Introduction
Growers rely on a variety of inoculants, agrochemicals, and plant growth regulators to control pests and manage peanut growth and development. Peanut is a leguminous crop, capable of biological nitrogen fixation (BNF) (Schiffman and Alper, 1968; Shimshi et al., 1967; Walker et al., 1976). The infection of the roots by rhizobium spp. is necessary for BNF to occur (Schiffman and Alper, 1968; Walker et al., 1976). Rhizobium spp. may occur naturally in the soil or may be applied in-furrow at planting using commercially available inoculants. Successful inoculation is important to prevent nitrogen deficiency in peanut. Inoculants are living organisms that are sensitive to temperature, sunlight, and have a limited shelf life (Anonymous, 2010). Fungicidal seed treatments have been shown to decrease inoculant viability resulting in reduced nodulation, BNF, plant growth, and crop yield (Hashem et al., 1997; Rubens Jose, 2009).
Tobacco thrips, spider mites (Tetranychus spp.), and heliothines (Helicoverpa virescens and Heliothis zea F.) are major insect pests in peanut that may limit growth and yield (Weiss, 2000; Wightman and Rao, 1994). Tobacco thrips may cause peanut stunting when plants are young and act as vectors of tomato spotted wilt (caused by Tospovirus) (Reddy et al., 1991). In-furrow insecticides and foliar-applied insecticides are commonly used to control tobacco thrips (Brandenburg, 2011; Chapin, 2010; Herbert, 2011). The heliothis complex may cause significant peanut canopy defoliation (Weiss, 2000). Control of heliothines may require a foliar-applied insecticide spray to protect plants from foliar damage that may result in yield loss (Brandenburg, 2011; Chapin, 2010; Herbert, 2011).
Early leaf spot, late leaf spot, and Sclerotinia blight may be prevalent when weather conditions and production practices are conducive for disease development (Middleton et al., 1994). These diseases are managed using crop rotation, resistant cultivars, fungicide sprays, or a combination of these practices (Phipps, 2011; Shew, 2011).
Peanut grown with adequate moisture often has excessive vine growth which can affect digging efficiency. The plant growth regulator prohexadione calcium may be applied to peanut to increase apical dominance of the peanut plant, ultimately increasing row visibility. Better defined rows at harvest enables growers to minimize pod loss at digging and ultimately increase field harvested yield (Beam et al., 2002; Culpepper et al., 1997; Mitchem et al., 1995). Excessive vine growth may also increase vine damage caused by equipment, resulting in increased incidence of Sclerotinia blight (Porter and Powell, 1978).
When applications are delayed and agrochemical remains in the tank for an extended period of time, there is the possibility that efficacy could be influenced. Xue et al. (2008) reported that permethrin remaining in ultra-low volume spray tanks for 4 months averaged 55.5% degradation of the product when analyzed by gas chromatography. In corn (Zea may L.), tank mixes of dimethenamid plus dicamba plus atrazine, isoxaflutole plus atrazine, and rimsulfuron plus S-metolachlor plus dicamba mixed one or more d prior to application resulted in reduced weed control (Stewart et al., 2009). Eure et al. (2011) reported changes in efficacy of pesticides and plant growth regulators applied to cotton (Gossypium hirsutum L.) although response was inconsistent, infrequent, and minor in scale of effect. Efficacy of ethephon applied as a tobacco (Nicotiana tabacum L.) ripening agent was reduced when prepared 3 or 7 d prior to application in 1 of 2 field studies (Eure et al., 2012). The effect of delayed application of spray solutions is poorly understood with respect to efficacy of fungicides, insecticides, inoculants, and plant growth regulators commonly used to manage peanut. Therefore, research was conducted to determine the effect of mixing interval on efficacy of insecticides and fungicides commonly applied to peanut, the inoculant Bradyrhizobium, and prohexadione calcium.
Materials and Methods
General Methodology
Field experiments were confoducted during 2009 and 2010 in North Carolina at the Peanut Belt Research Station near Lewiston-Woodville and the Upper Coastal Plain Refosearch Station near Rocky Mount. Soil at Lewiston-Woodville was a Norfolk loamy sand (fine-loamy, kaolinitic, thermic Typic Kandiudults). Soil at Rocky Mount was a Goldsboro sandy loam (fine-loamy, siliceous, subactive, thermic Aquic Paleudults).
Fungicides, insecticides, and prohexadione calcium were mixed individually the d of application or 3, 6, and 9 d prior to application, unless otherwise noted. Spray solutions were mixed in plastic spray bottles (3 L volume), sealed for storage in the dark at room temperature, and agitated every third d to bring agrochemicals back into solution. Unless otherwise stated, a municipal supply of water from Wake County, NC was used with a pH of 6.7, hardness of 31 ppm, and concentrations of boron, calcium, magnesium, and zinc of 0.04, 7.40, 0.0 and 0.06 ppm, respectively. On the d of application, agrochemicals were agitated thoroughly to ensure uniform distribution in spray solutions. Agrochemicals were applied using DG11002 nozzles (Tee Jet nozzles, Spraying Systems Co., Wheaton, IL 60189) calibrated to deliver 140 L/ha at 210 kPa.
The peanut cultivar ‘Phillips’ (Isleib et al., 2006) was planted in conventionally tilled, raised seed beds at a rate to establish 13 plants/m row in rows spaced 96 cm apart. Production and pest management practices other than specific treatments were held constant over the entire experiment to optimize peanut growth and development (Brandenburg, 2011; Jordan, 2011; Shew, 2011).
Data for all parameters were subjected to ANOVA using the PROC GLM procedure in SAS (SAS Institute Inc., Cary, NC 27513). Although present in all experiments, non-treated controls were not included in the statistical analysis to allow consideration of the factorial treatment arrangement of treatments. The treatment of agrochemicals applied the same d solutions were prepared is considered the most appropriate control in the experiments. However, data for the non-treated control are provided as a reflection of pest damage, population or peanut growth in the absence of pest control or management inputs. Means of significant main effects and interactions were separated using Fisher's Protected LSD test at p ≤ 0.05.
