Introduction
Cultivated peanut (Arachis hypogaea L.) is an important oil and food crop, grown on approximately 42 million acres worldwide. It is the third major oilseed of the world behind soybean and cotton. China, India and the United States have been the leading producers for over 25 years and grow about 70% of the world crop (American Peanut Council; www.peanutsusa.com). Peanut production in the U.S. is limited to three geographical regions: the Southeast region (Alabama, Florida, Georgia and Mississippi), the VC region (North Carolina, South Carolina and Virginia) and the Southwest region (Oklahoma, New Mexico and Texas). Each growing region differs from the others in climate and disease pressure and thus, breeding programs tend to develop peanut varieties which are specifically adapted and have superior performance in a particular growing region.
In the Southwestern U.S., the oil content of peanuts significantly affects flavor and shelf-life. Peanut seeds normally contain between 45–51% oil. Two fatty acids, oleic and linoleic acid, comprise over 80% of the oil content in peanut. In the fatty acid systhesis pathway, oleic acid is a precursor to linoleic acid. The enzyme responsible for the conversion of oleate to linoleate is oleoyl-PC desaturase, or Δ12 fatty acid desaturase. The fatty acid composition in most cultivated peanuts is 36–70% oleic and 15–43% linoleic acid (Norden et al., 1987). In general, saturated fatty acids are less susceptible to oxidative degradation than their less saturated counterparts. Thus, a high oleic to linoleic (O/L) acid ratio (≥ 10∶1) in peanut results in an increased shelf life (up to 10 times) and improved flavor when compared to a normal O/L ratio (∼1.5∶1). Other benefits of the high oleate trait have also been determined. Oils that have high oleic acid content and food products containing these oils have been shown to be nutritionally beneficial. Oleic acid has been shown to be associated with a reduction in blood pressure (Teres et al., 2008) and LDL cholesterol levels (O'Byrne et al., 1997). High-oleic peanuts have health benefits over conventional peanuts because the linoleic (polyunsaturated fat) and palmitic (saturated fat) fatty acids have been naturally replaced by the healthier oleic fatty acid (monounsaturated fat). High oleic peanuts and peanut oil high in oleic acid have also been shown to have a beneficial effect in type II diabetes and ultimately reverse the negative effects of inflammatory cytokines observed in obesity and non insulin dependent diabetes mellitus (Vassiliou et al., 2009).
The peanut industry in the Southwestern U.S. currently demands that peanuts be high oleic in nature and contracts are not offered to producers unless their product meets that requirement. Peanut genotypes possessing 75–80% oleic acid have been discovered and subsequently varieties containing the high oleate trait have been developed and released. Given the benefits of high oleic acid composition and the demands of the peanut industry, especially in the Southwestern U.S., breeding programs have focused on the rapid introgression of this trait into superior lines adapted for their growing region.
Since the discovery of the high O/L spontaneous mutant F435 (Norden et al., 1987), much work has been done to determine the inheritance of the high oleate trait in the different peanut market types (Chu et al., 2007, 2009; Jung et al., 2000a, 200b; Isleib et al., 1996; Lopez et al.,2001). Reports have determined that the fatty acid composition of peanut is quantitatively inherited and investigations proved that two loci, Ol1 and Ol2 , control the high O/L ratio in runner and Virginia market types (Moore and Knaft, 1989; Isleib et al., 2006b). Lopez et al. (2001) confirmed that these loci also control the high oleate trait in Spanish-type peanuts.
