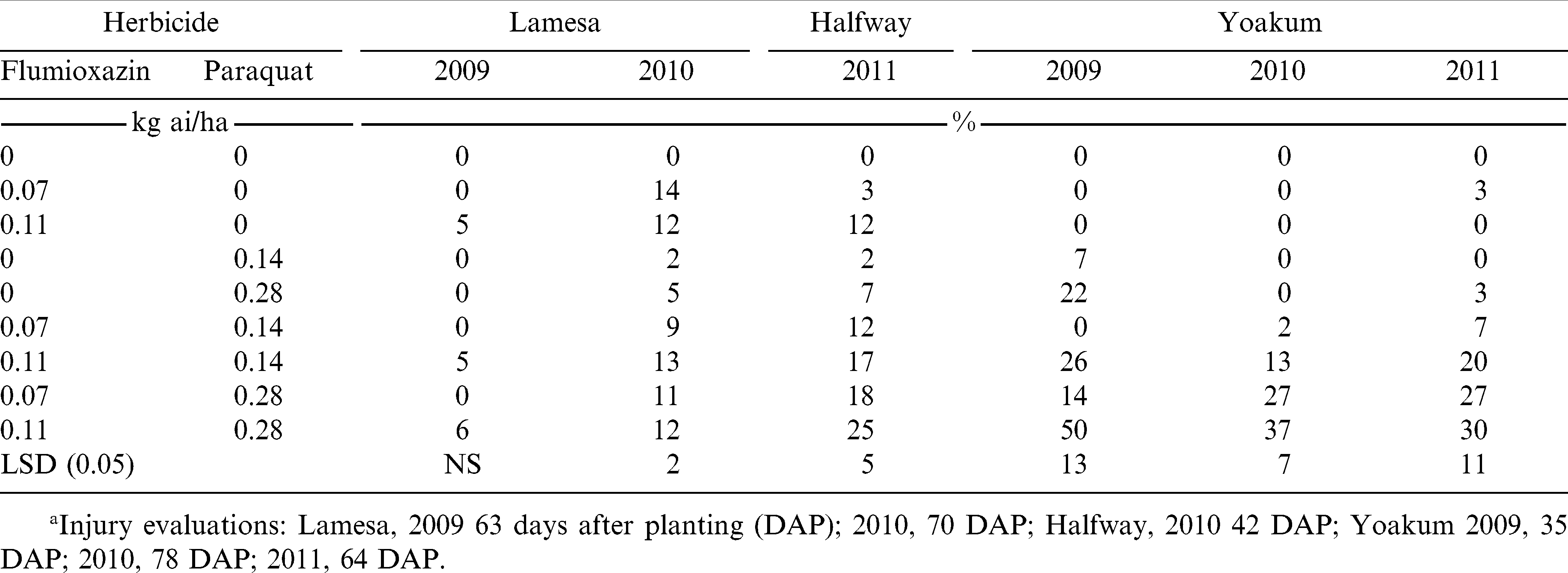
At Lamesa in 2009, no mid-season peanut injury was observed with any 7 DAC herbicide treatments (Table 4). In 2010, all herbicide treatments with the exception of paraquat alone at 0.14 kg/ha caused peanut injury which ranged from 5 to 14%. In 2011 at Halfway, peanut injury was greatest with the combination of flumioxazin at 0.11 kg/ha plus paraquat at 0.28 kg/ha while injury with flumioxazin alone at 0.07 kg/ha or paraquat alone at 0.14 kg/ha was 3% or less.
At Yoakum in 2009, paraquat alone at 0.28 kg/ha and herbicide combinations with the exception of flumioxazin at 0.07 kg/ha plus paraquat at 0.14 kg/ha resulted in 14 to 50% injury (Table 4). In 2010, herbicide combinations, with the exception of flumioxazin at 0.07 kg/ha plus paraquat at 0.14 kg/ha resulted in injury that was greater than the untreated check. Peanut injury was greatest with combinations that included paraquat at 0.28 kg/ha. Similar trends were noted in 2011 (Table 4). In work in the southeast, Johnson et al. (2006) reported that flumioxazin at 0.07 kg/ha applied 8 and 10 days after planting (DAP), injured peanut up to 39% at midseason. They also reported that flumioxazin at 1.05 kg/ha applied 4 DAP or later caused up to 49% peanut injury. However, despite the injury, there was no effect of flumioxazin application timing or rate on peanut stand. The flumioxazin label states that PRE applications of this product must be made within 2 d after planting and prior to peanut emergence (Anonymous, 2009).
