Introduction
Wild species of Arachis are native to a large region of South America in tropical and subtropical areas. Eighty species have been named (Krapovickas and Gregory, 1994; Valls and Simpson, 2005) and others will likely be described from uncollected regions of South America. Wild peanut species have been important to man since before the cultivated peanut evolved, and they are still used as forages, for their esthetic value, and as sources of germplasm for crop improvement. For example, A. glabrata and A. pintoi are utilized for grazing (Mathews et al., 2000; Hernandez-Garay et al., 2004) and A. repens is used as a ground cover in residential areas and roadsides in tropical regions. Several species have been consumed for their seeds, but only A. hypogaea is economically important today as a human food source. The primary interest in wild species of Arachis for the past 50–60 years has been as sources of disease and insect resistances for crop improvement. Several species, including ones that will hybridize with the cultivated peanut, have extremely high levels of disease resistance and a few (e.g., A. diogoi) have virus resistance genes that are not present in the cultivated gene pool.
Cytology and Evolution of Arachis
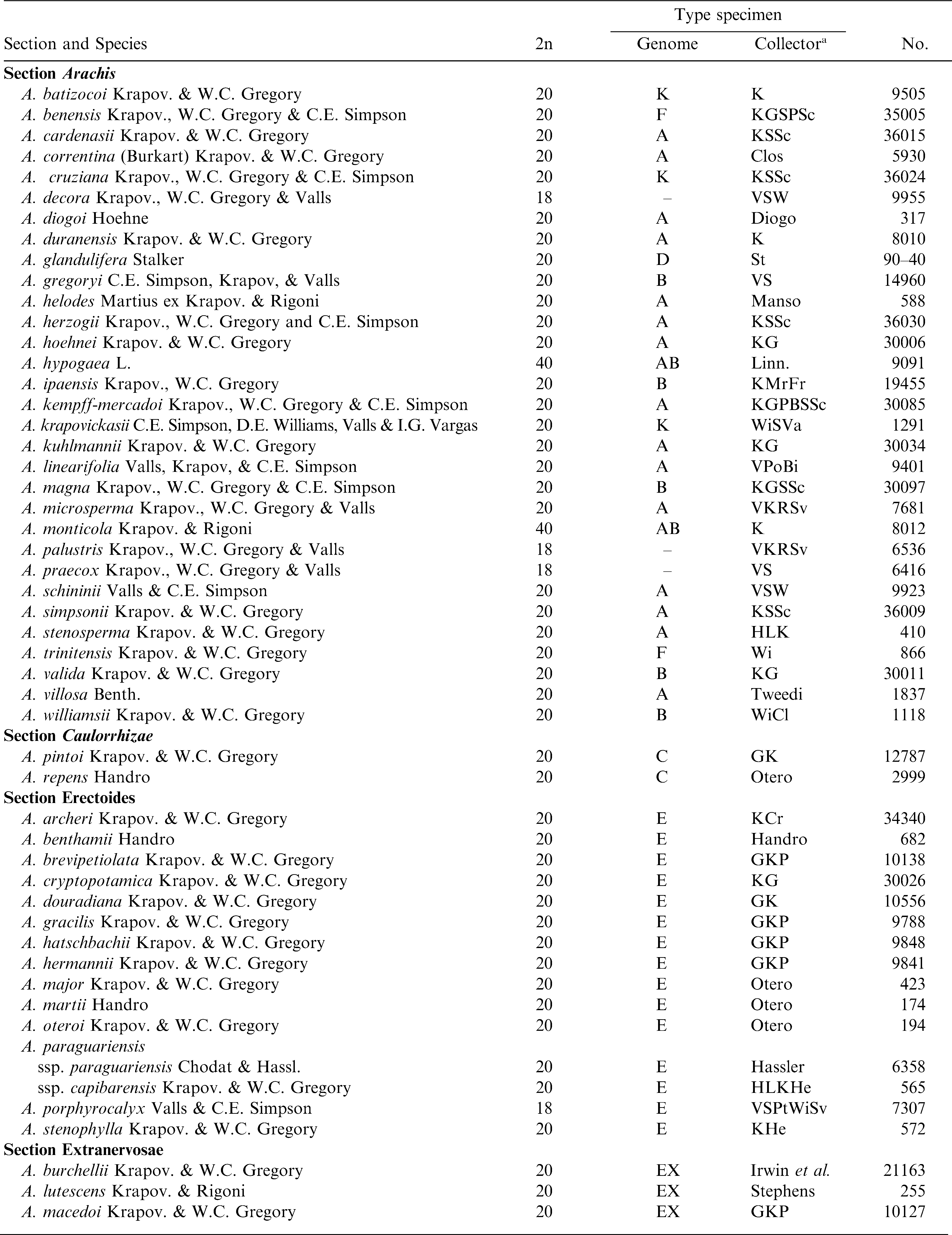
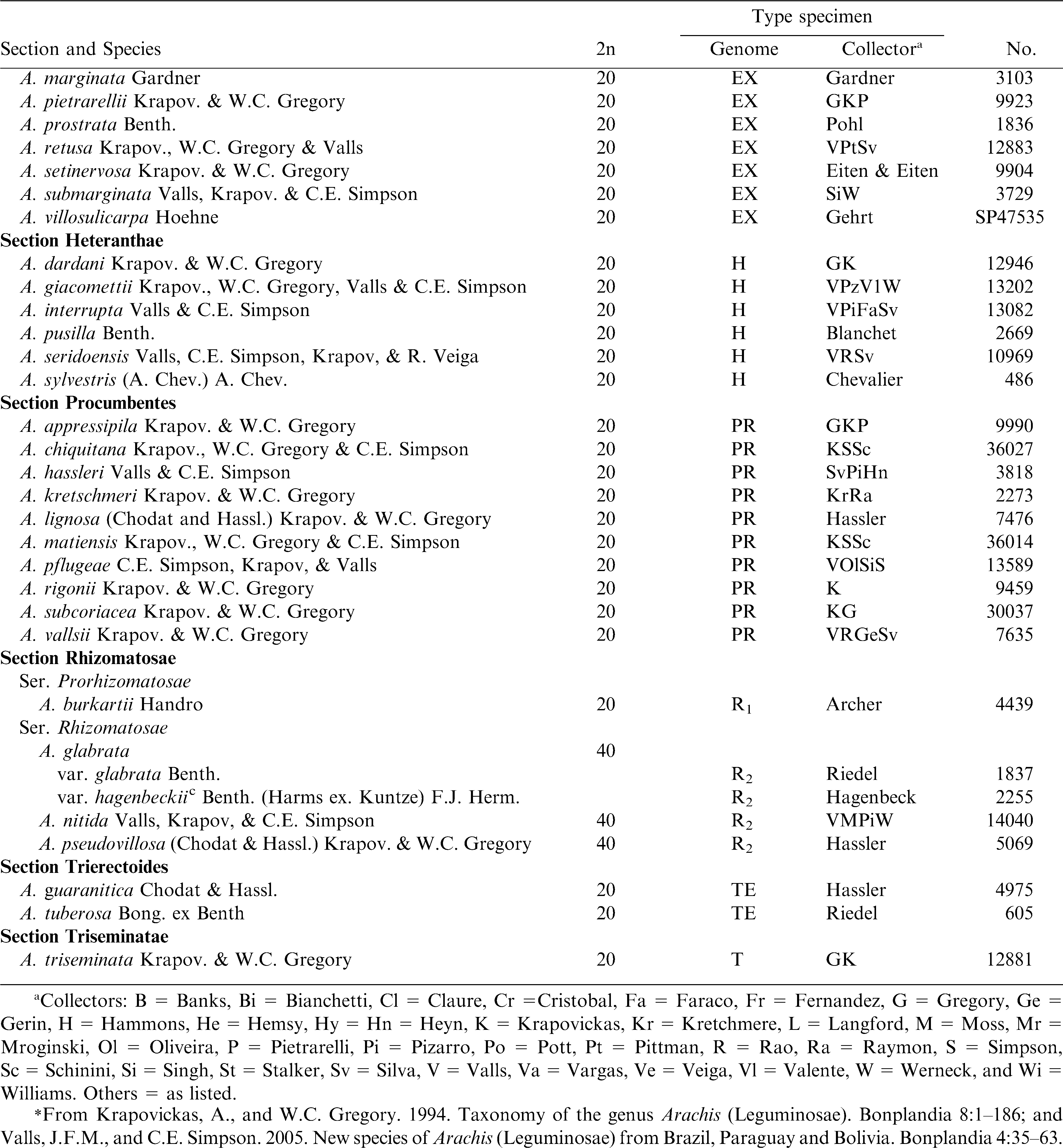
Arachis hypogaea is an allotetraploid (2n = 4x = 40) with a very large and complex genome. Chromosome behavior and morphology were reported by Husted (1936) and Stalker and Dalmacio (1986). Gregory (1946) reported the first chromosome number of a wild species (A. glabrata) as 2n = 4x = 40 and a year later Mendes (1947) reported diploid species (2n = 2x = 20). Not until 2005 were species with 18 chromosomes discovered (Penaloza and Valls, 2005). Most species in the genus are diploid, but tetraploids exist in sections Arachis and Rhizomatosae; and several species in sections Arachis and Erectoides are aneuploid (2n = 2x = 18) (Table 1). Polyploidy evolved independently in sections Arachis and Rhizomatosae (Smartt and Stalker, 1982); and Nelson et al. (2006) concluded that polyploidy evolved multiple times within section Rhizomatosae. Tallury et al. (2005) reported molecular evidence that the diploid section Rhizomatosae species (only one known) did not give rise to the tetraploids. Because A. glabrata will hybridize with species of both sections Erectoides and Arachis, Smartt and Stalker (1982) concluded that two diploids from sections Erectoides and Arachis likely hybridized and spontaneously doubled in chromosome number, evolving into the tetraploid species of section Rhizomatosae.
Krapovickas and Gregory (1994) concluded that Erectoides, Extranervosae, Heteranthae, Trierectoides and Triseminatae are “older” sections, while Arachis, Caulorrhizae, Procumbentes, and Rhizomatosae are more “recent” in origin. The largest subgeneric group is section Arachis, which includes the cultivated species, one other tetraploid (A. monticola), 26 diploid (2n = 2x = 20), and three aneuploid (2n = 2x = 18) species. These species are highly variable, and especially the annual species are continuing to differentiate. For example, A. duranensis has northern and southern groups which can be distinguished morphologically and with molecular markers (Stalker et al., 1995).
The first published attempt at interspecific hybridization in the genus was between the two tetraploids A. hypogaea (section Arachis) and A. glabrata (section Rhizomatosae) (Hull and Carver, 1938), but no hybrids were obtained. Krapovickas and Rigoni (1951) later hybridized A. hypogaea with A. villosa var. correntina and the F1s were vigorous, but sterile. The cultivated peanut has since been hybridized with most species in section Arachis. Similar to other genera which have a polyploid series, crosses are usually more successful when the species at the higher ploidy level (in this case A. hypogaea) is used as the female parent. Triploid interspecific hybrids usually have 10 bivalents and10 univalents, but trivalents are also observed in pollen mother cells which indicates that some chromosome homology exists between the A and B genomes (Stalker, 1985).
Early cytological research identified one pair of significantly smaller chromosomes (termed ‘A’ chromosome) in species of section Arachis and a unique chromosome pair that had a large secondary constriction (termed ‘B’ chromosome) in the species A. batizocoi (Husted, 1936). Hybridization between diploid species was first reported between A. duranensis and A. villosa var. correntina (Raman and Kesavan, 1962) and meiosis was regular. Later studies indicated that hybrids between species having the small chromosome pair were partially to fully fertile and most will produce F2 seeds; however, hybrids between the species with the small chromosome and A. batizocoi are sterile (Stalker and Simpson, 1995). Thus, the terminology ‘A’ and ‘B’ genome has been used in peanut to describe the two cytological groups. Because the cultivated peanut has one pair of smaller chromosomes and one pair of chromosomes with a large secondary constriction, it was described as an allotetraploid with AABB genomes. Stalker et al. (1991) crossed a series of species designated as having the A genome with A. batizocoi and found that F1s had many univalents, and bivalents were loosely associated. Hybrids between either A or B genome species with A. glandulifera (D genome) also have many univalents and are sterile (Stalker et al., 1991). Thus, there is a considerable amount of cytological differentiation between the three genomes.
Gregory and Gregory (1979) conducted an extensive hybridization program using 91 Arachis collections and reported cross-compatibility relationships among species of the genus. Their results indicated that hybridization between species within the same section is more successful than crosses between sections; F1s of intersectional crosses were highly sterile. To overcome crossing barriers, complex hybrids have been attempted (Gregory and Gregory, 1979; Stalker, 1981), but fertility has not been restored. Thus, introgression from wild Arachis species to A. hypogaea by conventional hybridization is believed to be restricted to members of section Arachis. Even within section Arachis there are many difficulties encountered to obtain hybrids due to genomic and/or ploidy differences.