Bradyrhizobium Efficacy
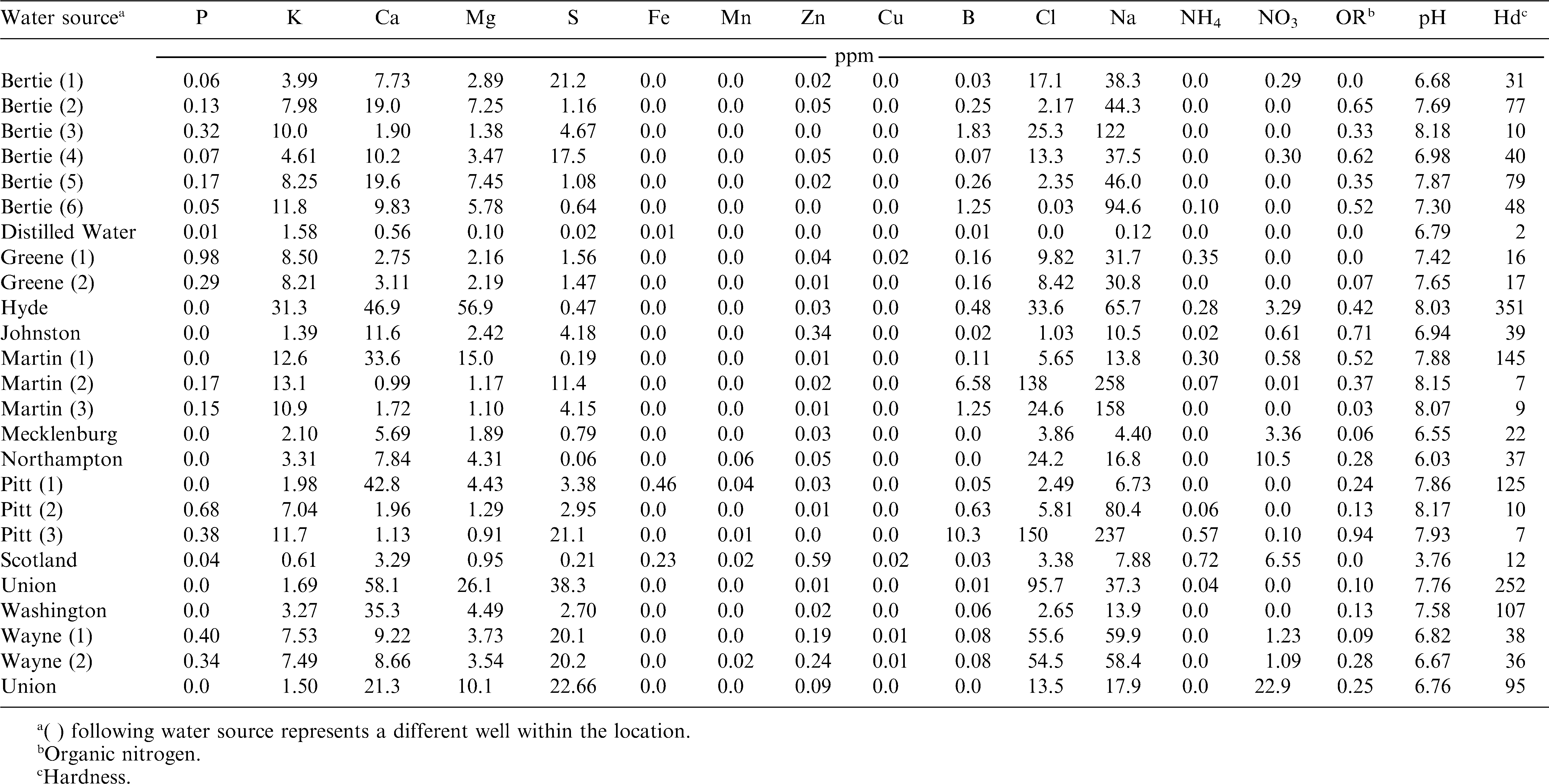
Efficacy of Bradyrhizobium (Optimize LIFT, EMD Crop BioScience, Milwaukee, WI 53209) was evaluated twice during 2011 in the greenhouse. Bradyrhizobium was prepared in 25 water sources (Table 1) immediately prior to planting or 4 and 8 d prior to planting. Water source pH, hardness, calcium, and chlorine concentration ranged from 3.76 to 8.18, 2 to 351 ppm, 0.56 to 58.10 ppm, and 0 to 150 ppm, respectively. Water sources were selected from locations throughout North Carolina to provide a wide range of water quality. Sterilized soil was placed in 15 cm diameter pots and a 14 cm long by 4 cm deep furrow was prepared. Each pot received 0.38 ml of 2.34% (v/v) Bradyrhizobium solution and three seeds of the peanut cultivar ‘Phillips’ were evenly spaced within each pot. Night and d temperatures were maintained at 20 and 30 C, respectively. A complete fertilizer without nitrogen was added 21 and 56 d after planting (DAP).
Plant height and the number of plants per pot were recorded 90 DAP. A Soil-Plant Analysis Development (SPAD) chlorophyll meter (Konica Minolta Sensing, Inc., Ramsey, NJ 07446) was used to determine leaf color from the upper one-third of the peanut canopy from 3 leaves per pot, 90 DAP. The SPAD chlorophyll meter value is derived based on the wavelength ranges of red and infrared light (Anonymous, 1989). Values are highly correlated with leaf chlorophyll content; higher values are associated with greater chlorophyll content (Kumagai et al., 2009; Leon et al., 2007; Reis et al., 2009). Chlorophyll content in leaves would be reflective of higher BNF as a result of great inoculant performance under conditions of this experiment. Additionally, dry weight of roots and shoots were determined 90 DAP. In the statistical analysis, water source and mixing interval were considered fixed effects and experiment and replication were considered random effects in the analysis.
Acephate Efficacy
Tobacco thrips control with acephate (Orthene 97, Valent Corp., Walnut Creek, CA 94596) at 1.1 kg ai/ha was evaluated in one field at Lewiston-Woodville during 2009 and two separate fields at this location during 2010 when applied 10 d after peanut emergence. Peanut was planted without in-furrow insecticide to promote tobacco thrips establishment. Plot size was two rows by 9 m.