In other studies, herbicide systems that included paraquat have resulted in severe peanut stunting while the herbicide system which included flumioxazin plus S-metolachlor applied PRE followed by lactofen applied POST caused moderate peanut stunting (Grichar and Dotray 2012). Although paraquat is registered for use on peanut within 28 d after emergence, Johnson et al. (1993) stated that paraquat use later than 28 d after emergence is discouraged due to the potential for crop injury with less time for plant recovery. Although paraquat was applied within the safety window excessive stunting was still observed. Peanut cultivar response to paraquat was thought to be the issue; however, Wehtje et al. (1991) reported that foliar absorption and translocation of paraquat did not vary between peanut cultivars and they attributed yield differences to the difference in yield potential between cultivars.
Peanut yield response
Since different peanut cultivars were used in each year of the study, no attempt was made to combine yield results over years. Peanut yields were not obtained from the Yoakum location in 2009 and 2010 due to the high populations of smellmelon which prevented proper digging of plots. Weed biomass slows field-drying of peanut vines and pods and increases the likelihood of exposure to rainfall, which can increase harvesting losses (Young et al., 1982; Wilcut et al., 1995). The fibrous root system and vines of smellmelon is extremely difficult to separate from peanut (Wilcut et al., 1994).
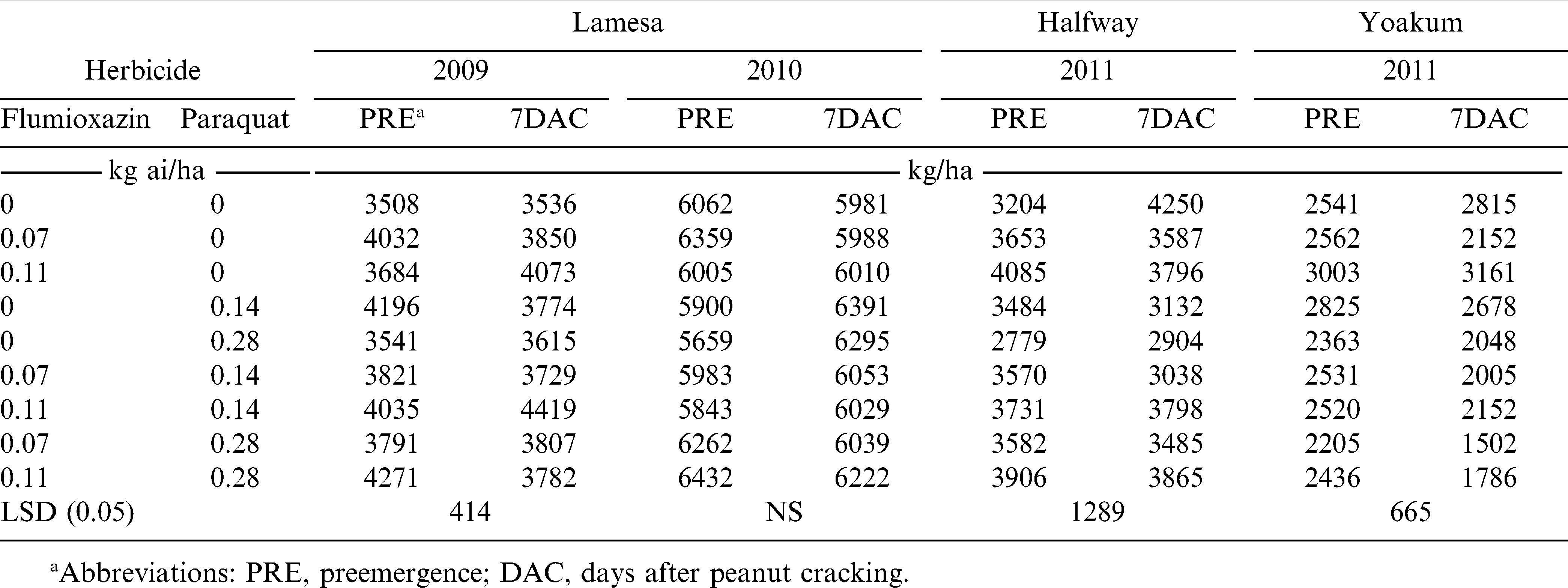
There was a herbicide by application timing interaction for yield; therefore those interactions are presented for each location where yield was obtained. In 2009, peanut yield increases from the untreated check (weed-free) were noted with flumioxazin alone at 0.07 kg/ha applied PRE or 0.11 kg/ha applied 7 DAC, paraquat alone at 0.14 kg/ha applied PRE, flumioxazin at 0.11 kg/ha plus prarquat 0.14 kg/ha applied PRE and 7 DAC, and flumioxazin at 0.11 kg/ha plus paraquat at 0.28 kg/ha applied PRE (Table 5). No yield reductions from the untreated check were noted with any other herbicide treatments. In 2010 at Lamesa, no differences in peanut yield were noted with any herbicide treatments whether applied PRE or 7 DAC (Table 5). However, at Halfway in 2010, paraquat alone at 0.28 kg/ha applied 7 DAC produced a lower yield than the untreated check for the 7 DAC applications. In 2011 at Yoakum, paraquat alone at 0.28 kg/ha, flumioxazin at 0.07 kg/ha plus paraquat at 0.14 or 0.28 kg/ha, or flumioxazin at 0.11 kg/ha plus paraquat at 0.28 kg/ha resulted in reduced yields compared with the untreated check for 7 DAC treatments (Table 5).