Based on cross-compatibility data, Smartt and Stalker (1982) and Stalker (1991) concluded that genomic groups have evolved in the genus which mostly follow sectional designations (Am – Ambinervosae, T – Triseminatae, C – Caulorrhizae, EX – Extranervosae, and E – Erectoides, R – Rhizomatosae, and A, B and D – Arachis). The B genome was recently divided into B, F, and K genomes by Seijo et al. (2004) and Robledo and Seijo (2010). Based on rDNA loci and chromosomes with centromeric heterochromatin, Robledo et al. (2009) described three karyotypic subgroups within the A genome and grouped the cultivated peanut with A. duranensis, A. villosa, A. schininii, and A. correntina. Other studies support placing A. hypogaea closely with A. duranensis (Moretzsohn et al., 2004; Milla et al., 2005a; Bravo et al., 2006; Koppolu et al., 2010; Calbrix et al., 2012). The chromosomes of B genome species are karyologically more diverse than those with an A genome (Fernandez and Krapovickas, 1994; Seijo et al., 2004). The B genome species (as opposed to the F and K genomes) does not have centromeric heterochromatin and includes A. ipaensis (the B component of A. hypogaea), A. magna, A. gregoryi, A. valida, and A. williamsii (Seijo et al., 2004; Robledo and Seijo, 2010). The D genome species A. glandulifera is more distantly related to A. hypogaea than other species of section Arachis. Also, molecular analysis has indicated that the aneuploids in section Arachis are more closely related to the B (now classified as the B, F, and K genomes) and D genome species than to A-genome species (Tallury et al., 2005). Evolution is apparently continuing in section Arachis at a rapid pace and multiple translocations have been observed in diploid accessions of A. duranensis (Stalker et al., 1995) and A. batizocoi (Stalker et al., 1991; Guo et al., 2012). At least five different secondary constriction types have been observed in A. hypogaea, which were most likely from translocation events (Stalker and Dalmacio, 1986), and this species is also evolving cytologically.
Analyses of species outside section Arachis have been infrequent. Stalker (1985) reported that the two diploid section Erectoides species A. rigonii × A. paraguariensis hybrids had many univalents and Krapovickas and Gregory (1994) later placed these species in different sections. Intersectional hybrids also were reported by Mallikarjuna (2005) who used in vitro techniques to obtain F1s.
In addition to morphological and cross-compatibility studies, molecular investigations have been used to better clarify the phylogenetic relationships among peanut species. Most of these investigations have involved species in section Arachis because of their close association with A. hypogaea. Many molecular systems have been utilized, including isozymes (Lu and Pickersgill, 1993; Stalker et al., 1994), seed storage proteins (Singh et al., 1991; Bianchi-Hall et al., 1993; Liang et al., 2006), Restriction Fragment Length Polymorphisms (RFLPs) (Kochert et al., 1991; Paik-Ro et al., 1992), Amplified Fragment Length Polymorphisms (AFLPs) (Milla-Lewis et al., 2005b); Simple Sequence Repeats (SSRs) (Hopkins et al., 1999; He et al., 2005; Hong et al., 2010; Guo et al., 2012; Nagy et al., 2012). Randomly Amplified Polymorphic DNA (RAPDS) (Halward et al., 1992; Lanham et al., 1992; Hilu and Stalker, 1995), and in situ hybridization (Raina and Mukai, 1999; Seijo et al., 2004). All of the studies have indicated that the cultivated peanut has significantly less molecular variation than diploid species, which supports the hypothesis that A. hypogaea originated from a single hybridization event. Additionally, there has been little or no apparent introgression from diploid species to A. hypogaea since its inception (Kochert et al., 1996).
As opposed to the cultivated species, large amounts of molecular variation have been documented among wild species of the genus. Although there have been differences observed among marker systems regarding species relationships, and there remain questions about species positions in sectional groupings (Friend et al., 2010), the molecular data generally fits the sectional relationship model proposed by Krapovickas and Gregory (1994). For example, Hoshino et al. (2006) used microsatellites to evaluate species in the nine peanut sections, and while most species grouped as expected, several species in the Procumbentes grouped with species from section Erectoides, and others clustered into sections Trierectoides and Heteranthae. Galgaro et al. (1998) also indicated that species in section Heteranthae did not group together. Friend et al. (2010) conducted a more comprehensive investigation of Arachis species and found that sections Extranervosae, Triseminatae, and Caulorrhizae each separated into distinct groups based on trnT-trnF sequences; but species in sections Erectoides, Heteranthae, Procumbentes, Rhizomatosae, and Trierectoides formed a major lineage. Species in section Arachis grouped into two major clades, with (i) the B (renamed the B, F, and K genomes), the D genome species, and 18-chromsome aneuploids being in one group and (ii) the A genome species being in the second group.
Desirable Traits in Arachis Species for Crop Improvement
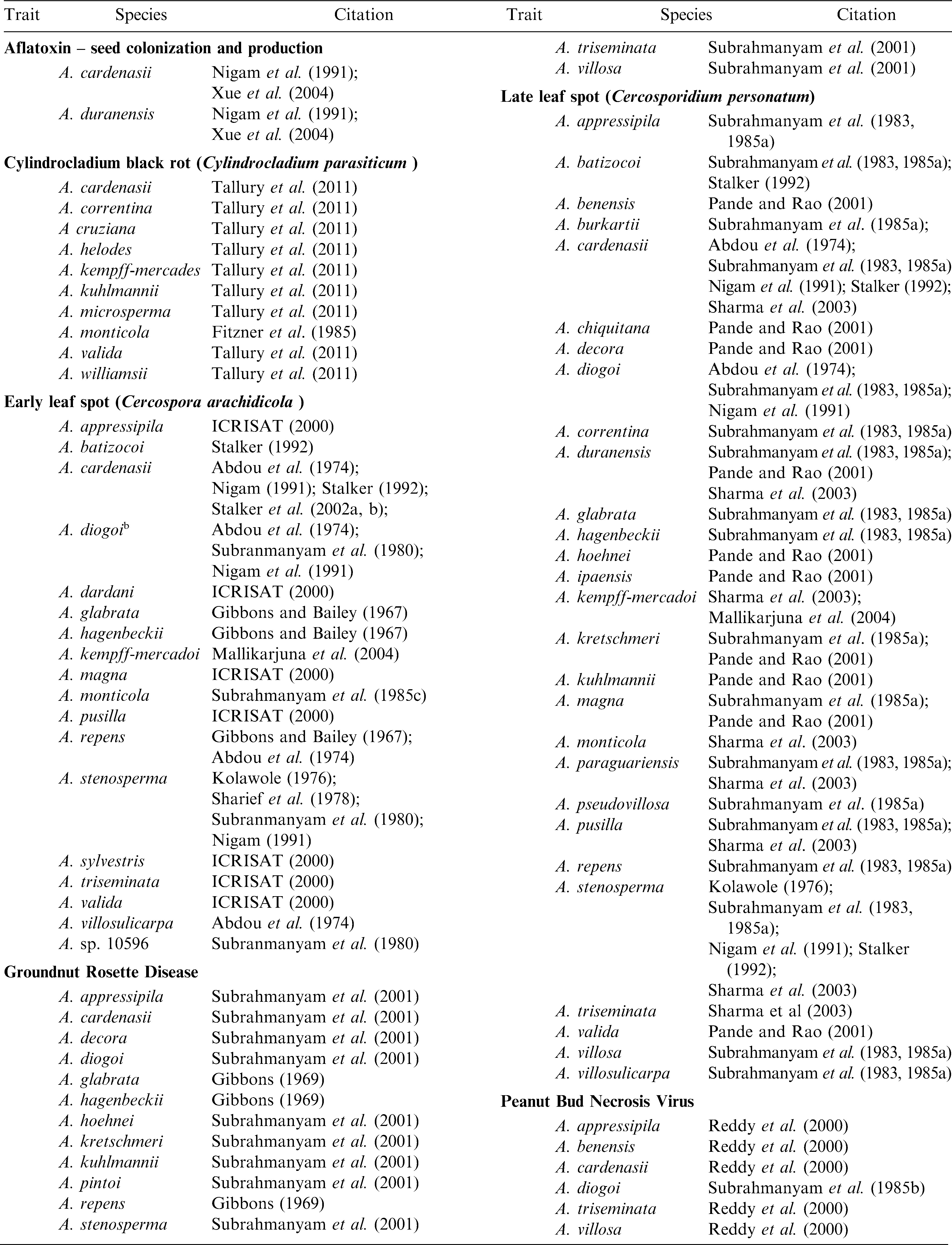
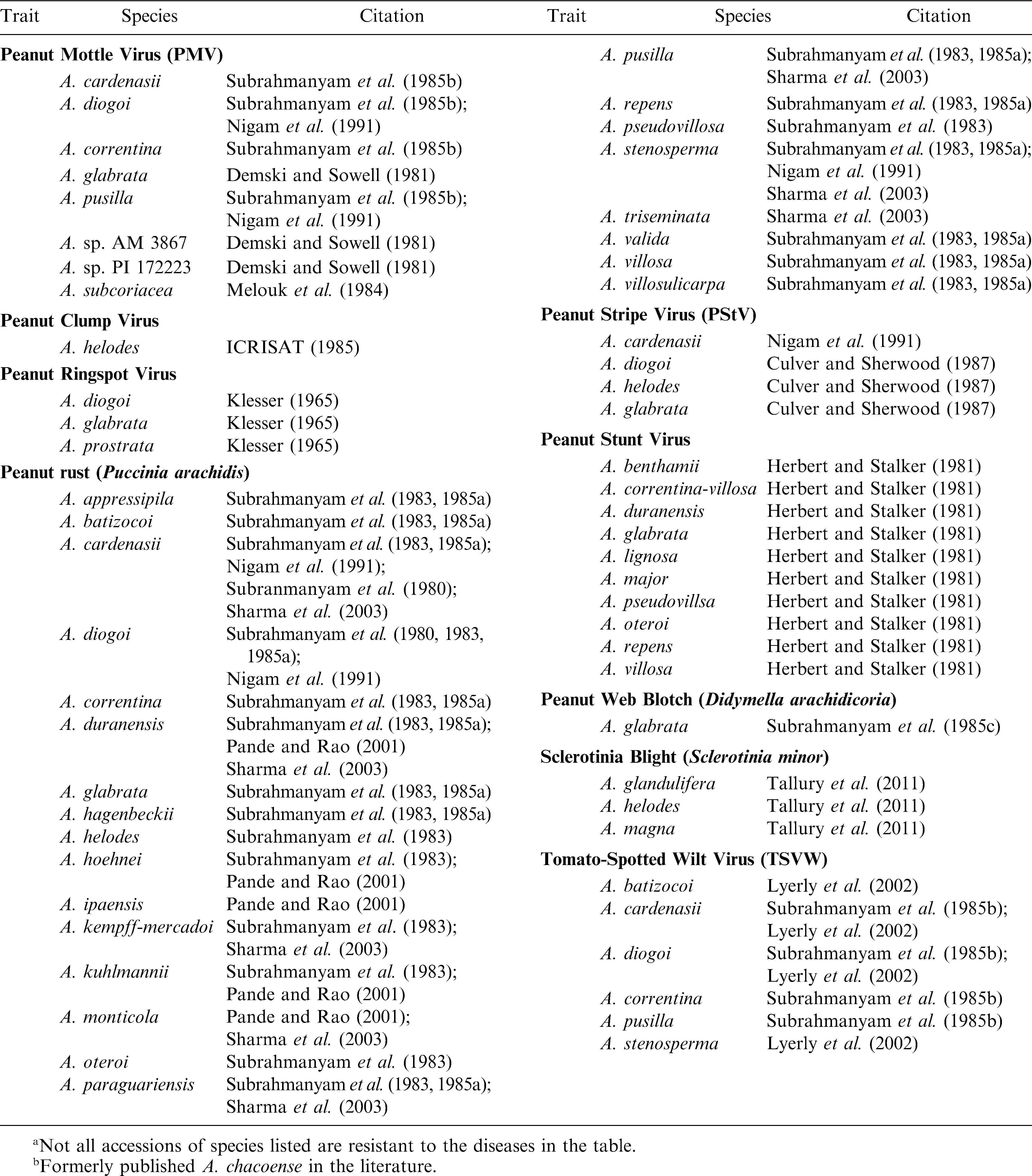
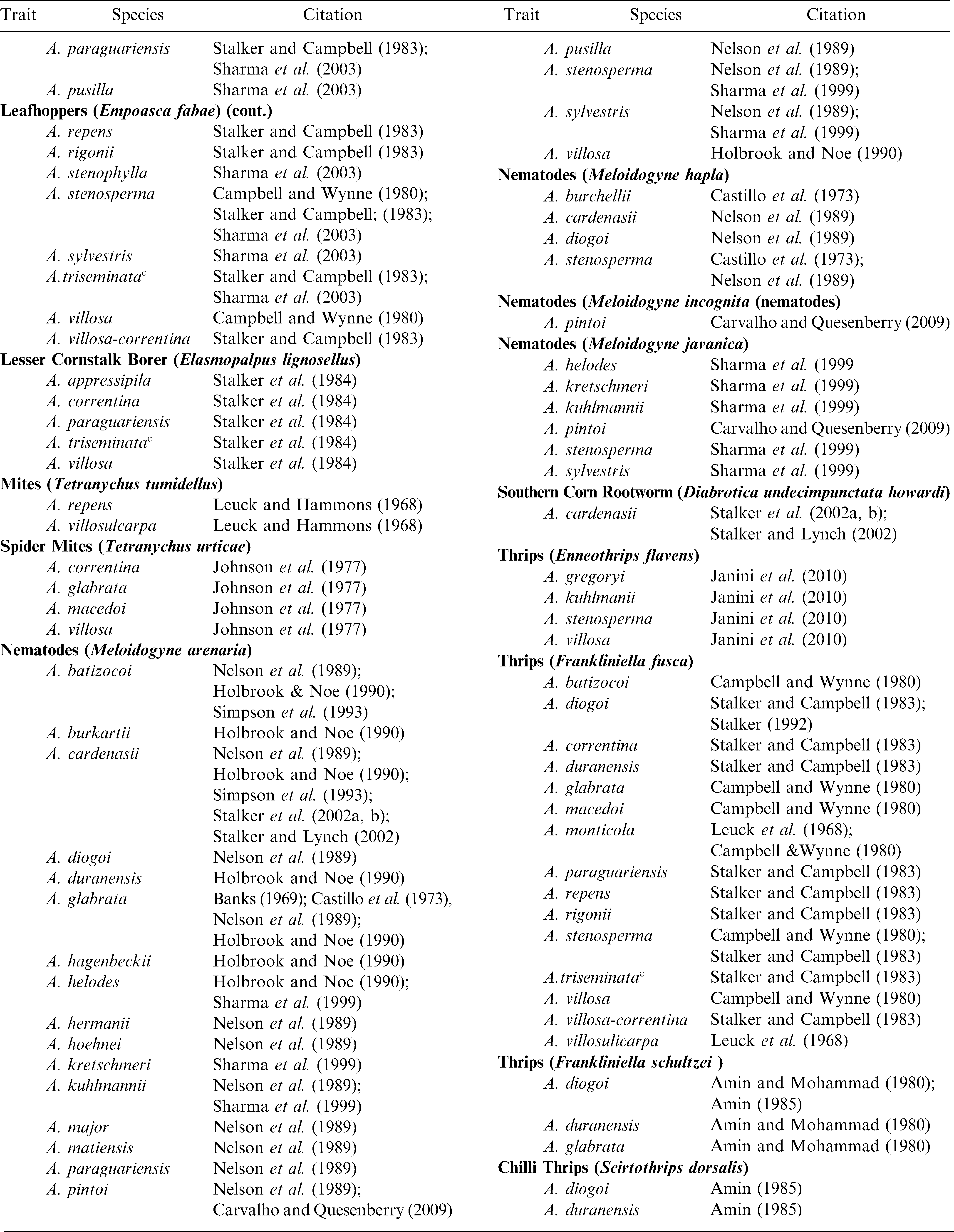
As compared to lines of A. hypogaea, extremely high levels of resistance have been identified in Arachis species for many important peanut pathogens (Stalker and Moss, 1987; Dwivedi et al., 2007) (Table 2). Mehan et al. (1986) identified four Arachis species that are resistant to aflatoxin production and Xue et al. (2004) found pre-harvest aflatoxin resistance in A. duranensis. Subrahmanyam et al. (2001) found 12 Arachis species accessions to be immune to groundnut rosette virus as opposed to none in the cultivated species. Arachis diogoi was the only species identified with no infection to peanut bud necrosis virus (Subrahmanyam et al., 1985b); this species is also the only one with immunity to tomato spotted wilt virus (Lyerly et al., 2002). None of 7,000 cultivated lines screened for peanut clump virus (PCV) had useful resistance whereas four Arachis accessions of A. kuhlmannii, A. duranensis, and A. ipaensis were immune (Dwivedi et al., 2007). ICRISAT scientists also have evaluated Arachis species for late and early leaf spots and they identified highly resistant materials (Dwivedi et al., 2007). Many Arachis species also have been evaluated for insect pests and extremely high levels of resistance observed as compared to the cultivated peanut (Table 3).
Introgressing Genes from Arachis Species to A. hypogaea
Because the domesticated peanut is an allotetraploid with two genomes and the species being utilized for introgression are diploids, sterility barriers result from ploidy differences and genomic incompatibilities between the species. Traits of interest from Arachis species have been difficult to follow in progenies of interspecific hybrids and disease and insect resistances in 40-chromosome progenies have been lost because of low population sizes and inadequate methods for selection in single plants. Utilizing molecular markers associated with traits of interest may help overcome many of these problems, but unfortunately, few molecular markers have been available to enhance selection efficiency. Molecular research to date indicates that introgression from Arachis species to A. hypogaea appears to be in large blocks (Garcia et al., 1995; Nagy et al., 2010) rather than as single genes or small chromosome segments. Thus, linkage drag of undesirable traits can restrict the use of genetic resources, and molecular markers also have great potential for selection against these characters.