Visible estimates of damage from tobacco thrips feeding were recorded 10 and 20 d after treatment (DAT) using a 0 to 5 ordinal scale, where 0 = no damage, 1 = noticeable feeding but no stunting, 2 = noticeable feeding and 25% stunting, 3 = feeding with blackened terminals and 50% stunting, 4 = severe feeding and 75% stunting, and 5 = severe feeding and 90% stunting (Carley et al., 2009). During 2010, five peanut leaves from five plants per plot were collected 5 and 10 DAT and gently placed in glass mason jars with 250 ml of water and 1 m of detergent (Palmolive Original, Colgate-Palmolive Company, New York, NY 10022) and sealed for transport to the laboratory. Following gentle agitation for 20 s, solutions were decanted onto a 250-µm mesh sieve (U.S. standard sieve, Fisher Scientific, Pittsburgh, PA 15275). Jars and lids were then rinsed thoroughly to remove any remaining thrips. Plants were thoroughly rinsed to separate thrips from plants. Thrips and small sediment were collected on the mesh screen and rinsed from the screen through a funnel into 25-ml vials using 70% isopropyl alcohol. Excess alcohol was decanted from the vials and remaining solution, thrips, and sediment were then poured onto a gridded petri dish and examined using a microscope (10X to 44X Stereoscope, Fisher Scientific, Pittsburgh, PA 15275). The number of immature and adult thrips was recorded for each sample. Additionally, canopy width of three plants per plot was recorded 10, 20, and 56 DAT. The experimental design was a randomized complete block with treatments replicated four times. Data were subjected to ANOVA and means separated using Fisher's Protected LSD at p ≤ 0.05.
Fungicide Efficacy
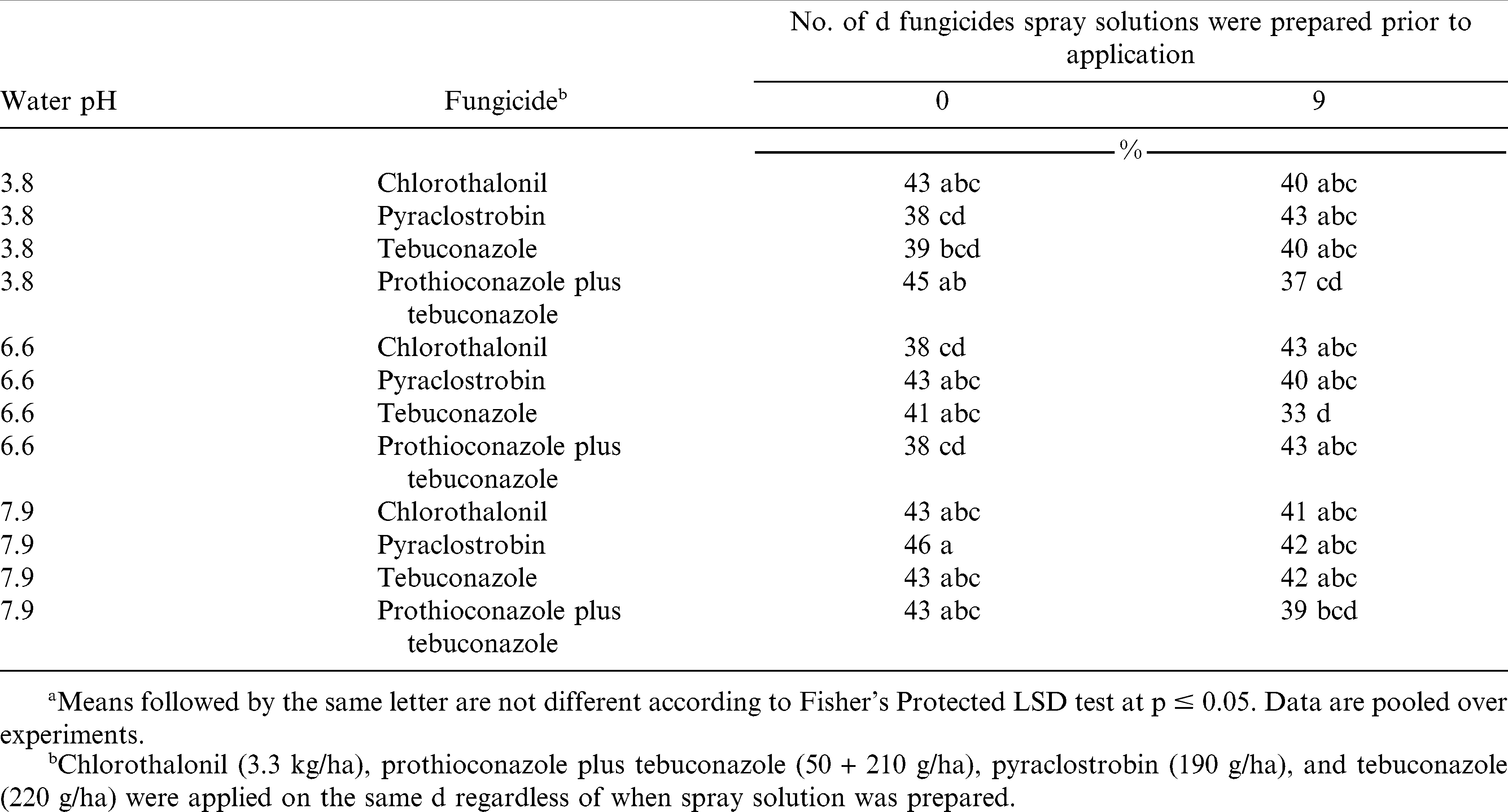
In one set of experiments during 2009 conducted in two separate fields, efficacy of chlorothalonil (Bravo WeatherStik fungicide, Syngenta Crop Protection Inc., Greensboro, NC 27419) at 3.3 kg ai/ha, prothioconazole plus tebuconazole (Provost fungicide, Bayer CropScience, Research Triangle Park, NC 27709) at 50 g ai/ha plus 210 g ai/ha, pyraclostrobin (Headline fungicide, BASF Corporation, Research Triangle Park, NC 27709) at 190 g ai/ha, and tebuconazole (Folicur fungicide, Bayer CropScience, Research Triangle Park, NC 27709) at 220 g ai/ha were applied at Lewiston-Woodville to determine impact of prior mixing interval on early and late leaf spot control. In a second set of experiments during 2010, fungicide solutions described previously were prepared the d of application and 9 d prior to application in water representing low, moderate, and high pH from Scotland, Bertie 1, and Martin 1 samples (Table 1).