Early-season peanut injury (Table 3) with 7 DAC treatments did not always correspond to a reduction in peanut yield (Table 5). Only at one instance at Halfway and also at Yoakum did the severe early season injury result in any effect on peanut yield. Other studies have not reported any yield reductions with the use of paraquat (Carley et al., 2009; Johnson et al., 1993; Wehtje et al., 1991). Previous research suggests that paraquat may affect peanut grade (especially Virginia types) by increasing the proportion of other kernels (Knauft et al., 1990), which may indicate that the herbicide affected grade by delaying maturity (Carley et al., 2009).
Wilcut et al. (2001) conducted field studies on the response of flumioxzin at 0.07 kg/ha to eight Virginai peanut cultivars and reported that flumioxain did not affect percentage grade or total yield of any cultivar. Johnson et al. (2006) reported under weed-free conditions flumioxazin applied within the recommended time interval (0 to 2 DAP) temporarily stunted early season growth, but peanut generally recovered by midseason with no yield reductions. They concluded if given ample time to compensate for delays in maturity, peanut yield should not be affected. Similar results were also reported by Jordan et al. (2009) in which they found that delaying the application of flumioxazin until peanut emergence increased injury regardless of rate but pod yield was not affected.
In conclusion, the use of flumioxazin plus paraquat applied PRE is a safe option, but the use of flumioxazin plus paraquat applied 7 DAC may result in significant peanut injury and as a consequence cause some yield reductions, most notably in the south Texas peanut growing region. Peanut injury with flumioxazin may also be enhanced by the high humidities that are common to the south Texas region. The activity of POST herbicides is usually favored under warm, humid conditions (Martinson et al., 2005). In general, a high relative humidity during and after herbicide application is likely to increase herbicide penetration and absorption and increase the probability of weed mortality (Hammerton, 1967; Koukkari and Johnson, 1979; Norsworthy et al. 1999; Prasad et al., 1967). Dew is another environmental factor that can impact the effect of herbicides on plants (Nalewaja et al., 1975). Nalewaja et al. (1975) concluded that dew may lead to increased uptake of herbicides; however, excessive dew can lead to the herbicide being washed off, thus reducing crop injury. Times of herbicide application in the south Texas region ranged from 6:30 AM to 8:30 AM when dews typically develop while in the High Plains area, herbicides were applied from 9:30 AM until noon (data not shown). Under most conditions, little or no dew develops in this region and will surely not be present at these later times.
Literature Cited
Anderson R.J Norris A.E and Hess F.D 1994 Synthetic organic chemicals that act through the prophyrin pathway In: Duke S.O and Rebeiz C.A (eds.) Porphyric Pesticides: Chemistry, Toxicity, and Pharmaceutical Applications ACS Symposium Series 559 Washington, DC. Amer. Chem. Soc. pp. 18 – 33 .
Anonymous 2009 Valor® WP Herbicide Product Label Walnut Creek, CA:, Valent USA .
Askew D.S Wilcut J.W and Cranmer J.R 1999 Weed management in peanut (Arachis hypogaea) with flumioxazin preemergence Weed Technol. 13 : 594 – 598 .
Baring R M Simpson C.E and Burow M.D 2006 Registration of ‘Tamrun OL07’ peanut Crop Sci. 46 ( 6 ): 2721 – 2722 .
Beasley J and Baldwin J 2009 Peanut cultivar options and descriptions, 2009 http://www.uga/commodities/fieldcrops/peanuts/production/cultivardescription.html. accessed March 21, 2011 .
Brecke B.J and Colvin D.L 1991 Weed management in peanuts In: Pimentel D (ed.) CRC Handbook of Pest Management in Agriculture 2nd edition, CRC Press , Boca Raton, FL . pp. 239 – 251 .