The high oleate trait in peanut has been shown to originate from two recessive loss-of-function mutations (ahfad2A and ahfad2B) in the genes that encode the Δ12 desaturase (Chu et al., 2007). The wild-type and dominant genes which govern Δ12 desaturase activity (ahFAD2A and ahFAD2B) are 99% homologous in nucleotide sequence and are homeologs as ahFAD2A belongs to the A subgenome and ahFAD2B to the B subgenome (Jung et al, 2000a). A close examination of properly generated F2 populations has shown that normal to high oleic segregation ratios are either 3∶1 or 15∶1 (normal∶high), depending upon the genotype of the normal oleate varieties used in the crosses (ol1ol1Ol1Ol1 or Ol1Ol1Ol1Ol1 , respectively). Those results indicated that only one expressed gene encoding a functional enzyme (Ol1 ) is necessary for a normal oleate phenotype. Isleib et al. (2006b) determined that the high oleate trait is not completely recessive, but instead that a dosage effect exists and the normal oleate exhibits incomplete dominance, allowing heterozygotes to be identified with confidence. In the naturally occurring mutant F435, the high O/L ratio is a result of greatly reduced transcription from the ahFAD2B gene along with a point mutation (G-to-A transition) in the ahFAD2A gene which resulted in an amino acid substitution (D150N) at position 448 and thus a non-functional desaturase (Jung et al., 2000 a,b). The genotypes of two other high O/L mutants (Flavorunner 458 and M2-225) generated by chemical mutagenesis have been dissected and it was determined that their high oleate phenotypes were the result of the 448G>A transition in ahFAD2A and a miniature-inverted-repeat-transposable element (MITE) insertion in ahFAD2B (Patel et al., 2004).
A better understanding of high oleate genetics has led to the development of several molecular marker systems which can be used to genotype F2 segregating populations and germplasm collections. Chu et al. (2007) designed cleaved amplified polymorphic sequence (CAPS) markers for the ahFAD2A gene and used them to screen the peanut U.S. mini-core collection and 27 accessions of Arachis duranensis for the 448G>A mutation. Later, a CAPS marker was also designed which would detect the mutation in the ahFAD2B gene (Chu et al., 2009), which provided a set of user-friendly high O/L molecular markers for use in marker assisted selection. Barkley et al. (2010, 2011) recently developed a real-time PCR genotyping assay using probes to detect an indel (insertion/deletion) in ahFAD2B which allows for rapid high through-put screening of segregating populations and is adaptable for seed or leaf tissue. Finally, Chen et al. (2010) have developed an allele-specific PCR assay which detects FAD2 alleles in the A and B genomes and thus completely genotypes any peanut with regards to the high O/L trait.
Although molecular markers can now define any genotype with regards to the high oleate trait, oil analysis should be done to confirm those results and determine the exact O/L ratio. Ideally, as with the genotype, breeders want to determine O/L ratios in early generations of selection (i.e. F2). Traditionally, oil composition of peanut has been determined by gas chromatography (GC) analysis (Phillips and Singleton, 1978; Singleton and Pattee, 1987) which, until recently, required a fairly large sample and was completely destructive, ruling out subsequent germination of tested seed. Using the traditional technique, oil composition of individual peanut seed can now be determined by GC analysis. Tillman et al. (2006) used near-infrared reflectance (NIR) spectroscopy to predict oleic and linoleic acid content of single peanut seed. NIR offers the advantage of being completely non-destructive, but lacks the accuracy that GC provides because it cannot determine exact fatty acid concentration but only classify peanut seed as either “high oleic” or “not high oleic”. We recently developed an oil extraction procedure and capillary electrophoresis (CE) protocol that enables one to extract sufficient quantities of oil from approximately 0.10 mg of peanut seed taken from the end distal to the embryo and determine the O/L ratio, ensuring subsequent seed germination and preservation of early generation breeding material (Bannore et al., 2008). In this study, we compare the CE and GC techniques relative to determining whether or not individual peanut seed have a high O/L ratio.
Materials and Methods
Plant Material
Eight genotypes of known oil composition (i.e. high oleate or normal) covering three peanut market- types were tested. Nine to ten random seed from each genotype were chosen for the CE/GC comparisons. Runner genotypes used included TamrunOL07 (high O/L, Baring et al., 2006), TX994313 (high O/L, USDA-ARS, Stillwater, OK) and Okrun (normal, Banks et al., 1989). Spanish genotypes used were OLin (high O/L, Simpson et al., 2003), TX996612 (high O/L, USDA-ARS, Stillwater, OK) and Tamspan 90 (normal, Smith et al., ). Virginia genotypes tested were Brantley (high O/L, Isleib et al., 2006a) and Jupiter (normal, Oklahoma State University, Ag. Exp. Station, 2000). No high oleate valencia-type varieties or breeding lines were available, so that market-type was omitted from these experiments.