The first peanut cultivars released from interspecific hybridization were from a cross between A. hypogaea and the second tetraploid species in section Arachis (A. monticola Krapov. & Rigoni). Biologically, A. monticola could be considered a weedy subspecies of A. hypogaea. Spancross was released by Hammons (1970) and Tamnut 74 was later released by Simpson and Smith (1975). Neither of these cultivars had phenotypic traits that could be identified as being derived from the wild species, which is not surprising because A. monticola has most of the same disease and insect problems as found in A. hypogaea.
Several methods have been utilized to create populations of fertile A. hypogaea interspecific hybrids and to restore plants to the tetraploid level. First, crosses can be made by hybridizing A. hypogaea with diploids to produce triploid (3x = 30) F1s, after which cuttings can be colchicine-treated to restore fertility at the hexaploid (6x = 60) level. Many triploids will also produce a few seeds through fusion of unreduced gametes, especially if they are left in the field or grown in the greenhouse for long periods of time (Singsit and Ozias-Akins, 1992). Backcrossing the hexaploids with A. hypogaea results in pentaploids (5x = 50) that are usually vigorous, but only partially fertile. Additionally, they produce few flowers and are difficult to use in crossing programs; but they sometimes yield a few seeds when selfed and the ploidy level stabilizes in progenies at the tetraploid level. A major problem with this scheme has been the few seeds produced at the hexaploid and pentaploid levels, and the lack of selection methods for traits of interest during the semi-sterile generations has resulted in many hundreds of tetraploid lines without traits of interest for crop improvement. To date, no useful germplasm has resulted from backcrossing hexaploids with A. hypogaea. Although backcrossing hexaploids with diploids will theoretically drop the chromosome number to the tetraploid level in one generation, these 6x × 2x crosses (or reciprocals) have not produced viable progenies.
An alternative method to backcrossing hexaploids with the cultivated species is to allow 6x plants to self-pollinate and, by selecting fertile progenies, a few plants may spontaneously lose chromosomes and stabilize at the 40-chromosome level. The loss of chromosomes appears to be infrequent and random, but the advantage of this procedure is associating chromosomes in different species at a high ploidy level which can increase the frequency of recombination. For example, A. hypogaea × A. cardenasii hexaploids were selfed for five generations after which they produced 40-chromosome progenies that were highly variable for seed size, color and other morphological traits (Company et al., 1982). Garcia et al. (1995) analyzed introgression from A. cardenasii to A. hypogaea with RFLPs and found wild species-specific markers on 10 of 11 linkage groups on the diploid RFLP map developed by Halward et al. (1993). Most of the introgression (88%) was apparently in the A genome of A. hypogaea, with the remaining 12% in the B genome. Germplasm lines have been released from this cross with resistance to early leaf spot, nematodes, and several insect pests (Stalker et al., 2002a, b; Stalker and Lynch, 2002; Isleib et al., 2006). The cultivar Bailey was released after utilizing these lines as sources of multiple disease resistances (Isleib et al., 2010).
A second method to introgress germplasm from diploid species to A. hypogaea is to first double the chromosome number of the diploid species to the tetraploid level. This method has the advantage of avoiding several generations of mostly sterile hybrids and recovering tetraploids and is much faster than by going through the triploid – hexaploid procedure; but autotetraploids generally have low vigor, and when annual species are used as parents, they are short lived. Ideally, A and B genome species would be hybridized at the diploid level and then the chromosomes doubled to produce AABB genome allopolyploids to be crossed with the cultivated species. However, chromosome doubling of the sterile AB genome diploids can be highly problematic. Examples of success with this methodology are TXAg-6 and TX Ag-7 (Simpson et al., 1993) which originated from the complex hybrid [A. batizocoi (B genome) × (A. cardenasii (A genome) × A. diogoi (A genome))]4x . TxAG-6 had very good nematode resistance, but also significant linkage drag which resulted in low yields and poor seed and pod quality. RFLP markers linked to the nematode gene conferring resistance were used to select favorable genotypes (Church et al., 2000). The nematode-resistant cultivars COAN (Simpson and Starr, 2001) and NemaTAM (Simpson et al., 2003) were released after introgressing genes from TXAg-6 to A. hypogaea. By using SSR markers, Nagy et al. (2010) showed that recombination was greatly reduced in the chromosome region where the nematode-resistance gene is located because a large introgressed segment from the wild species comprised a third to half of a chromosome. This germplasm was used in the development of the nematode-resistant cultivar Tifguard (Holbrook et al., 2008), which also has good resistance to tomato spotted wilt virus.
Tracking introgression from Arachis species to cultivated peanut would greatly facilitate selection of desirable progenies in advanced generations of interspecific hybrids. Molecular markers offer a method for following introgression from Arachis species to A. hypogaea. This is important because recombination between the cultivated genomes and those of other species is rare, thus restricting selection for desired traits in interspecific hybrid derivatives (Holbrook and Stalker, 2003). Guimarães et al. (2010, 2011) identified eight genes in A. stenosperma roots that correspond with resistance to M. arenaria, and QTLs for resistance to late leaf spot also have been identified (Leal-Bertioli et al., 2009) in section Arachis species.
For tracking introgressed segments from wild accessions into cultivated germplasm, markers that allow more agile assays would be ideal. A different type of marker, transposon-based, is being evaluated for their use for introgressed segments. For example, Shirasawa et al. (2012b) developed 535 markers derived from transposon-enriched genomic libraries. These markers show great potential and detect higher polymorphism levels than genomic microsatellite markers (Shirasawa et al., 2012b). Also, Single Nucleotide Polymorphism (SNP) markers constitute the most abundant molecular markers in the genome and can be carried out with high throughput genotyping methods. SNP markers have been widely used in many plant species. However, they have had limited use in peanut due to the difficulties of their implementation in polyploid plants, and in Arachis it will require separation of A and B-genome sequences. A SNP-based map of diploid Arachis was developed by Nagy et al. (2012) wherein a high-density genetic map of the A genome was developed from an intra-species cross within A. duranensis, and 598 SSRs, 37 single-stranded DNA conformation polymorphism (SSCP) markers, and 1,054 SNPs were mapped. SNP-based markers have not yet been extensively used on the tetraploid Arachis, but they are expected to greatly accelerate genetic mapping and marker-assisted selection when genotyping-by-sequencing (Elshire et al., 2011) can be routinely implemented.
Linking Agronomic Traits with Markers
The narrow genetic base of peanut makes the development of molecular markers a difficult task. The nature and the possibility of using molecular markers have been evolving as have all aspects of the agronomical sciences. The first molecular marker studies used for peanut were based on isozymes and proteins (Grieshammer and Wynne, 1990; Krishna and Mitra, 1988; Lu and Pickersgill, 1993), followed by Restriction Fragment Length Polymorphism - RFLPs (Kochert et al., 1991; Kochert et al., 1996; Paik-Ro et al., 1992), Random Amplified Polymorphic DNA - RAPDs (Halward et al., 1991; Halward et al., 1992; Hilu and Stalker, 1995; Subramanian et al., 2000; Dwivedi et al., 2001; Raina et al., 2001) and Amplified Fragment Length Polymorphism – AFLPs (He and Prakash, 1997; Gimenes et al., 2000; Gimenes et al., 2002; Herselman, 2003; Milla et al., 2005a, 2005b; Tallury et al., 2005).