Carley D.S Jordan D.L Brandenburg R.L and Dharmasri L.C 2009 Factors influencing response of Virginia market type peanut (Arachis hypogaea) to paraquat under weed-free conditions Peanut Sci. 36 : 180 – 189 .
Gorbet D.W and Tilman B.L 2009 Registration of ‘Florida-07’ peanut J. Plant Registrations. 3 : 14 – 18 doi: 10.3198/jpr2008.05.0276crc .
Grichar W.J and Colburn A.E 1996 Flumioxazin for weed control in Texas peanuts (Arachis hypogaea L.) Peanut Sci. 23 : 30 – 36 .
Grichar W.J Besler B.A Dotray P.A Johnson W.C and Prostko E.P 2004 Interaction of flumioxazin with dimethenamid or metolachlor in peanut (Arachis hypogaea L.) Peanut Sci. 31 : 12 – 16 .
Grichar W.J and Dotray P.A 2012 Weed control and peanut tolerance with ethalfluralin-based herbicide systems Int. J. Agronomy doi:10.1155/2012/597343 .
Jordan D.L Lancaster S.H Lanier J.E Lassiter B.R and Johnson P.D 2009 Peanut and eclipta (Eclipta prostrate) response to flumioxazin Weed Technol. 23 : 231 – 235 .
Johnson W.C Prostko E.P and Mullinix B.G 2006 Phytotoxicity of delayed applications of flumioxazin on peanut (Arachis hypogaea) Weed Technol. 20 : 157 – 163 .
Hammerton J.L 1967 Environmental factors and susceptibility to herbicides Weeds. 15 : 330 – 335 .
Henning R.J Allison A.H and Tripp L.D 1982 Cultural practices In: Pattee H.E and Young C.T (eds.) Peanut Science and Technology American Peanut Research Education Society, Incorporated Yoakum, TX . pp. 123 – 138 .
Johnson W.C Chamberlin J.R Brenneman T.B Todd J.W Mullinix B.G and Cardina J 1993 Effects of paraquat and alachlor on peanut (Arachis hypogaea) growth, maturity, and yield Weed Technol. 7 : 855 – 959 .
Knauft D.A Colvin D.L and Gorbet D.W 1990 Effect of paraquat on yield and market grade of peanut (Arachis hypogaea) genotypes Weed Technol. 4 : 866 – 870 .
Koukkari W.L and Johnson M.A 1979 Oscillations of leaves of Abutilon theophrasti (velvetleaf) and their sensitivity to bentazon in relation to low and high humidity Physiologia Plantarum. 47 : 158 – 162 .
Livingston S.D Janak J.D and Matthies A.Z 2004 Early and late-season suppression and control of Texas smellmelon in cotton Proc. South. Weed Sci. Soc. 57 : 27 .
Livingston S.D 2006 Over-the-top and lay-by herbicide treatments to control silverleaf nightshade, desert horse purslane, and Texas smellmelon in RR, RR-Flex, and Liberty Link transgenic cotton Proc. South. Weed Sci. Soc. 59 : 225 .
Martinson K.B Durgan B.R Gunsolus J.L and Sothern R.B 2005 Time of day of application effect on glyphosate and glufosinate efficacy Crop Management doi:10.1094/CM-2005-0718-02-RS .
Nalewaja J.D Pudelko J and Adamczewski K.A 1975 Influence of climate and additives on bentazon Weed Sci. 23 : 504 – 507 .
Norsworthy J.K Lawrence R Oliver R and Purcell L.C 1999 Diurnal leaf movement effects spray interception and glyphosate efficacy Weed Technol. 13 : 466 – 470 .
Prasad R Foy C.L and Crafts A.S 1967 Effects of relative humidity on absorption and translocation of foliarly applied dalapon Weeds 15 : 149 – 156 .
Price A.J Wilcut J.W and Cranmer J.R 2004 Physiological behavior of root-absorbed flumioxazin in peanut, ivyleaf morningglory (Ipomoea hederacea), and sicklepod (Senna obtusifolia) Weed Sci. 52 : 718 – 724 .
SAS Institute 2002 SAS User's Guide Version 9.1 Cary, NC. Statiscal Analysis System Institute Inc., 1028 pp.
Scott G.H D Askew S and Wilcut J.W 2001 Economic evaluation of diclosulam and flumioxazin systems in peanut (Arachis hypogaea) Weed Technol. 15 : 360 – 364 .
Senseman S.A 2007 Herbicide handbook. 9th edition Weed Sci. Soc. of America Lawrence, KS 458 pp.
Simpson E C Baring M.R Melouk H.A Lopez A and Kirby J.S 2003 Registration of ‘Olin’ peanut Crop Sci. 43 ( 6 ): 1880 – 1881 .