In each experiment, fungicides were applied at the R-3 stage of peanut growth, based on North Carolina Cooperative Extension Service recommendations (Shew, 2011). Half of each plot received additional applications of chlorothalonil (3.3 kg/ha) and pryraclostrobin (190 g/ha) 14 and 28 d after the initial fungicide application, respectively. These fungicide solutions were prepared immediately prior to application. Peanut often receives 4 or 5 fungicide applications per year to control early and late leaf spot in North Carolina (Shew, 2011). The second and third applications applied to only half of the plot were included to determine if any minor effect on efficacy caused by initial treatments would be observed by the end of the growing season when additional fungicides were applied. Plot size was one row by 4 m with non-treated rows separating treated rows.
Visible estimates of percent canopy defoliation were recorded 1 and 3 wk before digging peanut, using a 0 to 100 scale where 0 = no canopy defoliation and 100 = complete canopy defoliation. Peanut was not harvested. The experimental design was a split-plot with the number of total fungicides for the entire season serving as the whole plot unit. Combinations of fungicide and mixing interval served as the split-plot unit (one application with fungicides including various mixing intervals vs. three fungicide applications including the initial spray with various mixing intervals and the second and third sprays). Data for canopy defoliation were subjected to ANOVA for a two (trial) by four (fungicide) by four (mixing interval) factorial treatment arrangement in the experiment from 2009. In the experiment from 2010, data for canopy defoliation were subjected to ANOVA for a two (trial) by four (fungicide) by two (mixing interval) by three (water source) treatment arrangement. Means of significant main effects and interactions were separated using Fisher's Protected LSD test at p ≤ 0.05.
Efficacy of boscalid (Endura fungicide, BASF Corporation, Research Triangle Park, NC 27709) at 12 g ai/ha and fluazinam (Omega 500 fungicide, Syngenta Crop Protection Inc., Greensboro, NC 27419) at 145 g ai/ha in controlling Sclerotinia blight was evaluated in fields with a history of Sclerotinia blight incidence during 2009 at Lewiston-Woodville in two separate fields and during 2010 in one field at Lewiston-Woodville and one field at Rocky Mount. Visible estimates of plant condition were recorded 1 wk prior to harvest in 2009, using a 0 to 100 scale where 0 = healthy canopy and 100 = canopy completely damaged by disease. The experimental design was a randomized complete block with four replications.
Prohexadione Calcium Efficacy
The experiment was conducted during 2009 at Lewiston-Woodville in one field and during 2010 at Rocky Mount in two separate fields. Plot size was two rows by 9 m. Prohexadione calcium (Apogee plant growth regulator, BASF Corporation, Research Triangle Park, NC 27709) at 680 g ai/ha was applied when 50% of vines from adjacent rows were touching, based on Cooperative Extension Service recommendations (Jordan, 2011). A second application of prohexadione calcium was made to half of each plot 2 wk after the initial application using spray solution prepared the d of application. Two applications of prohexadione calcium are generally needed to maintain row visibility through harvest (Jordan et al., 2008).
Visible estimates of peanut vine architecture were recorded 3 and 6 wks after treatment (WAT) using a scale of 1 to 10 where 1 = flat canopy with vines completely overlapping in the row middles and 10 = triangular- shaped canopy with no vines from adjacent rows touching in the row middles (Mitchem et al., 1995). Main stem height of three plants per plot was recorded 3 and 6 WAT. The experimental design was a spit-plot with four replications. The number of total prohexadione applications for the entire season served as the whole plot unit. Mixing interval served as the split-plot unit with one application with prohexadione calcium including mixing intervals compared to two prohexadione calcium applications including the initial spray with various mixing intervals and the second spray.
Results and Discussion
Bradyrhizobium Efficacy
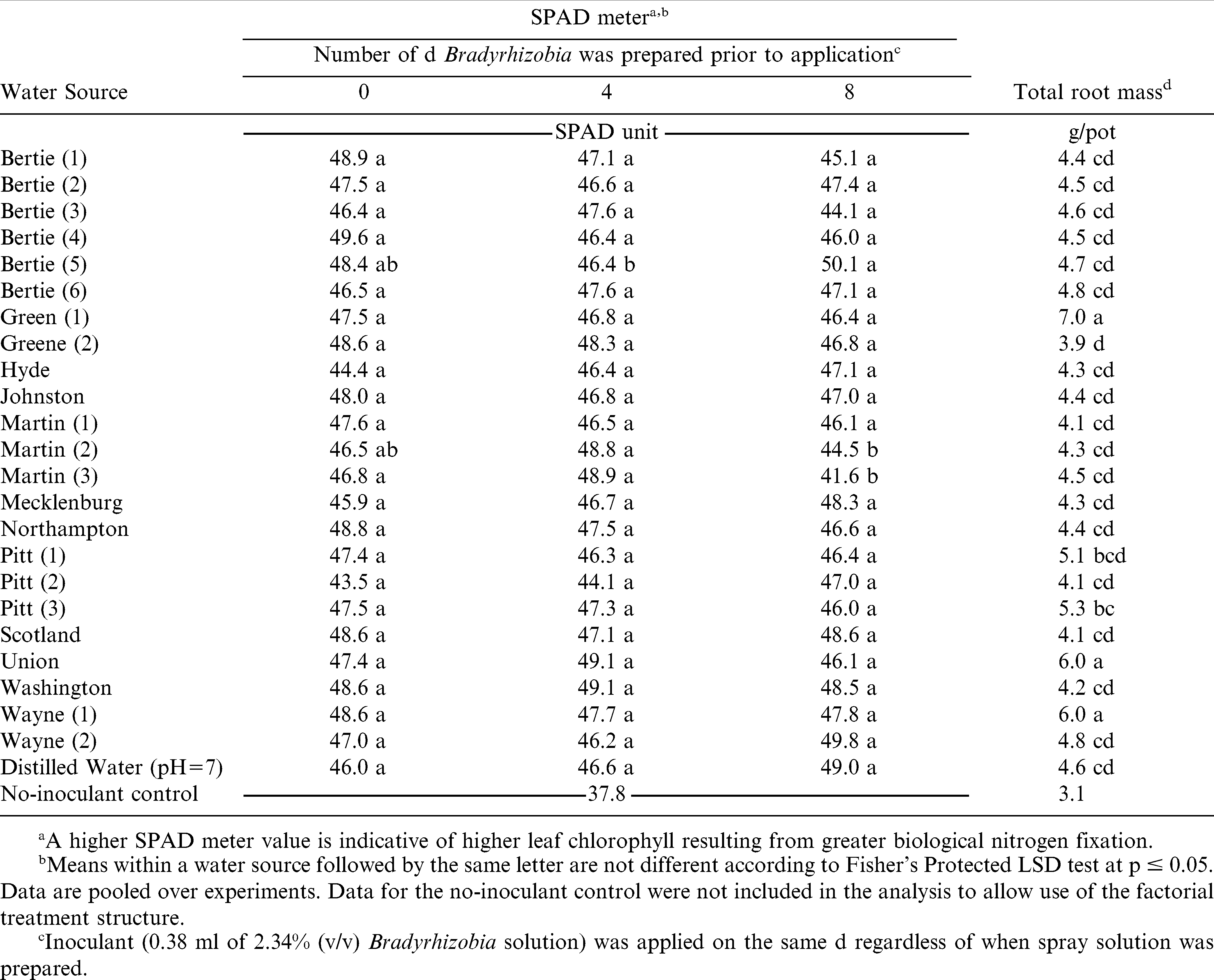
Plant stand, plant height, and total shoot mass were not influenced by the interaction of water source by mixing interval or the main effect of water source or mixing interval (data not shown). However, SPAD meter values were influenced by the interaction of water source and mixing interval; total root mass was influenced by the main effect of water source.