In recent years, microsatellite or Simple Sequence Repeat (SSR) markers have become the assay of choice for genetic studies in Arachis since they are multiallelic, co-dominant, transferable among related species, PCR-based markers, and usable in tetraploid genomes. Efforts by several research groups to develop microsatellite markers for peanut have resulted in more than 5,000 SSRs (see Pandey et al., 2011, 2012 for reviews). This large effort to produce and characterize SSRs has enabled the phylogenetic evaluation of the genus Arachis (Krishna et al., 2004; Barkley et al., 2007; Tang et al., 2007; Varshney et al., 2009b, Moretzsohn et al., 2013) and production of moderately dense genetic maps for cultivated peanut, which will be described below.
To date, the number of genes associated with molecular markers in peanut is relatively small, but the large number of molecular markers becoming available has great potential for utilization in crop improvement programs. Bertioli et al. (2003) described numerous linkages of resistance genes in peanut. Pandey et al. (2012) listed QTLs for some of the important traits found in the cultivated peanut. Chu et al. (2011) outlined a breeding scheme to utilize marker-assisted selection to pyramid nematode resistance and the high oleic acid trait in peanut cultivars, and the system has greatly increased efficiency for developing breeding lines.
Introgression of QTLs with the Aid of Molecular Markers
By the use of molecular markers to map genetically structured populations, a number of Quantitative Trait Loci (QTLs) have been identified and some are already used for tracking wild segments introgressed into A. hypogaea.
Root-knot Nematode (Meloidogyne spp.)
The first markers for an agronomically useful, wild species-derived trait in peanut were for resistance to root-knot nematode (M. arenaria) from A. cardenasii. Two closely-linked sequence characterized amplified region (SCAR) markers were identified for genes for reduced galling and egg number (Garcia et al., 1996). Simultaneously, three RAPD markers were associated with nematode resistance in several backcross breeding populations derived from the interspecific hybrid TxAG-6 [A. batizocoi × (A. cardenasii × A. diogoi)]4x (Burow et al., 1996). Marker-assisted selection then was used to develop NemaTAM, a high yielding, nematode resistant cultivar (Simpson et al., 2003). In this case, it was demonstrated that use of markers was more efficient than phenotypic selection because plants selected with markers for the homozygous resistance gene have fewer escapes compared to plants from phenotypic selection. Marker-assisted selection and an accelerated backcross breeding program were also used in development of a high-oleic variety with nematode and tomato spotted wilt virus resistances, called high O/L Tifguard (Chu et al. 2011). The effectiveness of selection of nematode resistance has been the most successful use of MAS in peanut to date. However, in spite of the success of this work, it is now thought that the use of a single gene trait that confers near immunity may be subject to breakdown of resistance under high selection pressure, and is cause for concern. Therefore, new sources of resistance for nematodes, such as amphidiploids derived from A. stenosperma, which is highly resistant to fungi and nematodes (Leal-Bertioli et al., 2010; Proite et al., 2008; Santos et al., 2011; Singsit et al., 1995), would be useful for peanut breeding.
Late Leaf Spot Resistance
Resistance to late leaf spot (LLS) (Cercosporidium personatum) has multiple components, including percent defoliation, incubation period, latency period, lesion number and diameter, sporulation, and pod yield (Green and Wynne, 1986; Chiteka et al., 1988a, 1988b; Anderson et al., 1993; Waliyar et al., 1993, 1995). High levels of resistance have also been associated with low yield suggesting linkage or pleotropic effects (Iroume and Knauft, 1987), thus breeding for high yielding cultivars with resistance requires this linkage to be broken.
Stalker and Mozingo (2001) identified three RAPD markers associated with early leaf spot lesion diameter in a peanut population derived from a cross between an A. hypogaea × A. cardenasii introgression line with NC 7. Two breeding lines developed from this material have been placed into advanced line trials.
Mapping of RFLP markers on BC3F1 lines in greenhouse studies identified five markers for leaf spot resistance (Burow et al., 2008), including three QTLs for incubation period, and one each for latency period, lesion number and diameter. Those QTLs for latency period and lesion number were overlapping, suggesting either linkage between the two or a QTL with pleiotropic effects.
Leal-Bertioli et al. (2009) reported the mapping of 34 resistance gene analogues (RGAs) and five QTLs for LLS resistance (% diseased leaf area) on detached leaves of the F2 plants of the A-genome mapping population derived from A. duranensis × A. stenosperma, and suggested additive or partial dominance gene action. One QTL explained almost half of the phenotypic variance observed and some QTLs mapped near RGA markers. In another QTL study based on cultivated genotypes, with GPBD-4 as one parent, Khedikar et al. (2010) reported 11 QTLs for LLS; each QTL explained 2 to 7% of phenotypic variation in three environments, suggesting that the genes controlling LLS resistance in this cross are relatively minor. In maps from two other populations, again using GPBD-4 and a larger number (188 and 181) of markers and six trials, a major QTL for LLS was reported, which explained from 10% to 62% of phenotypic variance, depending on the environment (Sujay et al., 2011). These are being used for marker assisted selection breeding at ICRISAT in India (Varshney, 2012, personal communication).
Rust
QTL analysis using a partial genetic map of a mapping population with 67 marker loci derived from the cross TAG 24 × GPBD-4 and multiple season phenotyping data on both rust and LLS detected 12 QTLs explaining between 1.70 and 55.20% of the phenotypic variation for each disease, respectively (Khedikar et al., 2010). The SSR marker tightly linked to the major QTL (IPAHM103; QTLrust01), was then validated among a diverse set of genotypes as well as another mapping population (Sarvamangala et al., 2011) derived from the cross TG 26 × GPBD-4. The SSR marker (IPAHM 103) was deployed to introgress the rust resistance QTL into three elite groundnut varieties (ICGV 91114, JL 24 and TAG 24) using the donor GPBD-4 through marker-assisted backcrossing. GPBD-4 was a common parent in all of these crosses, and since it was derived from the wild species A. cardenasii, resistance incorporated into cultivars was likely derived from wild species germplasm.
Groundnut Rosette Virus
The aphid-transmitted groundnut rosette virus is an important pathogen of peanut in Africa and Asia, causing severe stunting and loss of yield. Herselman et al. (2004) tested 308 AFLP primer combinations, and they were able to devise five linkage groups consisting of 12 markers with one marker linked to aphid resistance.
Tomato Spotted Wilt Virus (TSWV)
Tomato spotted wilt virus is transmitted by tobacco thrips (Frankliniella spp.) and causes serious losses in the U.S.A. segregating population of F2 plants of the A-genome diploid cross A. kuhlmannii × A. diogoi was screened for resistance to TSWV, and five linked AFLP markers on one chromosome were associated with resistance at a high statistical threshold (Milla et al., 2004; Milla, 2003). In Brazil, interspecific populations and wild species have also been found as promising for introgression of resistance to the thrips, Enne-othrips flavens (Janini et al., 2010).
Domestication Traits
Mapping of RFLP markers on BC3F2 lines of the cross A. hypogaea × A. cardenasii (Burow et al., 2011) identified 29 markers for the domestication-related traits of main stem length, number of lateral branches, pod size, and seed size. Foncéka et al. (2012) produced populations derived from crosses of cultivar Fleur 11 and an amphidiploid (A. ipaensis × A. duranensis)4x (Fávero et al., 2006) to investigate introgression of wild segments. A population composed of a mixture of BC3F1 and BC2F2 individuals, each self-pollinated to produce BC3F2 and BC2F3 families, was used for phenotyping and QTL detection. Domestication-related trait QTLs were found, including ones associated with days to flowering, plant architecture, pod and seed morphology, and yield components. Perhaps surprisingly, it was shown that wild alleles contributed positive variation to several agronomic traits such as flowering precocity, seed and pod number per plant, and length, size and maturity of pods. Moreover, the comparison of QTLs obtained under well-watered and water limited conditions revealed that QTLs for stress tolerance indices for pod and seed numbers with favorable alleles could be attributed to the wild parents. These could be involved in reproductive trade-offs between producing large seeds versus producing more, but smaller, seeds under water stress.
Genetic Maps
One of the main uses of molecular markers in peanut has been the construction of genetic linkage maps, which are used to study the genome structure and organization, identify regions of particular interest (e.g., resistance to diseases) and for marker-assisted selection in breeding programs. Due to the very low genetic variation in cultivated peanut, wild species were initially used for the construction of linkage maps in Arachis because of their simple genetic structure and higher polymorphism levels. The first map used RFLPs to analyze variation between the diploid species A. stenosperma and A. cardenasii where a total of 117 RFLP markers were mapped into 11 linkage groups (Halward et al., 1993). An AFLP-based A-genome map was generated from an F2 population developed from the cross A. kuhlmannii × A. diogoi (Milla, 2003), and a RAPD-based map of A. stenosperma × A. cardenasii also was developed by Garcia et al. (2005).
It was only with the large abundance of SSR markers that moderately saturated maps were created. The first SSR-based map was constructed for an F2 population derived from a cross of two A-genome diploid species (A. duranensis and A. stenosperma) where 170 loci mapped into 11 linkage groups covering 1231 cM of total map distance (Moretzsohn et al., 2005). Additional markers were subsequently included in the diploid A genome map, resulting in 369 markers (188 SSRs, 80 anchor markers, and 35 resistance gene analogues, among others), mapped into 10 linkage groups, which correspond to the 10 chromosomes of the haploid genome (Leal-Bertioli et al., 2009). Nagy et al. (2012) published a more saturated map having 1724 SNP, SSR, and single stranded DNA conformation polymorphism (SSCP) makers from a cross between two A. duranensis accessions.
A diploid F2 population derived from the cross A. ipaënsis × A. magna was used to develop a map for the B genome of Arachis, with 149 codominant markers (mostly microsatellites) mapped into 10 linkage groups covering of 1294.4 cM (Moretzsohn et al., 2009). Fifty-one common markers presented evidence of the high synteny of the B and the A genomes. Guo et al. (2012) created a more dense map by crossing two accessions of A. batizocoi. They compared high density A and B genome maps and observed a large amount of synteny between the A and B genomes, but also several inversions and translocations.
The first tetraploid map also was created with RFLPs by using progenies of a cross between the cultivar Florunner × the synthetic amphidiploid TxAG-6 [A. batizocoi × (A. cardenasii × A. diogoi)]4x where 383 markers were mapped (Burow et al., 2001). A genetic map was created based on a population of 88 BC1F1 individuals of a cross of a synthetic amphidiploid (A. ipaënsis × A. duranensis) by Fávero et al. (2006) with A. hypogaea cv. Fleur11. This SSR-based linkage map for the tetraploid genome was composed of 298 markers and 21 linkage groups, spanning 1843.7 cM (Fonceka et al., 2009). A comparative analysis of this map with the A genome map of Moretzsohn et al. (2005) suggested the occurrence of a chromosomal translocation event prior to the peanut's tetraploidization (Fonceka et al., 2009).
The partial first linkage map from a cross between accessions of A. hypogaea was constructed using an F2 population (Herselman et al., 2004), in which five linkage groups with 12 markers spanned 139 cM of the genome. More complete maps were later reported, with the first being in 2008 by using 142 individuals of a recombinant inbred line (RIL) population derived from a cross between one accession of A. hypogaea subsp. hypogaea and one accession of the fastigiata subspecies (Hong et al., 2008). New markers were added to this map and two additional maps were constructed based on RIL populations having accessions of the subspecies fastigiata as parents (Hong et al., 2010). A reference map was developed, with 175 loci and 22 linkage groups, covering a total distance of 885.4 cM. The marker order was in general collinear to the A genome map (Moretzsohn et al., 2005). Another intraspecific map for peanut was developed using a RIL population composed of 318 F8/F9 plants (Varshney et al., 2009a); and of the 1145 microsatellite markers screened, only 135 mapped in 22 linkage groups spanning 1270.5 cM. More recently, Wang et al. (2012) published a SSR map based on an F2 population of 94 individuals derived from an A. hypogaea subsp. hypogaea × A. hypogaea subsp. fastigiata hybrid. This map consisted of 385 polymorphic SSRs covering 318 loci and 21 linkage groups that spanned 1674.4 cM. To date, there are 23 maps of peanut published using RFLPs, AFLPs, SSRs, SNP, SCAR and CAPS markers which are summarized in Pandey et al. (2012). A recent consensus map of A. hypogaea map comprised of 897 marker loci was constructed by Gautami et al. (2012).
These maps are very useful for breeding because they incorporate QTLs for agronomically important traits, such as disease resistance and drought related traits. They also have been used to develop markers closely associated with a nematode resistance gene (Nagy et al., 2010). Further, as the cultivated genome is sequenced, the diploid maps will be of great importance for sorting out the chromosome and genetic organization of the tetraploid genome.