Reagents
Saturated free fatty acid (FFAs) standards such as stearic acid (C18:0), palmitic acid (C16:0) and nonadecanoic acid (C19:0) and unsaturated FFAs such as oleic acid (C18:1), linoleic acid (C18:2) and linolenic acid (C18:3), adenosine 5′-monophosphate (AMP) monohydrate from yeast, tris (hydroxymethyl) aminomethane (Tris), α-cyclodextrin (α-CD) was obtained from Sigma (St. Louis, MO, USA). Dioxane, HCl, diethyl ether and anhydrous sodium sulphate were from Fischer Scientific (Fairlawn, NJ, USA) and N-methyformamide (NMF), was from Aldrich (St. Louis, MO, USA). Hexane was obtained from EM Science (Cherry Hill, NJ, USA) and KOH was from Mallinckrodt, Inc. (Paris, Kentucky, USA). Ethanol and methanol were purchased from AAPER Alcohol and Chemical Co. (Shelbyville, Kentucky, USA).
Extraction of Oil from Individual Peanut Seed
Extraction of oil from peanut seed was performed as previously reported (Bannore et al., 2008). The seeds used for the determination of FFAs were sound and mature. A small portion (≤ 0.10 mg) of the seed was cut from the distal end (away from the embryo) and the testa was removed from that portion. The sample was then weighed and ground to paste in a mortar-pestle in a sufficient volume of hexane. The slurry was transferred into a vial. The remaining paste was scraped and washed into the vial. The mortar was washed 2-3 times with hexane as required for complete transfer of the contents. The vial was vortexed for approximately one min and centrifuged for about 20-25 min at 10,000 rpm. The supernatant layer was collected and the hexane was evaporated using speed vacuum. The peanut oil left behind was further used for extraction of FFAs.
The isolation of FFAs from the total peanut oil was accomplished using the method of Dermaux et al. (1999) with some modifications. To 20-25 mg of oil, 0.55 mL of 1 M potassium hydroxide in 95% ethanol solution was added and subjected to overnight reflux in a heat block at 98 °C. Thereafter, the mixture was cooled to room temperature, and then transferred to a separatory funnel by rinsing the reflux vial with 1.5 mL water. The non-saponifiable matter was extracted using 1.5 mL of diethyl ether. The aqueous layer containing fatty acid salts was separated from the organic layer and then acidified to pH 2.0 with 1 M HCl. The FFAs formed were extracted with 6 × 1 mL diethyl ether. All organic layers were collected, washed with 1 mL of water and dried over anhydrous sodium sulphate. The solvent was evaporated at room temperature in the fume hood overnight. The extracted FFAs were dissolved in 0.5 mL of NMF-dioxane (1∶1, v/v) mixture. This was done by first dissolving the FFAs in dioxane followed by addition of NMF and then vortexing for 10-15 s. The sample was divided in-half, with one portion being used directly for CE analysis and the other modified for GC analysis.
Capillary Electrophoresis Analysis
The CE analysis of all standards and peanut samples was performed on a P/ACE MDQ (Beckman Instruments, Inc., Fullerton, CA, USA) equipped with a photodiode array detector and a 0-30 kV high-voltage power supply. The data were collected on an IBM personal computer configured with P/ACE MDQ 32 Karat software version 8.0. The capillary columns used for separation were untreated fused-silica capillaries with 50 µm I.D. and 363–359 µm O.D from Polymicro Technologies (Phoenix, AZ, USA). The total and effective lengths were 60.2 cm and 50 cm, respectively. The experiments were performed at a constant voltage of 28 KV and the temperature was maintained at 20 C. All standards and samples were injected hydrodynamically for 3 sec by application of a pressure of 0.5 psi. The indirect UV detection was carried out at a wavelength of 254 nm with adenosine 5′-monophosphate (AMP) as a background UV absorber.