SPAD meter values ranged from 41.6 to 50.1 regardless of water source or the number of d inoculant solution was prepared prior to application (Table 2). The SPAD value for non-inoculated peanut was 37.8. When comparing within a water source, differences in SPAD values were noted for 3 of 25 water samples [Bertie (5), Martin (2), and Martin (3)] when inoculant remained in solution for 4 or 8 d prior to application. Inoculant solution prepared 8 d prior to use in water collected from Martin (3) resulted in an 11% reduction in SPAD value as compared to inoculant prepared the d of application. Inoculant prepared in Martin (2) the d of application or 4 d prior to application resulted in a similar SPAD value but solution prepared 8 d prior to application was 9% less than solution prepared 4 d prior to application. The SPAD values were similar for inoculant solution prepared using water source Bertie (5) the d of application and 4 or 8 d prior to application. However, the SPAD value for inoculant prepared in Bertie (5) water 8 d prior to application was 3.7 units greater than solution prepared 4 d prior to application.
In inoculant treatments, total root mass ranged from 3.9 to 6.0 g/pot while root mass of non-inoculated peanut was 3.1 g/pot (Table 2). Inoculant solution prepared in water source Green (1), Union, and Wayne (1) resulted in a total root mass 1.4 to 2.4 g/pot higher than inoculant solution prepared in distilled water. The difference in root mass of these samples could not be explained based on analyses of water sources.
When comparing inoculant solutions prepared in 25 water sources the d of application or 4 and 8 d prior to application over all parameters; Bertie (5), Martin (2), and Martin (3) showed sporadic changes in efficacy due to water source or number of d prepared prior to application (Table 2). Review of the water analysis report did not suggest any nutrient or pH trends that explain variations in SPAD values, height, and root dry weight among treatments.
Acephate Efficacy
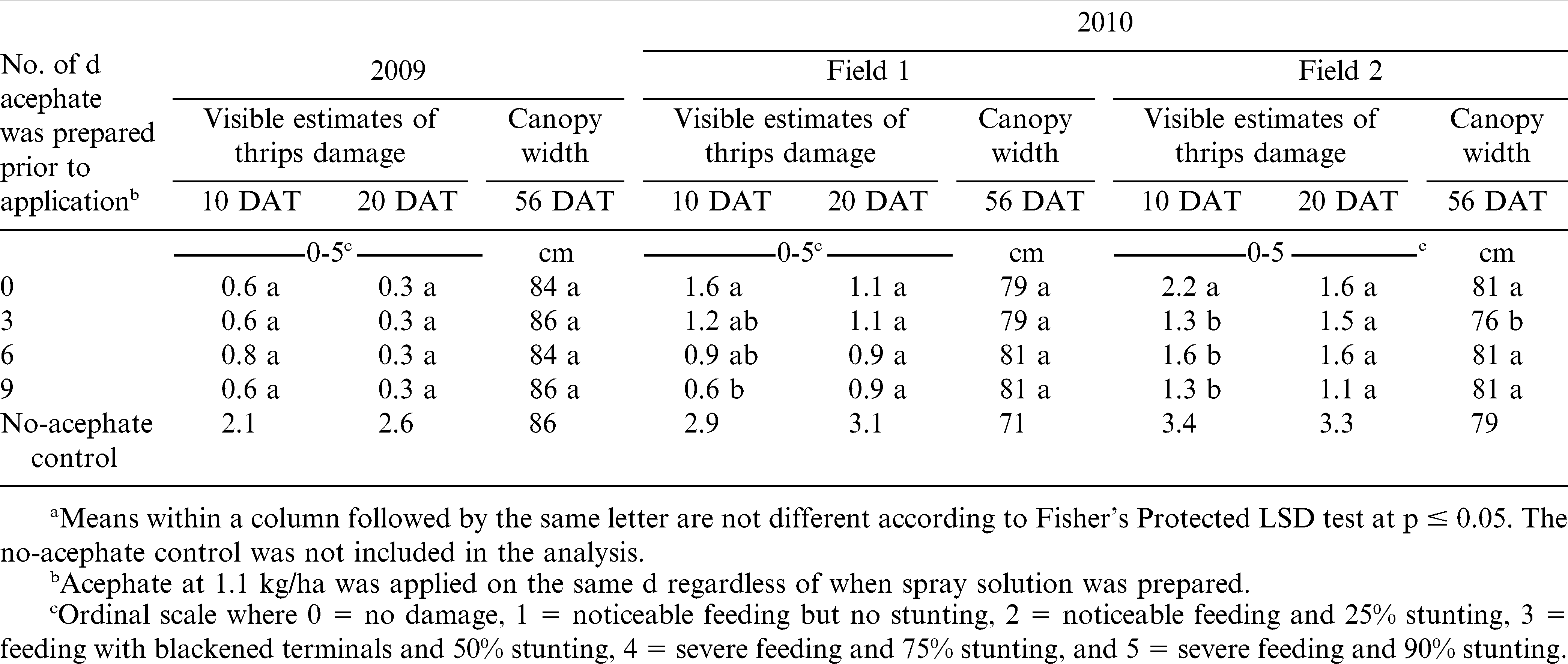
The interaction of experiment by mixing interval was significant for immature thrips populations 5 DAT (Table 3, p ≤ 0.0001) but not for adult thrips populations (data not shown, p = 0.3611). The interaction of experiment by mixing interval did not influence immature and adult thrips populations 10 DAT (p = 0.4710), visible estimates of tobacco thrips damage 10 (p = 0.3318) and 20 DAT (p = 0.7757), and canopy width 10 (p = 0.4439), 20 (p = 0.5116), and 56 DAT (p = 0.4585). However, the main effect of mixing interval for visible estimates of tobacco thrips damage 10 DAT (p ≤ 0.0001) and the main effect of experiment for immature and adult thrips populations 10 DAT (p ≤ 0.0001), for visible estimates of tobacco thrips damage 20 DAT (p = 0.0238) and canopy width 56 DAT (p = 0.0359) were significant (Table 4).