Map Synteny
In general, it is possible to map only limited numbers of molecular markers in a given mapping population due to polymorphism constraints, and this is especially true for peanuts. In order to improve the knowledge on the genetic and genomic structure, assignment of marker and QTL positions, genetic maps developed for different mapping populations can be used for creating consensus maps, which can be also be anchored to other species maps. Consensus maps allow investigators to (i) map a large number of marker loci onto a single map, (ii) determine relative position of common markers across different mapping populations, (iii) determine stability of marker locus position across the genomes, and (iv) provide evidence for chromosomal rearrangements, gene duplication and assists in the assignment of linkage groups to chromosomes (Gautami et al., 2012).
With the present availability of markers, the international Arachis community has been striving towards developing a consensus genetic map. By integration of the eleven linkage maps of the artificial amphidiploids and cultivated tetraploid peanuts, a reference consensus map consisting of 897 marker loci has been constructed and given the consensus nomenclatures for the linkage groups (a01 to a10 and b01 to b10) (Gautami et al., 2012).
Subsequently, Shirasawa et al. (2012a) created the most comprehensive consensus map to date. They mapped a large number of DNA markers from two cultivated peanut maps (Shirasawa et al., 2012b) onto the previously reported linkage maps of the A and B genomes (Moretzsohn et al., 2005) and the artificial amphidiploid map (Fonceka et al., 2009), and integrated these with nine maps of cultivated peanut (Gautami et al., 2012; Hong et al., 2010; Khedikar et al., 2010; Sarvamangala et al., 2011; Qin et al., 2012; Ravi et al., 2011; Sujay et al., 2011; Varshney et al., 2009b; Shirasawa, 2012a, 2012b). This consensus map was then subjected to comparative analysis with four legume genomes (Cajanus cajan, Glycine max, Lotus japonicus, and Medicago truncatula) to clarify the feature of genome structure in the genus Arachis. The comparison of diploid and tetraploid maps revealed probable genome rearrangements within the Arachis genomes. Bertioli et al. (2009) used sequence characterized markers, and a high proportion of low or single copy gene markers to anchor the AA-diploid Arachis map onto the fully sequenced genomes of L. japonicus and M. truncatula (Sato et al., 2008); www.medicago.org). This alignment was represented as “genome plots” (Bertioli et al., 2009). Inspection of these plots shows surprising degrees of synteny considering the time of species divergence (estimated 55 million years). Although there are some regions of double affinities between Arachis and these model legumes, most synteny blocks have a single main affinity and not multiple affinities. Genome evolution (e.g., chromosomal translocations and inversions) progressively breaks down syntenic relationships between species over evolutionary time.
The comparative genomics of the legumes revealed that the Arachis genome structure was moderately divergent from those of the compared legumes. This information will be useful for selecting highly informative and uniformly distributed markers for developing new genetic maps, background selection and diversity analysis, aligning new genetic and physical maps, performing QTL analysis in a multi-populations design, evaluating the genetic background effect on QTL expression, and serving other genetic and molecular breeding activities in peanut.
Value of Diploids for Genomic Investigations
The chromosomes of A. hypogaea are highly diplodized which indicates that there may be little recombination between the A and B genomes except when the infrequent quadrivalent is formed. To support this, genomic comparison of A. duranensis and A. ipaensis indicates that there is significant divergence in repetitive DNA; however, the repetitve DNA in the tetraploid species has not significantly recombined since A. hypogaea evolved (Araujo et al., 2012). Thus, genomic investigations of the progenitor diploid species should have direct application to the cultivated peanut. For example, sequencing the diploid genome of either A. duranensis or A. ipaensis could serve as a template for sequencing the genomes of the tetraploid species. This is important because developing a high quality diploid genetic sequence will be significantly less expensive than developing an entire sequence for the tetraploid genomes and trying to sort out the genetic duplications between the A and B genomes. Using a diploid progenitor species also will lead to fewer assembly errors. In addition, utilizing A. duranensis or A. ipaensis for sequencing templates will solve problems associated with chimeric contigs in de-novo assemblies of the tetraploid genomes.
Conclusions and Future Prospects
The incorporation of wild alleles into crops is a proven strategy for the development of improved varieties with pest and disease resistance, tolerance to abiotic stress, improved yield and quality, and even male fertility and fertility restoration. However, the extent of utilization of the useful allele reservoir in wild species and its impact on peanut breeding has been limited, mainly due to the restrictions of the plant itself in terms of crossability, multiplication rate, and, until recently, to the lack of appropriate molecular tools to analyze and follow traits in hybrids. Arachis cardenasii has been one of the most useful sources of genes from wild species to date, but crosses involving other species also have been used. The recent use of the two most probable ancestors of peanut A. duranensis and A. ipaënsis in a systematic introgression program opens the way for extensive and detailed characterization of the peanut genome and wild allele interactions for a wide range of traits. As new materials are being created and genotyping strategies are becoming more advanced, variability from the wild species is being harnessed to the benefit of world agriculture.
Literature Cited
Abdou Y.A.M Gregory W.C and Cooper W.E 1974 . Sources and nature of resistance to Cercospora arachidicola Hori and Cercosporidium personatum (Berk et Cutis) Deighton in Arachis species . Peanut Sci. 1 : 6 – 11 .
Amin P.W 1985 . Resistance of wild species of groundnut to insect and mite pests, pp. 57 – 60 . In: Proceedings of an International Workshop on Cytogenetics of Arachis, 31 Oct-2 Nov 1983 . ICRISAT Center , Patancheru, A.P., India .
Amin P.W and Mohammad A.B 1980 . Groundnut pest research at ICRISAT, pp. 158 – 166 . In: Proceedings of an international workshop on groundnuts, 13–17 Oct 1980 . ICRISAT Center , Patancheru, A.P., India .
Amin P.W and Mohammad A.B 1982 . ICRISAT's Research in groundnut entomology. Occasional Paper Groundnut Entomology. 82/1, 34 pp, ICRISAT , Patancheru, A.P. 502 324, India .
Anderson W.F Holbrook C.C and Brenneman T.B 1993 . Resistance to Cercosporidium personatum within peanut germplasm . Peanut Sci. 20 : 53 – 57 .
Araujo A Nielen S Vidigal B Moretzsohn M Leal-Bertioli S Ratnaparkhe M Kim C Bailey J Paterson A Guimaraes P Schwarzacher T Heslop-Harrision P and Bertioli D 2012 . An analysis of the repetitive component of the peanut genome in the evolutionary context of the Arachis A-B genome divergence . XX Plant and Animal Genome Confr , San Diego, CA , 2012.
Banks D.J 1969 . Breeding for northern root-knot nematode, Meloidogyne hapla, resistance in peanuts . Proc. Amer. Peanut Res. Educ. Assoc. 1 : 23 – 28 .
Barkley N.A Dean R.E Pittman R.N Wang M.L Holbrook C.C and Pederson G.A 2007 . Genetic diversity of cultivated and wild-type peanuts evaluated with M13-tailed SSR markers and sequencing . Genet. Res. Camb. 89 : 93 – 106 .
Bertioli D Moretzsohn M Madsen L Sandal N Leal-Bertioli S Guimaraes P Hougaard B Fredslund J Schauser L Nielsen A Sato S Tabata S Cannon S and Stougaard J 2009 . An analysis of synteny of Arachis with Lotus and Medicago sheds new light on the structure, stability and evolution of legume genomes . BMC Genomics 10 : 45 .
Bertioli D.J Leal-Bertioli S.C Lion M.B SantosPappas V.L.G Cannon S.B and Guimaraes P.M 2003 . A large scale analysis of resistance gene homologues in Arachis . Mol. Genet. Genomics 270 : 34 – 45 .
Bianchi-Hall C Keys R.D and Stalker H.T 1993 . Diversity of seed storage proteins in wild peanuts (Arachis species) . Plant Syst. Evol. 186 : 1 – 15 .
Bravo J.P Hoshino A.A Angelici C.M.L.C.D Lopes C.R and Gimenes M.A 2006 . Transferability and use of microsatellite markers for the genetic analysis of the germplasm of some Arachis section species of the genus Arachis . Genet. Mol. Biol. 29 : 516 – 524 .
Burow M.D Simpson C.E Paterson A.H and Starr J.L 1996 . Identification of peanut (Arachis hypogaea) RAPD markers diagnostic of root-knot nematode (Meloidogyne arenaria (Neal) Chitwood) resistance . Molecular Breed. 2 : 369 – 379 .
Burow M.D Simpson C.E Starr J.L Park C.H and Paterson A.H 2011 . QTL analysis of early leaf spot resistance and agronomic traits in an introgression population of peanut. Advances in Arachis through Genomics and Biotechnology . Brasilia. (abstr.) .
Burow M Simpson C Starr J and Paterson A 2001 . Transmission genetics of chromatin from a synthetic amphidiploid to cultivated peanut (Arachis hypogaea L.). Broadening the gene pool of a monophyletic polyploid species . Genetics 159 : 823 – 837 .
Burow M.D Starr J.L Park C.H Simpson C.E and Paterson A.H 2008 . Identification of QTLs for resistance to early leafspot (Cercospora arachidicola S. Hori) in an introgression population of peanut (Arachis hypogaea L.) . 2008 Plant and Animal Genomes Confr , San Diego, CA. (abstr.) .
Calbrix R.G Beilinson V Stalker H.T and Neilson N.C 2012 . Diversity of seed storage proteins of Arachis hypogaea and related species . Crop Sci. 52 : 1676 – 1688 .
Campbell W.V and Wynne J.C 1980 . Resistance of groundnut to insects and mites, pp. 149 – 157 . In: Proceedings of an International Workshop on Groundnuts, 13–17 Oct 1980 . ICRISAT , Patancheru, A. P. 502 324, India .
Campbell W.V Wynne J.C and Stalker H.T 1982 . Screening groundnut for Heliothis resistance, pp. 267 – 276 . In: Proceedings of the International Workshop on Heliothis Management, 15–20 Nov 1981 . ICRISAT Center , Patancheru A. P. 502 324, India .
Carvalho M.A and Quesenberry K.H 2009 . Characterization of Meloidogyne arenaria, M. javanica, and M. incognita reaction of the USA Arachis pintoi (Krapov. & W.C. Gregory) germplasm collection . Peanut Sci. 36 : 121 – 125 .
Castillo M.B Morrison L.S Russell C.C and Banks D.J 1973 . Resistance to Meloidogyne hapla in peanut . Jour. Nematol. 5 : 281 – 285 .
Chiteka Z.A Gorbet D.W Knauft D.A Shokes F.M and Kucharek T.A 1988a . Components of resistance to late leafspot in peanut. II. Correlations among components and their significance in breeding for resistance . Peanut Sci. 15 : 76 – 81 .
Chiteka Z.A Gorbet D.W Shokes F.M Kucharek T.A and Knauft D.A 1988b . Components of resistance to late leafspot in peanut. I. Levels and variability - implications for selection . Peanut Sci. 15 : 25 – 30 .
Chu Y Wu C.L Holbrook C.C Tillman B.L Person G and Ozias-Akins P 2011 . Marker-assisted selection to pyramid nematode resistance and the high oleic trait in peanut . Plant Genome 4 : 110 – 117 .
Church G.T Simpson C.E Burow M.D Paterson A.H and Starr J.L 2000 . Use of RFLP markers for identification of individuals homozygous for resistance to Meloidogyne arenaria in peanut . Nematology 2 : 575 – 580 .
Company M Stalker H.T and Wynne J.C 1982 . Cytology and leafspot resistance in Arachis hypogaea × wild species hybrids . Euphytica 31 : 885 – 893 .
Culver J.N and Sherwood J.L 1987 . Resistance to Peanut Stripe Virus in Arachis germplasm . Plant Dis. 71 : 1080 – 1082 .
Demski J.W and Sowell G 1981 . Resistance to peanut mottle virus in Arachis spp . Peanut Sci. 8 : 43 – 44 .
Dwivedi S.L Bertioli D.J Crouch J.H Valls J.F Upadhyaya H.D Favero A Moretzsohn M and Paterson A.H 2007 . Peanut, pp. 115 – 151 . In: Kole C (ed.) . Genome Mapping and Molecular Breeding in Plants, Vol. 2 Oilseeds . Springer-Verlab , Berlin Heidelberg .
Dwivedi S.L Gurtu S Chandra S Yuejin W and Nigam S.N 2001 . Assessment of genetic diversity among selected groundnut germplasm. I: RAPD analysis . Plant Breeding 120 : 345 – 349 .