A new capillary column was flushed successively with a manual syringe using the following solutions and duration: 1M NaOH for 10 min, followed by water for 3 min, 0.1 M HCl for 10 min, water again for 3 min, and finally with the running electrolyte for 5 min. This successive washing of the capillary was done daily at the start of the experiments using the P\ACE MDQ instrument setting and applying a pressure of 65 psi to the vial for each washing step. Before injections, equilibration of the capillary was carried out at the running voltage (i.e., 28 kV) for 20–30 min with freshly prepared running electrolyte solution (40 mM Tris, 2.5 mM AMP and 7 mM of α-CD in a mixture of NMF-dioxane-water (5∶3∶2, v/v).
The stock solutions of all standard FFAs were prepared by dissolving appropriate amount of acids in NMF-dioxane (4∶1 v/v) to give concentration of 5 mM. All standard solutions were prepared by diluting an aliquot of the stock solutions in the running electrolyte. For the quantitative determination of oleic and linoleic acids in peanut oil samples, an internal standard that shares similar properties with these solutes is the best choice. In this regards, the nonadecanoic acid (C19:0), which is usually absent or present in trace amounts in peanut oils, was selected as the internal standard for establishing the standard calibration curve. The concentrations of standard solutions for the two FFAs oleic acid (C18:1) and linoleic acid (C18:2) used for calibration were 0.2, 0.4, 0.6, 0.8, 1.0, 1.2 and 1.4 mM while the FFA C19:0 used as the internal standard had a concentration of 0.5 mM. The internal standard was first dissolved in dioxane, and then NMF was added. All stock and standard solutions were stored in the refrigerator. In all cases, the quantification of oleic acid and linoleic acid was achieved by comparing peak heights of the fatty acids in the sample with that of the standards from the calibration curve in the range of 0.2 mM to 1.4 mM. The calibration curves for oleic acid and linoleic acid were linear in the concentration range studied (R2 equal to 0.9985 and 0.9961, respectively).
An aliquot (1 or 2 µL) of the purified peanut FFAs was taken and diluted (50-fold or 100-fold) in the final running buffer and vortexed for 4-5 sec. Internal standard FFA C19:0 (0.50 mM) was added to every sample. The sample was then pressure injected in the CE instrument. Hydrodynamic injection of all standards and samples was done for 3 sec at 0.5 psi (1psi = 6895 Pa) at the anodic end. The capillary was pressure rinsed with the running electrolyte for 2 min at 65 psi between injections. Each run was performed at 28 kV with the capillary and sample temperature maintained at 20 °C, and the detection was set at 254 nm. The running electrolyte in the inlet reservoir was changed several times a day while the outlet reservoir electrolyte was changed daily. When not in use, the capillaries were stored in water.
Gas Chromatography Analysis
50µl of peanut extract in 1∶1 n-methylformamide:dioxane was first dried under nitrogen. 100µl of 0.5% (w/v/) butylated hydroxytoluene was added to prevent oxidation and 37.5µl of 10mM methyl nonadecanoate (19∶0) was added as an internal standard (ISTD). Samples were again dried under N2 and saponified with 5% methanolic-potassium hydroxide followed by transmethylation with 14% methanolic-boron trifluoride to form fatty acid methyl esters (FAMEs) (Shipley et.al. 1993). Water was removed by putting samples through magnesium sulfate mini-columns and FAMEs were purified on silicic acid mini-columns. Each sample was reconstituted in 500µl chloroform for a final ISTD concentration of 0.75mM. FAMEs were analyzed by gas chromatography (GC) on a HP5890 Series II gas chromatograph with HP7673 auto-sampler. Samples were introduced onto a DB-225 column (30m × 0.25mm with 0.15µm film thickness) (J&W Scientific) using a split injector set at 250 C with a 1∶25 split ratio. Ultrapure helium was the carrier gas at 1ml/min. The GC program was as follows: 100 C for 2 min, 25 C/min to 180 C, 15 C/min to 200 C, 4 +-C/min to 225 °C and held 6 min. A flame ionization detector set at 300 C was used and peak areas were recorded and quantified using HP-Chemstation software. FAMEs were quantified based on the internal standard added to the sample prior to derivitization and peaks were identified by using retention times compared to standard fatty acid methyl esters (FAMEs, NuCheck Prep, Inc., Elysian MN).