Thrips damage was considerably higher and a relatively higher population of thrips was noted in non-treated controls compared with acephate-treated peanut (Table 3 and 4). When analyzed by experiment, the number of immature thrips per five leaves 5 DAT was less in field 2 when acephate was mixed 3 d prior to application as compared to acephate prepared the d of application, or 6 and 9 d prior to application (Table 3).
In 2009, mixing interval did not affect visual estimates of thrips damage 10 or 20 DAT or canopy width 56 DAT (Table 4). In 2010, visual estimates of thrips damage 10 DAT was lower when acephate solutions were prepared 9 d prior to application (Field 1) or 3, 6, or 9 d prior to application (Field 2) compared with application the d of mixing. By 20 DAT, no difference in thrips damage was noted irrespective of mixing interval. Although canopy width 56 DAT did not differ in Field 1, mixing acephate 3 d prior to application resulted in a narrower canopy width compared with other mixing intervals in field 2 in 2010.
Fungicide Efficacy
In the experiment evaluating fungicide and mixing interval, the interaction of experiment by mixing interval by fungicide by number of fungicide sprays was not significant for peanut defoliation 1 wk before harvest. However, the main effect of mixing interval and number of fungicide sprays was significant. The primary goal of this research was to determine if defoliation, an indicator of fungicide performance, was affected by mixing interval. These data suggest that mixing fungicides at various intervals 9 d prior to application will not reduce efficacy if fungicide solutions can be brought back into solution (Table 5). When pooled over fungicides, a single application resulted in 74% defoliation at 1 wk before harvest while three applications resulted in 24% defoliation (data not shown). Five fungicide sprays are generally made to control early and late leaf spot in North Carolina (Shew, 2011), and these data suggest that significant defoliation can occur with only three fungicide sprays.
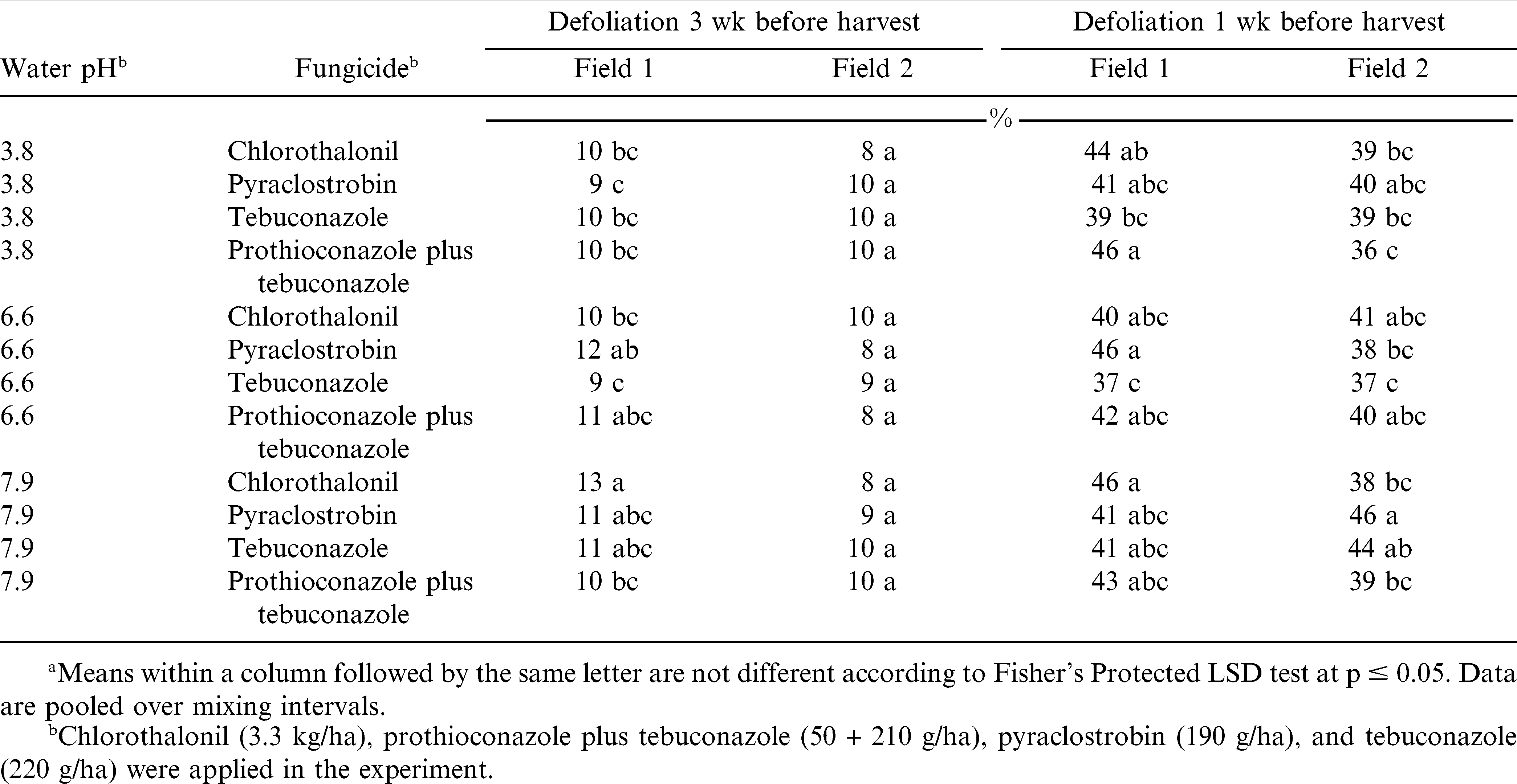
In the experiment comparing mixing intervals, fungicides, and water sources, defoliation 3 wk and 1 wk before harvest was not influenced by the interaction of experiment by mixing interval by water source by fungicide by number of fungicide sprays. However, 1 wk prior to harvest the interaction of experiment by water source by fungicide was significant (p = 0.0111; cv = 32.9%; sd = 2.1) (Table 6) as well as the interaction of experiment by water source by fungicide (p = 0.0188; cv = 22.0%; sd = 5.3) (Table 7). The number of fungicide sprays influenced canopy defoliation 3 wk and 1 wk before harvest. Regardless of water source, efficacy of prothioconazole plus tebuconazole was not influenced (Table 6). However, efficacy of chlorothalonil, pyraclostrobin, and tebuconazole was influenced when spray solutions were prepared using different water sources (Table 6). Canopy defoliation 3 wk before harvest was greater when chlorothalonil was prepared using high pH water (Field 1) when compared to chlorothalonil prepared using low and moderate pH waters. Similar results using chlorothalonil were not observed 1 wk before harvest. Pyraclostrobin prepared using low pH water (Field 1) resulted in less canopy defoliation when compared to pyraclostrobin prepared using moderate pH water 3 wk before harvest. Canopy defoliation when pyraclostrobin and tebuconazole were prepared using moderate pH water was 8 and 7% less respectively (Field 2), when compared to pyraclostrobin or tebuconazole prepared using high pH water.