Simpson E.C Baring M.R Schubert A.M Black M.C Melouk H.A and Lopez Y 2006 Registration of ‘Tamrun OL 02’ peanut Crop Sci. 46 : 1813 – 1814 .
[SWSS] Southern Weed Science Society 1999 Weed Identification Guide Weed Identification Committee, C.D. Elmore, chairman and ed Champaign, IL 450 pp.
Thompson A.M Rosales-Robles E Chandler J.M Nester P.R and Tingle C.H 2005 Crop tolerance and weed management systems in imidazolinone-tolerant corn (Zea mays L.) Weed Technol. 19 : 1037 – 1044 .
Tingle C.H Chandler J.M Jones C.A and Steele G.L 2000 Competition and control of smellmelon (Cucumis melo L. Var dudaim Naud.) in cotton Proc. South. Weed Sci. Soc. 53 : 207 – 208 .
Tingle C.H and Chandler J.M 2004 The effect of herbicides and crop rotations on weed control in glyphosate-resistant crops Weed Technol. 18 : 940 – 946 .
Tredaway-Ducar J Clewis S.B Wilcut J.W L Jordan D Brecke B.J Grichar W.J Johnson W.C and Wehtje G.R 2009 Weed management using reduced rate combinations of diclosulam, flumioxazin, and imazapic in peanut Weed Technol. 23 : 236 – 242 .
USDA 2008 Farmers Stock Peanut Inspection Instructions Agric. Marketing Serv. Washington, D.C .
Walker R.H Wells L.W and McGuire J.A 1989 Bristly starbur (Acanthospermum hispidum) interference in peanuts (Arachis hypogaea) Weed Sci. 37 : 196 – 200 .
Wehtje G Mcguire J Walker R.H and Patterson M.G 1986 Texas panicum (Panicum texanum) control in peanuts (Arachis hypogaea) with paraquat Weed Sci. 34 : 308 – 311 .
Wehtje G Wilcut J.W McGuire J.A and Hicks T.V 1991 Foliar penetration and phytotoxicity of paraquat as influenced by peanut cultivar Peanut Sci. 18 : 67 – 71 .
Wehtje G Wilcut J.W and McGuire J.A 1992 Influence of bentazon on the phytotoxicity of paraquat to peanuts (Arachis hypogaea) and associated weeds Weed Sci. 40 : 90 – 95 .
Wilcut J.W Wehtje G Hicks T.V and McGuire J.A 1989 Postemergence weed control systems without dinoseb for peanuts Weed Sci. 37 : 385 – 391 .
Wilcut J.W and Swann C.W 1990 Timing of paraquat applications for weed control in Virginia-type peanuts (Arachis hypogaea) Weed Sci. 38 : 558 – 562 .
Wilcut J.W York A.C and Wehtje G.R 1994 The control and interaction of weeds in peanut (Arachis hypogaea) Review of Weed Sci. 6 : 177 – 205 .
Wilcut J.W York A.C Grichar W.J and Wehtje G.R 1995 The biology and management of weeds in peanut (Arachis hypogaea) In: Pattee H.E and Stalker H.T (eds.) Advances in Peanut Science Amer Peanut Res. Educ. Soc. Stillwater, OK . pp. 207 – 244 .
Wilcut J.W 1997 Summary of flumioxazin performance in southeastern peanuts Proc. South. Weed Sci. Soc. 50 : 7 .
Wilcut J.W Askew S.D Bailey W.A Spears J.F and Isleib T.G 2001 Virginia market-type peanut (Arachis hypogaea) cultivar tolerance and yield response to flumioxazin preemergence Weed Technol. 15 : 137 – 140 .
Yoshida R.M Sakaki R Sato R Haga T Nogano E Oshio H and Kamoshita K 1991 S-53482-a new N-phenyl phthalimide herbicide Proc. Brighton Crop Protect. Conf. Weeds. 1 : 69 – 75 .
Young J.H Person N.K Donald J.O and Mayfield W.H 1982 Harvesting, curing, and energy utilization In: Pattee H.E and Young C.T (eds.) Peanut Science and Technology American Peanut Research Education Society Yoakum, TX . pp. 458 – 487 .
Notes
- Senior Research Scientist Texas A&M AgriLife Research, 10345 State Hwy 44, Corpus Christi, TX 78406.; Professor, Texas Tech University, Texas A&M AgriLife Research, and Texas A&M AgriLife Extension Service, Lubbock, TX79403 * Corresponding author (email: w-grichar@tamu.edu)
Author Affiliations