Statistical Analysis
All statistical analyses were conducted with use of PC SAS Version 9 (SAS Institute, Cary, NC). GC ratios and CE ratios were compared with two procedures. First, the possible linear relationship between these ratios was evaluated with a correlation coefficient (PROC CORR). The means ratios for each method and each variety were also compared with paired t-tests (PROC MIXED). A significance level of 0.05 was used to judge significance. Means and standard errors of the two methods (GC and CE) are reported for each variety.
Results and Discussion
As previously shown, it is possible to extract FFAs from small portions of individual peanut seed and determine their oil composition (Bannore et al., 2008). However, until now the accuracy of the CE method had not been compared with the traditional method of gas chromatography. This study is the first written report validating the CE method by direct comparison with GC.
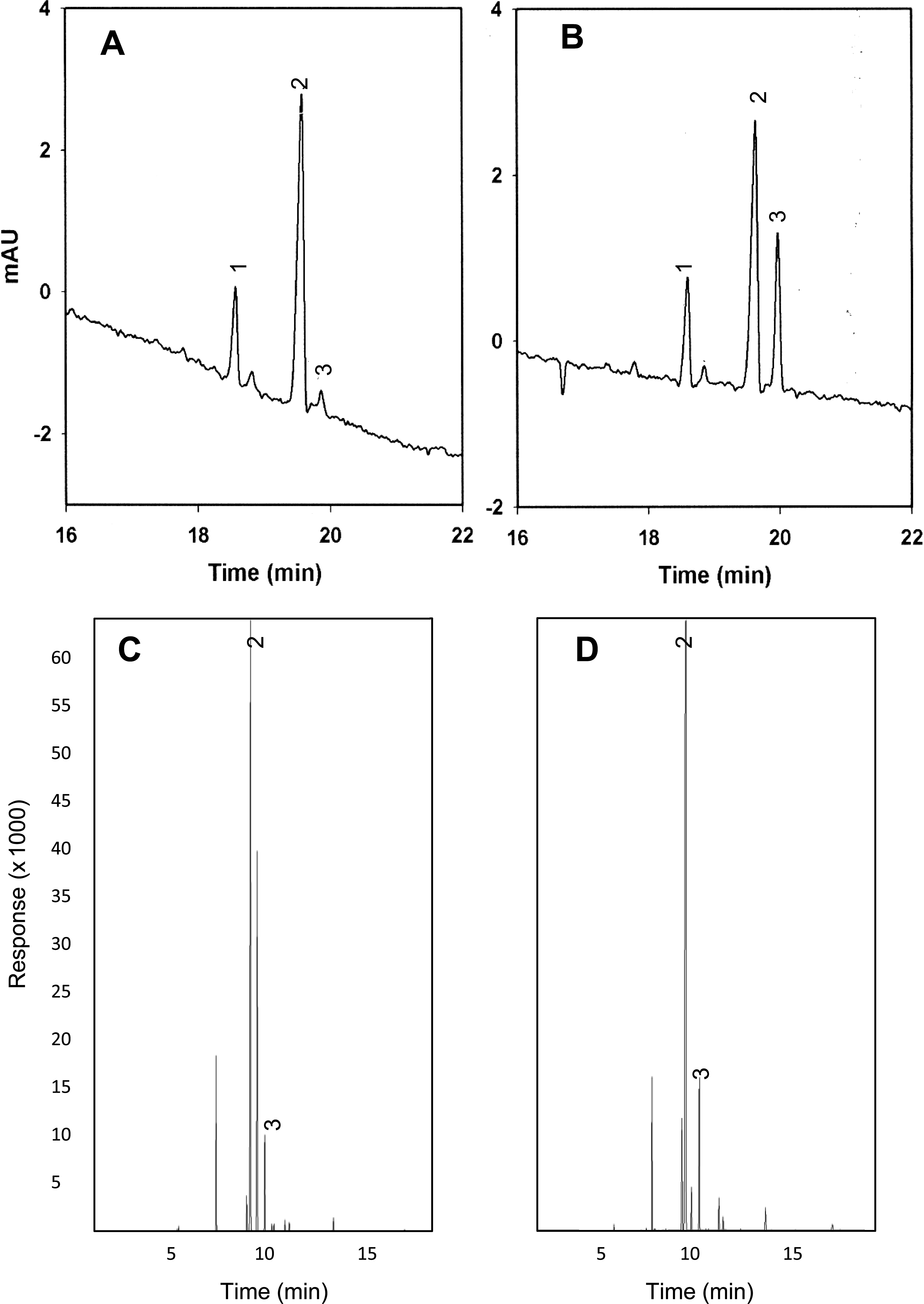