When considering canopy defoliation 1 wk before harvest, neither chlorothalonil or pyraclostrobin alone were influenced by mixing interval regardless of water source (Table 7). Canopy defoliation 1 wk before harvest was 8% greater when prothioconazole plus tebuconazole was prepared using low pH water and applied the d of application, compared to prothioconazole plus tebuconazole prepared using low pH water and left in spray solution for 9 d. Canopy defoliation 1 wk before harvest using tebuconazole prepared in moderate pH water and left in solution for 9 d resulted in 8% less defoliation compared to tebuconazole prepared in moderate pH water and applied the d of mixing. These data suggest that preparing fungicide solutions in various pH water sources and leaving them in solution for 9 d prior to application will not reduce the efficacy of the fungicides if fungicide can be brought back into solution.
Applying a single fungicide application resulted in 15 and 55% defoliation 3 wk and 1 wk before harvest, respectively (data not shown). Three fungicide applications resulted in 5 and 27% defoliation 3 wk and 1 wk before harvest, respectively. These data reflect differences noted in the previous experiment.
The interaction of experiment and mixing interval and the main effect of mixing interval were not significant for plant condition rating associated with Sclerotinia blight (data not shown). However, the main effect of experiment was significant for plant condition rating. At Lewiston-Woodville in 2009, plant condition ratings were equivalent when comparing non-treated controls to treatments of boscalid or fluazinam (21 to 31%, data not shown). During 2010, plant condition rating following fluazinam ranged from 28% (Rocky Mount) to 84% (Lewiston-Woodville) in absence of fluazinam compared with 14% (Rocky Mount) and 42% (Lewiston-Woodville) when fluazinam was applied. In 2010, plant condition rating following boscalid was 15 to 37% compared with 40 to 83% without boscalid (data not shown). In 2010, boscalid increased the plant condition rating from 25 to 46% and fluazinam reduced plant condition rating 14 to 42%. While plant canopy expressed disease in 2009, differences between treatments and non-treated controls were not evident, thus these data may not be representative of the level of disease control boscalid and fluazinam may offer.
Prohexadione Calcium Performance
Canopy height and row visibility 8 WAT were not influenced by the interaction of experiment by mixing interval by number of sprays (data not shown). The main effect of mixing interval was not significant for canopy height or row visibility. However, canopy height and row visibility were influenced by the main effects of number of sprays and experiment. Greater main stem height and lower row visibility were noted in non-treated controls when compared with two sequential application of prohexadione calcium was applied twice (data not shown).
When comparing results for acephate, fungicides, and prohexadione calcium over all pests or peanut growth parameters, performance of these agrochemicals was not affected adversely when agrochemicals remained in spray solution up to 9 d. Over all parameters evaluated in the Bradyrhizobium study, SPAD value was negatively influenced when using inoculant solution prepared in only one of 25 water sources 8 d prior to application. Water quality did influence control of canopy defoliation by chlorothalonil, prothioconazole plus tebuconazole, pyraclostrobin, or tebuconazole. However, these differences would likely be difficult to observe at the farm level. Although leaf spot fungicides and water quality were evaluated, results may be different when using spray water with different pH values or cation concentrations. Spray water quality has been shown to reduce efficacy of herbicides (Buhler and Burnside, 1983; Nalewaja and Matysiak, 1993; Thelen et al. 1995). Insecticides, Sclerotinia blight, and plant growth regulator experiments were conducted using a single water source at pH 6.7 with relatively low water hardness. Leaving agrochemicals in spray tanks in water with other characteristics might lead to different responses in performance than observed in these experiments. However, other experiments evaluating agrochemical efficacy when left in spray solution for extended periods of time have not observed a product failure (Eure et al., 2011; Eure et al., 2012; Stewart et al., 2009).
Acknowledgements
This research was supported in part by funds administered through the North Carolina Peanut Growers Association, Phillip Morris U.S.A., R. J. Reynolds, and Monsanto Company. Appreciation is expressed to staff at the Peanut Belt Research Station and the Upper Coastal Plain Research Station for technical assistance.
Literature Cited
Anonymous 1989 Chlorophyll meter SPAD-502. Technical Bulletin Minolta , Osaka, Japan 22 pp.
Anonymous 2010 Optimize, Lift and LCO Promoter Technology Label EMD Crop Biosciences, Milwaukee, WI. Pub. 1760-0311 .
Beam J.B Jordan D.L York A.C Isleib T.G Bailey J.E McKemie T.E Spears J.F and Johnson P.D 2002 Influence of prohexadione calcium on pod yield and pod loss of peanut Agron. J. 94 : 331 – 336 .
Brandenburg R.L 2011 Peanut insect and mite management . pp. 86 – 104 In 2011 Peanut Information North Carolina Cooperative Extension Service Pub AG-331 (Revised) , Raleigh, NC 156 pp.
Buhler D.D and Burnside O.C 1983 Effect of water quality, carrier volume, and acid on glyphosate phytoxicity Weed Sci. 31 : 163 – 169 .
Carley D.S Jordan D.L Brandenburg R.L and Dharmasri L.C 2009 Factors influencing response of Virginia market type peanut to paraquat under weed-free conditions Peanut Sci. 36 : 180 – 189 .