Elshire R.J Glaubitz J.C Sun Q Poland J.A Kawamoto K Buckler E.S and Mitchell S.E 2011 . A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species . PLoS ONE 6 : e19379 .
Fávero A.P Simpson C.E Valls F.M.J and Velo N.A 2006 . Study of evolution of cultivated peanut through crossability studies among Arachis ipaënsis, A duranensis and A hypogaea . Crop Sci. 46 : 1546 – 1552 .
Fernandez A and Krapovickas A 1994 . Cromosomas y evolucion en Arachis (Leguminosae) . Bonplandia 8 : 1064 – 1070 .
Fitzner M.S Alderman S.C and Stalker H.T 1985 . Greenhouse evaluation of cultivated and wild peanut species for resistance to Cylindrocladium black rot . Proc. Amer. Peanut Res. Educ. Soc. 17 : 28 . (abstr.)
Fonceka D Hodo-Abalo T Rivallan R Faye I Sall M.N Ndoye O Favero A.P Bertioli D.J Glaszmann J.C Courtois B and Rami J.F 2009 . Genetic mapping of wild introgressions into cultivated peanut: a way toward enlarging the genetic basis of a recent allotetraploid . BMC Plant Biol. 9 : 103 .
Fonceka D A Tossim H Rivallan R Vignes H Faye I Ndoye O Moretzsohn M Bertioli D C Glaszmann J Courtois B and F Rami J 2012 . Fostered and left behind alleles in peanut: interspecific QTL mapping reveals footprints of domestication and useful natural variation for breeding . BMC Plant Biol. 12 : 26 .
Friend S.A Quandt D Tallury S.P Stalker H.T and Hilu K.W 2010 . Species, genomes and section relationships in genus Arachis (Fabaceae): A molecular phylogeny . Plant Syst. Evol. 290 : 185 – 199 .
Galgaro L Lopes C.R Gimenes M Valls J.F.M and Kochert G 1998 . Genetic variation between several species of sections Extranervosae, Caulorrhizae, Heteranthae, and Triseminatae (genus Arachis) estimated by DNA polymorphism . Genome 41 : 445 – 454 .
Garcia G.M Stalker H.T and Kochert G.A 1995 . Introgression analysis of an interspecific hybrid population in peanuts (Arachis hypogaea L.) using RFLP and RAPD markers . Genome 38 : 166 – 176 .
Garcia G.M Stalker H.T Shroeder E and Kochert G 1996 . Identification of RAPD, SCAR, and RFLP markers tightly linked to nematode resistance genes introgressed from Arachis cardenasii into Arachis hypogaea . Genome 39 : 836 – 845 .
Garcia G.M Stalker H.T Schroeder E Lyerly J.H and Kochert G 2005 . A RAPD-based linkage map of peanut based on a backcross population between the two diploid species Arachis stenosperma and A. cardenasii . Peanut Sci. 32 : 1 – 8 .
Gautami B Foncéka D Pandey M.K Moretzsohn M.C Sujay V Qin H Hong Y Faye I Chen X BhanuPrakash A Shah T.M Gowda M.V.C Nigam S.N Liang X Hoisington D.A Guo B Bertioli D.J F Rami J and K Varshney R 2012 . An international reference consensus genetic map with 897 marker loci based on 11 mapping populations for tetraploid groundnut (Arachis hypogaea L.) . PLoS ONE. 7 : e41213 .
Gibbons R.W 1969 . Groundnut rosette research in Malawi, pp. 1 – 8 . In: Proceedings of the Third Eastern African Cereals Research Conference, Mar 1969, Zambia and Malawi . Ministry of Agriculture and Natural Resources , Lilongwe, Malawi .
Gibbons R.W and Bailey B.E 1967 . Resistance to Cercospora arachidicola in some species of Arachis . Rhod. Zam. Mal. Jour. Agric. Res. 5 : 57 .
Gimenes M.A Lopes C.R Galgaro M.L Valls J.F.M and Kochert G 2000 . Genetic variation and phylogenetic relationships based on RAPD analysis in section Caulorrhizae, genus Arachis (Leguminosae) . Euphytica 116 : 187 – 195 .
Gimenes M.A Lopes C.R Galgaro M.L Valls J.F and Kochert G 2002 . RFLP analysis of genetic variation in species of section Arachis, genus Arachis (Leguminosae) . Euphytica 123 : 421 – 429 .
Green C.C and Wynne J.C 1986 . Diallel and generation means analyses for the components of resistance to Cercospora arachidicola in peanut . Theor. Appl. Genet. 73 : 228 – 235 .
Gregory M.P and Gregory W.C 1979 . Exotic germplasm of Arachis L. interspecific hybrids . Jour. Hered. 70 : 185 – 193 .
Gregory W.C 1946 . Peanut breeding program underway. Research and farming . Annual Report, North Carolina Agricultural Experiment Station 69 : 42 – 44 .
Grieshammer U and Wynne J.C 1990 . Isozyme variability in mature seeds of U.S. peanut cultivars and collections . Peanut Sci. 17 : 72 – 75 .
Guimarães P.M Brasileiro A.C.M Araújo A.C.G Leal-Bertioli S.C.M da Silva F.R and Morgante C.V 2010 . A study of gene expression in the nematode resistant wild peanut relative, Arachis stenosperma, in response to challenge with Meloidogyne arenaria . Trop. Plant Biol. . DOI 10.1007/s12042-010-9056-z .
Guimarães P.M Brasileiro A.C.M Leal-Bertioli S.C.M Pappas G Togawa R and Bonfim O 2011 . Comparative 454 pyrosequencing of transcripts from two wild Arachis genotypes under biotic and abiotic stress . In Proceedings: Plant and Animal Genome XIX Conference, 15–19th Jan 2011 , San Diego, USA. (abstr.) .
Guo Y Khanal S Tang S Bowers J.E Heesacker A.F Khalilian N Nagy E.D Zhang D Taylor C.A Stalker H.T Ozias-Akins P and Knapp S.J 2012 . Comparative mapping in intraspecific populations uncovers a high degree of macrosynteny between A- and B- genome diploid species of peanut . BMC Genomics 13 : 608 .
Halward T.M Stalker H.T and Kochert G 1993 . Development of an RFLP linkage map in diploid peanut species . Theor. Appl. Genet. 87 : 379 – 384 .
Halward T Stalker T LaRue E and Kochert G 1992 . Use of single-primer DNA amplifications in genetic studies of peanut (Arachis hypogaea L.) . Plant Molec. Biol. 18 : 315 – 25 .
Halward T.M Stalker H.T Larue E.A and Kochert G 1991 . Genetic variation detectable with molecular markers among unadapted germ-plasm resources of cultivated peanut and related wild species . Genome 34 : 1013 – 1020 .
Hammons R.O 1970 . Registration of Spancross peanut . Crop Sci. 10 : 459 .
He G and Prakash C.S 1997 . Identification of polymorphic DNA markers in cultivated peanut (Arachis hypogaea L.) . Euphytica 97 : 143 – 149 .
He G.H Meng R Gao H Guo B Gao G Newman M Pittman R.N and Prakash C.S 2005 . Simple sequence repeat markers for botanical varieties of cultivated peanut (Arachis hypogaea L.) . Euphytica 142 : 131 – 136 .
Hebert T.T and Stalker H.T 1981 . Resistance to peanut stunt virus in Arachis species . Peanut Sci. 8 : 45 – 47 .
Hernandez-Garay A Sollenberger L.E Staples C.R and Pedreria C.G.S 2004 . ‘Florigraze’ and ‘Arbrook’ rhizome peanut as pasture for growing Holstein heifers . Crop Sci. 44 : 1355 – 1360 .
Herselman L 2003 . Genetic variation among Southern African cultivated peanut (Arachis hypogaea L.) genotypes as revealed by AFLP analysis . Euphytica 133 : 319 – 327 .
Herselman L Thwaites R Kimmins F.M Courtois B van der Merwe P.J and Seal S.E 2004 . Identification and mapping of AFLP markers linked to peanut (Arachis hypogaea L.) resistance to the aphid vector of groundnut rosette disease . Theor. Appl. Genet. 109 : 1426 – 1433 .
Hilu K.W and Stalker H.T 1995 . Genetic relationships between peanut and wild species of Arachis sect. Arachis (Fabaceae): Evidence from RAPDs . Plant Syst. Evol. 198 : 167 – 178 .
Holbrook C.C and Noe J.P 1990 . Resistance to Meloidogyne arenaria in Arachis spp. and the implications on development of resistant peanut cultivars . Peanut Sci. 17 : 35 – 38 .
Holbrook C.C Timper P Culbreath A.K and Kvien C.K 2008 . Registration of ‘Tifguard’ peanut . Jour. Plant Reg. 2 : 2 .
Holbrook C.C and Stalker H.T 2003 . Peanut breeding and genetic resources . Plant Breed. Rev. 22 : 297 – 356 .
Hong Y.B Chen X.P Liang X.Q Liu H.Y Zhou G.Y Li S.X Wen S.J Holbrook C.C and Guo B.Z 2010 . A SSR-based composite genetic linkage map for the cultivated peanut (Arachis hypogaea L.) genome . BMC Plant Biol. 10 : 1 – 13 .
Hong Y.B Liang X.Q Chen X.P Liu H.Y Zhou G.Y Li S.X and Wen S.J 2008 . Construction of genetic linkage map based on SSR markers in peanut (Arachis hypogaea L.) . Agric. Sci. in China 7 : 915 – 921 .
Hopkins M.S Casa A.M Wang T Mitchell S.E Dean R.E Kochert G.D and Kresovich S 1999 . Discovery and characterization of polymorphic simple sequence repeats (SSRs) in cultivated peanut (Arachis hypogaea L.) . Crop Sci. 39 : 1243 – 1247 .
Hoshino A.A Bravo J.P Angelici C.M.L.C.D Barbosa A.V.G Lopes C.R and Gimenes M.A 2006 . Heterologous microsatellite primer pairs informative for the whole genus Arachis . Genet. Mol. Biol. 29 : 665 – 675 .
Hull F.H and Carver W.A 1938 . Peanut improvement . Ann. Report, Florida Agric. Exp. Sta. 39 – 40 .
Husted L 1936 . Cytological studies of the peanut Arachis. II. Chromosome number, morphology and behavior and their application to the origin of cultivated forms . Cytologia 7 : 396 – 423 .
ICRISAT (International Crops Research institute for the Semi-Arid Tropics) . 1985 , ICRISAT Annual Report 1984 , Patancheru, A.P. 502 324, India .
ICRISAT (International Crops Research institute for the Semi-Arid Tropics) . 1990 , ICRISAT Annual Report 1989 , Patancheru, A.P. 502 324, India .
ICRISAT (International Crops Research institute for the Semi-Arid Tropics) . 2000 , ICRISAT Annual Report 1999 . ICRISAT , Patancheru, A.P. 502 324, India .
Iroume R.N and Knauft D.A 1987 . Selection indices for simultaneous selection for pod yield and leafspot resistance in peanut (Arachis hypogaea L.) . Peanut Sci. 14 : 51 – 54 .
Isleib T.G Milla-Lewis S.R Pattee H.E Copeland S.C Zuleta M.C Shew B.B Hollowell J.E Sanders T.H Dean L.O Hendrix K.W Balota M and Chapin J.W 2010 . Registration of ‘Bailey’ peanut . Jour. Plant Reg. 5 : 27 – 39 .
Isleib T.G Rice P.W Mozingo R.W Copeland S.C Graeber J.B and Stalker H.T 2006 . Registration of N96076L peanut germplasm . Crop Sci. 46 : 2329 – 2330 .
Janini J.C Boiça Júnior A.L Godoy I.J Michelotto M.D and Fávero A.P 2010 . Avaliação de espécies silvestres e cultivares de amendoim para resistência a Enneothrips flavens Moulton . Bragantia 69 : 891 – 898 .
Johnson D.R Wynne J.C and Campbell W.V 1977 . Resistance of wild species of Arachis . Peanut Sci. 4 : 9 – 11 .
Klesser P.J 1965 . Groundnut Ringspot Virus-A new sap-transmissible virus of Arachis hypogaea and A. monticola . S. Afr. Jour. Agr. Sci. 9 : 711 – 720 .
Khedikar Y Gowda M Sarvamangala C Patgar K Upadhyaya H and Varshney R 2010 . A QTL study on late leaf spot and rust revealed one major QTL for molecular breeding for rust resistance in groundnut (Arachis hypogaea L.) . Theor. Appl. Genet. 121 : 971 – 984 .
Kochert G Halward T Branch W.D and Simpson C.E 1991 . RFLP variability in peanut (Arachis hypogaea L.) cultivars and wild species . Theor. Appl. Genet. 81 : 565 – 570 .