Examples of chromatographs generated by both methods are shown in Figure 1. Excellent resolution of FFAs was achieved using this oil extraction method and subsequent separation by either CE or GC. Chromatographs for analysis of high oleic sample number 87 are shown in Figure 1A and C, while those for the normal oleic sample number 25 are shown in panels B and D. Peaks for the CE internal standard (nonadecanoic acid), oleic and linoleic acid are labeled.
Capillary electrophoresis (A and B) and gas chromatography (C and D) chromatographs determining the O/L ratio in peanut FFA samples. Labeled peaks are 1 = internal standard, 2 = oleic acid, 3 = linoleic acid. Panels A and C = high O/L sample number 87; Panels B and D = normal O/L ratio sample number 25.
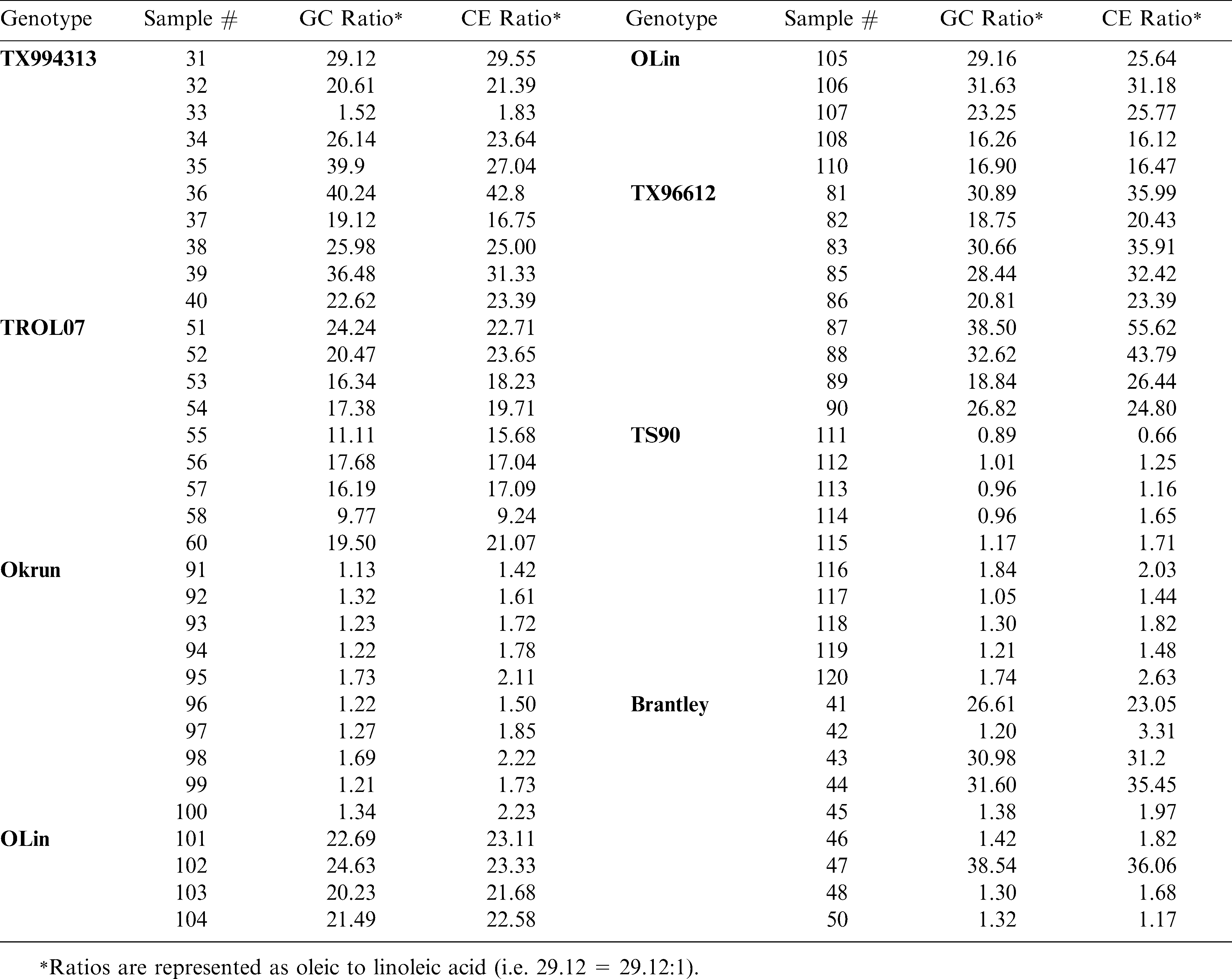
The O/L ratios measured from peanut seed samples are shown in Table 1. Oil was extracted from at least nine and sometimes ten individual seed of eight different genotypes, varying in oleic acid content. All but two of the genotypes tested are released peanut varieties. Genotypes TX994313 and TX996612 are high oleic runner and Spanish (respectively) breeding lines. In all samples tested, the GC and CE O/L ratios measured were in 100% agreement with respect to the sample being labeled as high O/L (ratio ≥ 