Chapin J.W 2010 Insects . pp. 4 – 40 In Peanut Money-Maker Production Guide-2010: Clemson Extension Circular 588 , Blackville, SC 62 pp.
Culpepper A.S Jordan D.L Batts R.B and York A.C 1997 Peanut response to prohexadione calcium as affected by cultivar and digging date Peanut Sci. 24 : 85 – 89 .
Eure P.M Jordan D.L Bacheler J.S York A.C Fisher L.R and Wells R 2011 Performance of cotton agrochemicals when spray solution application is delayed J. Cotton Sci. 15 : 215 – 223.
Eure P.M Fisher L.R Stewart A.M and Jordan D.L 2012 Evaluation of growth regulators and ripening agent used in flue-cured tobacco when spray solution application is delayed Tobacco Sci. 49 : 21 – 24 .
Hashem F.M Saleh S.A van Berkum P and Voll M 1997 Survival of Bradyrhizobium sp. (Arachis) on fungicide-treated peanut seed in relationship to plant growth and yield World J. of Microbio. and Biotech. 13 : 335 – 340 .
Herbert D.A 2011 Insects: peanuts . pp. 4.97 – 4.107 In Pest Management Guide-2011 Field Crops Virginia Cooperative Extension Service Pub 456-016 , Blacksburg, VA 7-1 pp.
Isleib T.G Rice P.W Mozingo II R.W Copeland S.C Graeber J.B Pattee H.E Sanders T.H and Coker D.L 2006 Registration of ‘Phillips’ peanut Crop Sci. 46 : 2308 – 2309 doi:10.2135/cropsci2005.12.0491 .
Jordan D.L 2011 Peanut production practices . pp. 25 – 54 In 2011 Peanut Information North Carolina Cooperative Extension Service Pub AG-331 (Revised) , Raleigh, NC 156 pp.
Jordan D.L Nuti R.C Beam J.B Lancaster S.H Lanier J.E Lassiter B.R and Johnson P.D 2008 Peanut (Arachis hypogaea L.) cultivar response to prohexadione calcium Peanut Sci. 35 : 101 – 107 .
Kumagai K Takuya A and Kubota F 2009 Correlation of chlorophyll meter readings with gas exchange and chlorophyll fluorescence in flag leaves of rice (Oryza sativa L.) plants Plant Prod. Sci. 12 : 50 – 53 .
Leon A.P Vina S.Z Frezza D Chaves A and Chiesa A Estimation of chlorophyll contents by correlation between SPAD-502 meter and chroma meter in butterhead lettuce Comm. In Soil Sci. and Plant Analysis. 38 : 2877 – 2885 .
Middleton K.J Pande S Sharma S.B and Smith D.H 1994 Diseases . pp. 336 – 394 In The Groundnut Crop Chapman and Hall , London, UK 756 pp.
Mitchem W.E York A.C and Batts R.B 1995 Evaluation of chlorimuron as a growth regulator for peanut Peanut Sci. 22 : 62 – 66 .
Nalewaja J.D and Matysiak R 1993 Spray carrier salts affect herbicide toxicity to kochia (Kochia scoparia) Weed Tech. 7 : 154 – 158 .
Phipps P.M 2011 Diseases and nematodes: peanuts . pp. 3.43 – 3.50 In Pest Management Guide-2011 Field Crops Virginia Cooperative Extension Service Pub 456-016 , Blacksburg, VA 7-1 pp.
Porter D.M and Powell N.L 1978 Sclerotinia blight development in peanut vines injured by tractor tires Peanut Sci. 5 : 87 – 90 .
Reddy D.V.R Wightman J.A and Beshear R.J 1991 Bud necrosis: a disease of groundnut caused by tomato spotted wilt virus Information Bulletin 31, International Crops Research Institute for the Semi-Arid Tropics , Patancheru .
Reis A.R Favarin J.L Malavolta E Junior J.L and Moraes M.F 2009 Photosynthesis, chlorophylls, and SPAD readings in coffee leaves in relation to nitrogen supply Comm. In Soil Sci. and Plant Analysis. 40 : 1512 – 1528 .
Rubens Jose C 2009 Nitrogen fixation with the soybean crop in Brazil: Compatibility between seed treatment with fungicides and bradyrhizobial inoculants Symbiosis. 48 : 154 – 163 .
Schiffman J and Alper T 1968 Effects of Rhizobium – inoculum placement on peanut inoculation Exp. Agric. 4 : 203 – 208 .
Shew B.B 2011 Peanut disease management . pp. 103 – 119 In 2011 Peanut Information North Carolina Cooperative Extension Service Pub AG-331 (Revised) , Raleigh, NC 156 pp.
Shimshi D Schiffman J Kost T Bielorai H and Alper T 1967 Effects of soil moisture regime on nodulation of inoculated peanuts Agron. J. 59 : 397 – 400 .
Stewart C.L Nurse R.E Cowbrough M and Sikkema P.H 2009 How long can a herbicide remain in the spray tank without losing efficacy ? Crop Prot. 28 : 1086 – 1096 .
Thelen K.D Jackson E.P and Penner D 1995 The basis for the hard-water antagonism of glyphosate activity Weed Sci. 43 : 541 – 548 .
Walker M.E Minton N.A and Dowler C.C 1976 Effects of herbicides, a nematicide and Rhizobium inoculant on yield, chemical composition and nodulation of Starr peanuts (Arachis hypogaea L.) Peanut Sci. 3 : 49 – 51 .
Wightman J.A and Rao G.V.R 1994 Groundnut pests . pp. 395 – 469 In The Groundnut Crop, eds. J. Smartt Chapman and Hall , London, UK 756 pp.
Weiss E.A 2000 Groundnut . pp. 53 – 93 In Oilseed Crops Blackwell Science , Oxford, UK, 411 pp.
Xue R Qualls W.A Zhong H and Brock C.L 2008 Permethrin decomposition after four months storage in the spray truck tanks during mosquito off season J. of the Am. Mosquito Control Assoc. 24 : 127 – 129 .
Notes
- Graduate Research Assistant, Professor, Associate Professor, and William Neal Reynolds Professor Emeritus; Department of Crop Science, North Carolina State University, Box 7620, Raleigh, NC 27695-7620 * Corresponding author: david_jordan@ncsu.edu.
Author Affiliations