Kochert G Stalker H.T Gimenes M Galgaro L Lopes C.R and Moore K 1996 . RFLP and cytogenetic evidence on the origin and evolution of allotetraploid domesticated peanut, Arachis hypogaea (Leguminosae) . Amer. Jour. Bot. 83 : 1282 – 1291 .
Kolawole K.B 1976 . A short progress report on transfer of Cercospora resistant traits to the cultivated Arachis hypogaea L. Samaru Agric . Newslett. 18 : 40 – 43 .
Koppolu R Upadhyaya H.D Dwivedi S.L Hoisington D.A and Varshney R.K 2010 . Genetic relationships among seven sections of the genus Arachis studied by using SSR markers . BMC Plant Biol. 10 : 15 .
Krapovickas A and Gregory W.C 1994 . Taxonomy of the genus Arachis (Leguminosae) . Bonplandia 8 : 1 – 186 .
Krapovickas A and Rigoni V.A 1951 . Estudios citologicas en el genero Arachis . (In Spanish) Revista de Investigaciones Agricolas 5 : 289 – 293 .
Krishna T.G and Mitra R 1988 . The probable genome donors to Arachis hypogaea L. based on arachin seed storage protein . Euphytica 37 : 47 – 52 .
Krishna G.K Zhang J Burow M Pittman R.N Delikostadinov S.G Lu Y and Puppala N 2004 . Genetic diversity analysis in Valencia peanut (Arachis hypogaea L.) using microsatellite markers . Cell. Mol. Biol. Lett. 9 : 111 – 120 .
Lanham P.G Fennell S P Moss J and Powell W 1992 . Detection of polymorphic loci in Arachis germplasm using random amplified polymorphic DNAs . Genome 35 : 885 – 889 .
Leal-Bertioli S.C.M Jose A.C.V.F Alves-Freitas D.M.T Moretzsohn M.C Guimaraes P.M Nielen S Vidigal B.S Pereira R.W Pike J Favero A.P Parniske M Varshney R.K and Bertioli D.J 2009 . Identification of candidate genome regions controlling disease resistance in Arachis . BMC Plant Biology 9 : 112 .
Leal-Bertioli S.C.M Farias M.P Silva P.T Guimarães P.M Brasileiro A.C.M Bertioli D.J and Araujo A.C.G 2010 . Ultrastructure of the initial interaction of Puccinia arachidis and Cercosporidium personatum with leaves of Arachis hypogaea and Arachis stenosperma . Jour. Phytopathology 158 : 792 – 796 .
Leuck B D and Hammons R.O 1968 . Resistance of wild peanut plants to the mite Tetranychus tumidellus . Jour. Econ. Entomol. 61 : 73 – 82 .
Leuck D.B Hammons R.O and Morgan L.W 1968 . Peanut host plant resistance studies-1967 , 12 pp. USDA Spec. Rpt. No. X321 .
Liang X.Q Luo M Holbrook C.C and Guo B.Z 2006 . Storage protein profiles in Spanish and runner market type peanuts and potential markers . BMC Plant Biol. 6 : 24 .
Lu J and Pickersgill B 1993 . Isozyme variation and species relationships in peanut and its wild relatives (Arachis L. - Leguminosae) . Theor. Appl. Genet. 85 : 550 – 560 .
Lyerly J.H Stalker H.T Moyer J.W and Hoffman K 2002 . Evaluation of the wild species of peanut for resistance to tomato spotted wilt virus . Peanut Sci. 29 : 79 – 84 .
Lynch R.E Branch W.D and Garner J.W 1981 . Resistance of Arachis species to the fall armyworm, Spodoptera frugiperda . Peanut Sci. 8 : 106 – 109 .
Mallikarjuna N 2005 . Production of hybrids between Arachis hypogaea and A. chiquitana (section Procumbentes) . Peanut Sci. 32 : 148 – 152 .
Mallikarjuna N Pande S Jadhav D.R Sastri D.C and Rao J.N 2004 . Introgression of disease resistance genes from Arachis kempff-mercadoi into cultivated groundnut . Plant Breed. 123 : 573 – 576 .
Mathews B.W Carpenter J.R Cleveland E Gibson Z and Niino-Duponte R.Y 2000 . Perrennial forage peanut (Arachis pintoi) in pastures for raising replacement heifers/stocker steers in Hawaii . Jour. Hawaiian Pacific Agric. 11 : 1 – 10 .
Mehan V.K McDonald D Ranakrishna N and Williams J.H 1986 . Effect of genotype and date of harvest on infection of peanut seed by Aspergillus flavus and subsequent contamination with aflatoxin . Peanut Sci. 13 : 46 – 50 .
Mendes A.G.T 1947 . Estudos citológicos no gênero Arachis . Bragantia 7 : 257 – 267 .
Melouk H.A Sanborn M.R and Banks D.J 1984 . Sources of resistance to Peanut Mottle Virus in Arachis germplasm . Plant Dis. 68 : 563 – 564 .
Milla S.R 2003 . Relationship and utilization of Arachis germplasm in peanut improvement, Ph.D. Dissertation, North Carolina State University, Raleigh .
Milla S.R Tallury S.P Stalker H.T and Isleib T.G 2004 . Identification of molecular markers associated with tomato spotted wilt virus in a genetic linkage map of Arachis kuhlmannii x A. diogoi . Proc. Amer. Peanut Res. Educ. Soc. 36 : 27 . (abstr.)
Milla S.R Isleib T.G and Stalker H.T 2005a . Taxonomic relationships among Arachis sect. Arachis species as revealed by AFLP markers . Genome 48 : 1 – 11 .
Milla S.R Isleib T.G and Tallury S.P 2005b . Identification of AFLP markers linked to reduced aflatoxin accumulation in A. cardenasii-derived germplasm lines of peanut . Proc. Amer. Peanut Res. Educ. Soc. 37 : 89 . (abstr.)
Moretzsohn M Barbosa A Alves-Freitas D Teixeira C Leal-Bertioli S Guimaraes P Pereira R Lopes C Cavallari M Valls J Bertioli D and Gimenes M 2009 . A linkage map for the B-genome of Arachis (Fabaceae) and its synteny to the A-genome . BMC Plant Biol. 9 : 40 .
Moretzsohn M.C Gouvea E.G Inglis P.W Leal-Bertioli S.C.M Valls J.F.M and Bertioli D.J 2013 . A study of the relationships of cultivated peanut (Arachis hypogaea) and its most closely related wild species using intron sequences and microsatellite markers . Annals of Botany 111 : 113 – 126 .
Moretzsohn M.C Hopkins M.S Mitchell S.E Kresovich S Valls J.F.M and Ferreira M.E 2004 . Genetic diversity of peanut (Arachis hypogaea L.) and its wild relatives based on the analysis of hypervariable regions of the genome . BMC Plant Biol. 4 : 11 .
Moretzsohn M Leoi L Proite K Guimarães P Leal-Bertioli S Gimenes M Martins W Valls J Grattapaglia D and Bertioli D 2005 . A microsatellite-based, gene-rich linkage map for the AA genome of Arachis (Fabaceae) . Theor. Appl.Genet. 111 : 1060 – 1071 .
Nagy E.D Chu Y Guo Y Khanal S Tang S Li Y Dong W.B Timper P Taylor C Ozias-Akins P Holbrook C.C Beilinson V Neilson N.C Stalker H.T and Knapp S.J 2010 . Recombination is suppressed in an alien introgression in peanut harboring Rma, a dominant root-knot nematode resistance gene . Molec. Breed. 26 : 357 – 370 .
Nagy E.D Guo Y Tang S Bowers J.E Okashah R.A Taylor C.A Zhang D Khanal S Heesacker A.F Khalilian N Farmer A.D Carrasquilla-Garcia N Penmetsa R.V Cook D Stalker H.T Nielsen N Ozias-Akins P and Knapp S.J 2012 . A high-density genetic map of Arachis duranensis, a diploid ancestor of cultivated peanut . BMC Genomics 13 : 469 .
Nelson A Samuel D.M Tucker J Jackson C and Stahlecker-Roberson A 2006 . Assessment of genetic diversity and sectional boundaries in tetraploid peanuts (Arachis) . Peanut Sci. 33 : 64 – 67 .
Nelson S.C Simpson C.E and Starr J.L 1989 . Resistance to Meloidogyne arenaria in Arachis sp. germplasm . Jour. Nematol. 21 : 654 – 660 .
Nigam S.N Dwivedi S.L and Gibbons R.W 1991 . Groundnut breeding: Constraints, achievements, and future possibilities . Plant Breed. 61 : 1127 – 1136 .
Paik-Ro O.G Smith R.L and Knauft D.A 1992 . Restriction fragment length polymorphism evaluation of six peanut species within the Arachis section . Theor. Appl. Genet. 84 : 201 – 208 .
Pande S and Rao J.N 2001 . Resistance of wild Arachis species to late leaf spot and rust in greenhouse trials . Plant Dis. 85 : 851 – 855 .
Pandey M Gautami B Jayakumar T Sriswathi M Upahyaya H.D Gowda M.V.C Radhakrishnan T Bertioli D.J Knapp S.J Cook D.R and Varshney R.K 2011 . Highly informative genic and genomic SSR markers to facilitate molecular breeding in cultivated groundnut (Arachis hypogaea) . Plant Breed. 131 : 139 – 147 .
Pandey M.K Monyo E Ozias-Akins P Liang X Guimarães P Nigam S.N Upadhyaya H.D Janila P Zhang X Guo B Cook D.R Bertioli D.J Michelmore R and Varshney R.K 2012 . Advances in Arachis genomics for peanut improvement . Biotechnol. Adv. 30 : 639 – 651 .
Penaloza A.P.S and Valls J.F.M 2005 . Chromosome number and satellite chromosome morphology of eleven species of Arachis (Leguminosae) . Bonplandia. 14 : 65 – 72 .
Proite K Carneiro R Falcão R Gomes A Leal-Bertioli S Guimarães P and Bertioli D 2008 . Post-infection development and histopathology of Meloidogyne arenaria race 1 on Arachis spp . Plant Pathology 57 : 974 – 980 .
Qin H Feng S Chen C Guo Y Knapp S Culbreath A He G Wang M Zhang X Holbrook C Ozias-Akins P and Guo B 2012 . An integrated genetic linkage map of cultivated peanut (Arachis hypogaea L.) constructed from two RIL populations . Theor. Appl. Genet. 124 : 653 – 664 .
Raina S.N and Mukai Y 1999 . Genomic in situ hybridization in Arachis (Fabaceae) identifies the diploid wild progenitors of cultivated (A. hypogaea) and related wild (A. monticola) peanut species . Plant Syst. Evol. 214 : 251 – 262 .
Raina S.N Rani V Kojima T Ogihara Y Singh K.P and Devarumath R.M 2001 . RAPD and ISSR fingerprints as useful genetic markers for analysis of genetic diversity, varietal identification, and phylogenetic relationships in peanut (Arachis hypogaea) cultivars and wild species . Genome 44 : 763 – 772 .
Raman V.S and Kesavan P.C 1962 . Studies on a diploid interspecific hybrid in Arachis . Nucleus 5 : 123 – 126 .
Ravi K Vadez V Isobe S Mir R Guo Y Nigam S Gowda M Radhakrishnan T Bertioli D Knapp S and Varshney R 2011 . Identification of several small main-effect QTLs and a large number of epistatic QTLs for drought tolerance related traits in groundnut (Arachis hypogaea L.) . Theor. Appl. Genet. 122 : 1119 – 1132 .
Reddy A.S Reddy L.J Mallikarjuna N Abdurahman M.D Reddy Y.V Bramel P.J and Reddy D.V.R 2000 . Identification of resistance to peanut bud necrosis virus (PBNV) in wild Arachis germplasm . Ann. Appl. Biol. 137 : 135 – 139 .
Robledo G Lavia G.I and Seijo J.G 2009 . Species relations among wild Arachis species with the A genome as revealed by FISH mapping of rDNA loci and heterochromatin detection . Theor. Appl. Genet. 118 : 1295 – 1307 .
Robledo G and Seijo G 2010 . Species relationships among the wild B genome of Arachis species (section Arachis) based on FISH mapping of rDNA loci and heterochromatin detection: A new proposal for genome arrangement . Theor. Appl. Genet. 121 : 1033 – 1046 .
Santos S.P Dantas K Leal-Bertioli S.C.M Nielen S Moretzsohn M.C Guimarães P.M Micas G and Bertioli D.J 2011 . New synthetic tetraploids for the introgression of wild alleles into cultivated peanut . Advances in Arachis through Genomics and Biotechnology , Brasília , Embrapa. (abstr.)
Sarvamangala C Gowda M.V.C and Varshney R.K 2011 . Identification of quantitative trait loci for protein content, oil content and oil quality for groundnut (Arachis hypogaea L.) . Field Crops Res. 122 : 49 – 59 .
Sato S Nakamura Y Kaneko T Asamizu E Kato T Nakao M Sasamoto S Watanabe A Ono A Kawashima K Fujishiro T Katoh M Kohara M Kishida Y Minami C Nakayama S Nakazaki N Shimizu Y Shinpo S Takahashi C Wada T Yamada M Ohmido N Hayashi M Fukui K Baba T Nakamichi T Mori H and Tabata S 2008 . Genome structure of the legume, Lotus japonicus . DNA Res. 15 : 227 – 239 .
Seijo J.G Lavia G.I Fernandez A Krapovickas A Ducasse D and Moscone E.A 2004 . Physical mapping of the 5S and 18S–25S rRNA genes by FISH as evidence that Arachis duranensis and A. ipaensis are the wild diploid progenitors of A. hypogaea (Leguminosae) . Amer. Jour. Bot. 91 : 1294 – 1303 .
Sharief Y Rawlings J.O and Gregory W.C 1978 . Estimates of leafspot resistance in three interspecific hybrids of Arachis . Euphytica 27 : 741 – 751 .
Sharma H.C Pampapathy G Dwivedi S.L and Reddy L.J 2003 . Mechanisms and diversity of resistance to insect pests in wild relatives of groundnut . Jour. Econ. Entomol. 96 : 1886 – 1897 .
Sharma S.B Ansari M.A Varaprasad K.S Singh A.K and Reddy L.J 1999 . Resistance to Meloidogyne javanica in wild Arachis species . Genet. Resour. Crop Evol. 46 : 557 – 568 .
Shirasawa K Bertioli D.J Varshney R.K Moretzsohn M.C Leal-Bertioli S.C.M Thudi M Pandey M.K F Rami J Foncéka D Gowda M.V.C Qin H Guo B Hong Y Liang X Hirakawa H Tabata S and Isobe S 2012a . Integrated consensus map of cultivated peanut and wild relatives reveals structures of the A and B genomes of Arachis and divergence of the legume genomes, . DNA Research doi:10.1093/dnares/dss042 .
Shirasawa K Koilkonda P Aoki K Hirakawa H Tabata S Watanabe M Hasegawa M Kiyoshima H Suzuki S Kuwata C Naito Y Kuboyama T Nakaya A Sasamoto S Watanabe A Kato M Kawashima K Kishida Y Kohara M Kurabayashi A Takahashi C Tsuruoka H Wada T and Isobe S 2012b . In silico polymorphism analysis for the development of simple sequence repeat and transposon markers and construction of linkage map in cultivated peanut . BMC Plant Biol. 12 : 80 .
Simpson C.E Nelson S.C Starr J.L Woodard K.E and Smith O.D 1993 . Registration of TxAG-6 and TxAG-7 peanut germplasm lines . Crop Sci. 33 : 1418 .
Simpson C.E and Smith O.D 1975 . Registration of Tamnut 74 peanut . Crop Sci. 15 : 603 – 604 .
Simpson C and Starr J 2001 . Registration of ‘COAN’ peanut . Crop Sci. 41 : 918 .
Simpson C.E Starr J.L Church G.T Burow M.D and Paterson A.H 2003 . Registration of “NemaTAM” Peanut . Crop Sci. 43 : 1561 .
Singh A.K Krishnan S.S Mengesha M.H and Ramaiah C.D 1991 . Phylogenetic relationships in section Arachis based on seed protein profiles . Theor. Appl. Genet. 82 : 593 – 597 .
Singsit C Holbrook C.C Culbreath A.K and Ozias-Akins P 1995 . Progenies of an interspecific hybrid between Arachis hypogaea and A. stenosperma - pest resistance and molecular homogeneity . Euphytica 83 : 9 – 14 .
Singsit C and Ozias-Akins P 1992 . Rapid estimation of ploidy levels in in vitro-regenerated interspecific Arachis hybrids and fertile triploids . Euphytica 64 : 183 – 188 .
Smartt J and Stalker H.T 1982 . Speciation and cytogenetics in Arachis, pp. 21 – 49 . In: Pattee H.E and Young C.E (eds.) . Peanut Science and Technology . Amer. Peanut Res. Educ. Soc. , Yoakum, TX .
Stalker H.T 1981 . Hybrids in the genus Arachis between sections Erectoides and Arachis . Crop Sci. 21 : 359 – 362 .
Stalker H.T 1985 . Cytotaxonomy of Arachis, pp. 65 – 79 . In: International Workshop on Cytogenetics of Arachis . International Crops Research Institute for Semi-Arid Tropics , Hyderabad, A.P., India .
Stalker H.T 1991 . A new species in section Arachis of peanuts with a D genome . Amer. Jour. Bot. 78 : 630 – 637 .
Stalker H.T 1992 . Utilizing Arachis germplasm resources, pp. 281 – 295 . In: Nigam S.N. (ed) Groundnut, a Global Perspective: Proceedings of an International Workshop, 25–29 Nov 1991 . ICRISAT Center , Patancheru, Andhra Pradesh, India .
Stalker H.T Beute M.K Shew B.B and Barker K.R 2002a . Registration of two root-knot nematode-resistant peanut germplasm lines . Crop Sci. 42 : 312 – 313 .
Stalker H.T Beute M.K Shew B.B and Isleib T.G 2002b . Registration of five leaf spot-resistant peanut germplasm lines . Crop Sci. 42 : 314 – 316 .
Stalker H.T and Campbell W.V 1983 . Resistance of wild species of peanuts to an insect complex . Peanut Sci. 10 : 30 – 33 .
Stalker H.T Campbell W.V and Wynne J.C 1984 . Evaluation of cultivated and wild peanut species for resistance to the lesser cornstalk borer (Lepidoptera: Pyralidae) . Jour. Econ. Entom. 77 : 53 – 57 .
Stalker H.T and Dalmacio R.D 1986 . Karyotype analysis and relationships among varieties of Arachis hypogaea L . Cytologia 58 : 617 – 629 .
Stalker H.T Dhesi J.S and Kochert G.D 1995 . Variation within the species A. duranensis, a possible progenitor of the cultivated peanut . Genome 38 : 1201 – 1212 .
Stalker H.T Dhesi J.S Parry D.C and Hahn J.H 1991 . Cytological and interfertility relationships of Arachis section Arachis . Amer. Jour. Bot. 78 : 238 – 246 .
Stalker H.T and Lynch R.E 2002 . Registration of four insect-resistant peanut germplasm lines . Crop Sci. 42 : 313 – 314 .
Stalker H.T and Moss J.P 1987 . Speciation, cytogenetics, and utilization of Arachis species . Adv. Agron. 41 : 1 – 40 .
Stalker H.T and Mozingo I.G 2001 . Molecular markers of Arachis and marker-assisted selection . Peanut Sci. 28 : 117 – 123 .
Stalker H.T Phillips T.D Murphy J.P and Jones T.M 1994 . Variation of isozyme patterns among Arachis species . Theor. Appl. Genet. 87 : 746 – 755 .
Stalker H.T and Simpson C.E 1995 . Germplasm resources in Arachis, pp. 14 – 53 . In: Advances in Peanut Science , Pattee H.E and Stalker H.T (eds.) . Amer. Peanut Res. and Educ. Soc. , Stillwater, OK .
Stevenson P.C Blaney W.M Simmonds M.J.S and Wightman J.A 1993 . The identification and characterization of resistance in wild species of Arachis to Spodoptera litura (Lepidoptera: Noctuidae) . Bull. Entom. Res. 83 : 421 – 429 .
Subrahmanyam P Mehan V.K Nevill D.J and McDonald D 1980 . Research on fungal diseases of groundnut at ICRISAT, pp. 193 – 198 . In: Proceedings of the International Workshop on Groundnuts, 13–17 Oct. 1980, India: International Crops Research Institute for the Semi-Arid Tropics . Patancheru, A.P. 502 324, India .
Subrahmanyam P Moss J.P and Rao V.R 1983 . Resistance to peanut rust in wild Arachis species . Plant Dis. 67 : 209 – 212 .
Subrahmanyam P Nolt A.M Reddy B.L Reddy D.V.R and McDonald D 1985b . Resistance to groundnut diseases in wild Arachis species, pp. 49 – 55 . In: Proceedings of an international workshop on cytogenetics of Arachis, 31 Oct–2 Nov 1983 . ICRISAT Centre , Patancheru, Andhra Pradesh, India .
Subrahmanyam P Smith D.H and Simpson C.E 1985c . Resistance to Didymella arachidicola in wild Arachis species . Oleagineux 40 : 53 – 56 .
Subrahmanyam P Moss J.P McDonald D Rao P.V.S and Rao V.R 1985a . Resistance to leaf spot caused by Cercosporidium personatum in wild Arachis species . Plant Dis. 69 : 951 – 954 .
Subrahmanyam P Naidu R.A Reddy L.J Lava Kumar P and Ferguson M.E 2001 . Resistance to groundnut rosette disease in wild Arachis species . Ann. Appl. Biol. 139 : 45 – 50 .
Subramanian V Gurtu S Nageswara Rao R.C and Nigam S.N 2000 . Identification of DNA polymorphism in cultivated groundnut using random amplified polymorphic DNA (RAPD) assay . Genome 43 : 656 – 660 .
Sujay V Gowda M.V.C Pandey M.K Bhat R.S Khedikar Y.P Nadaf H.L Gautami B Sarvamangala C Lingaraju S Radhakrishan T Knapp S.J and Varshney R.K 2011 . Quantitative trait locus analysis and construction of consensus genetic map for foliar disease resistance based on two recombinant inbred line populations in cultivated groundnut (Arachis hypogaea L.) . Molec. Breed. . 10.1007/s11032-011-9661-z .
Tallury S.P Hilu K.W Milla S.R Friend S.A Alsaghir M Stalker H.T and Quandt D 2005 . Genomic affinities in Arachis section Arachis (Fabaceae): Molecular and cytogenetic evidence . Theor. Appl. Genet. 111 : 1229 – 1237 .
Tallury S.P Hollowell J and Isleib T.G 2011 . Greenhouse evaluation of section Arachis wild species for Sclerotinia blight and CBR resistance . Proc. Amer. Peanut. Res. and Educ. Soc. 43 : 69 .
Tang R Gao G He L Han Z Shan S Zhong R Zhou C Jiang J Li Y and Zhuang W 2007 . Genetic diversity in cultivated groundnut based on SSR markers . Jour. Genet. Genomics 34 : 449 – 459 .
Valls J.F.M and Simpson C.E 2005 . New species of Arachis (Leguminosae) from Brazil, Paraguay and Bolivia . Bonplandia 14 : 35 – 63 .
Varshney R.K Bertioli D.J Moretzsohn M.C Vadez V Krishnamurty L Aruna R Nigam S.N Ravi K He G Knapp S.J and Hoisington D.A 2009a . The first SSR based genetic linkage map for cultivated groundnut (Arachis hypogaea L.) . Theor. Appl. Genet. 118 : 729 – 39 .
Varshney R.K Mahendar T Aruna R Nigam S.N Neelima K Vadez V and Hoisington D.A 2009b . High level of natural variation in a groundnut (Arachis hypogaea L.) germplasm collection assayed by selected informative SSR markers . Plant Breed. 128 : 486 – 494 .
Waliyar F McDonald D Subba Rao P.V and Reddy P.M 1993 . Components of resistance to an Indian source of Cercospora arachidicola in selected peanut lines . Peanut Sci. 20 : 93 – 96 .
Waliyar F Shew B.B Sidahmed R and Beute M.K 1995 . Effects of host resistance on germination of Cercospora arachidicola on peanut leaf surfaces . Peanut Sci. 22 : 154 – 157 .
Wang H Penmetsa R.V Yuan M Gong L Zhao Y Guo B Farmer A Rosen B Gao J Isobe S Bertioli D Varshney R Cook D and He G 2012 . Development and characterization of BAC-end sequence derived SSRs, and their incorporation into a new higher density genetic map for cultivated peanut (Arachis hypogaea L.) . BMC Plant Biol. 12 : 10 .
Xue H.Q Isleib T.G Stalker H.T Payne G.A and O'Brian G 2004 . Evaluation of Arachis species and interspecific tetraploid lines for resistance to alfatoxin production by Aspergillus flavus . Peanut Sci. 31 : 134 – 131 .
Notes
- Department of Crop Science, North Carolina State University, Raleigh, NC 27695
- Department of Horticulture, The University of Georgia, Tifton, GA, 31973
- Department of Gentics and Morphology, University of Brasilia, Campus Darcy Ribeiro, Brasília, DF. Brazil
- PBI, EMBRAPA Recursos Genéticos e Biotecnologia, Brasilia, Brazil * Corresponding author (email: tom_stalker@ncsu.edu)
Author Affiliations