10∶1) or normal in oil composition. Note that in some cases (i.e. Brantley and TX994313) both methods were able to detect off-types (normal seed within the high oleic lot), which suggests these techniques can serve as a tool for screening segregating populations or determining seed lot purity.
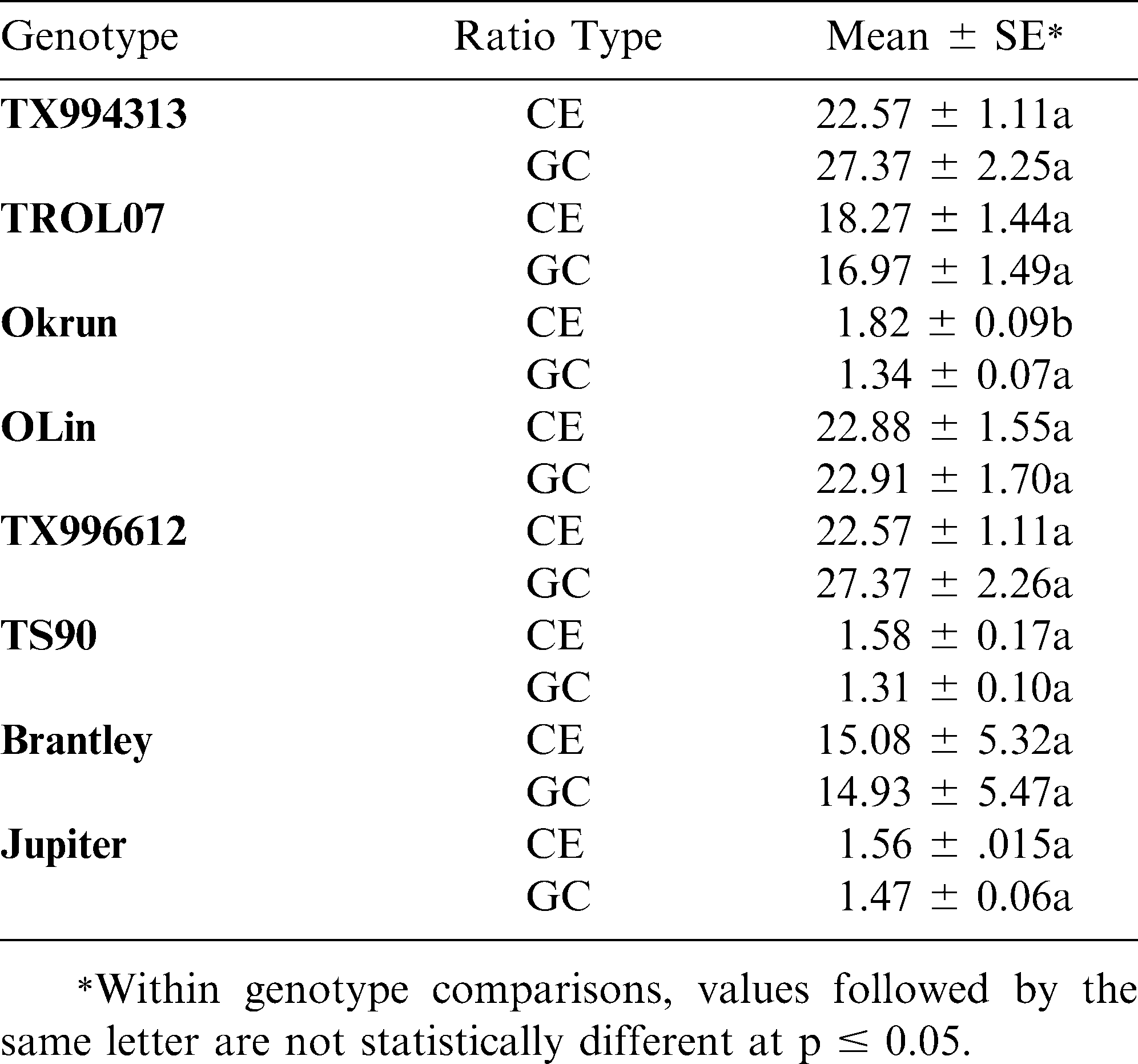
Table 2 shows the results of statistical analysis comparing the GC and CE ratios within individual genotypes. In all cases except one, the methods are statistically indistinguishable, not only in determining high or normal O/L content, but also in accuracy of determining O/L ratio. In the case of the genotype Okrun, the statistical difference between the two methods is only slight and can be explained by the extremely small margin of error within that data set. In general, standard errors for both methods were low, except in the case where a mixed seed lot was tested (i.e. Brantley). The two methods were highly correlated (r = 0.96; p < 0.0001) with respect to determining the exact oleic/linoleic acid ratio from each sample.
Others have compared capillary electrophoresis analysis with separation methods such as high pressure liquid chromatography (HPLC), gas chromatography-mass spectrometry (GC-MS) and gas chromatography. Kronholm et al. (2004) compared CE with GC-MS in the analysis of phenolic compounds and found the analytical values obtained were generally of similar magnitude. Wang et al. (2008) used GC, HPLC and CE to separate nine tryptamine standards and found the methods to be of similar sensitivity and selectivity. The results from this study support previous findings, indicating that CE and GC are interchangeable with regards to sensitivity and accuracy. These results also indicate that the method we developed to extract oil from single peanut seed provides oil of sufficient quality and quantity for further modification and GC analysis. Thus, using our methods, researchers may choose either GC or CE to screen segregating F2 populations while preserving tested seed for further germination and line propagation.
The time required to generate results from start to finish for the discussed methods will vary depending on number of samples being processed. NIR is the obvious choice for a quick classification “high” or “normal” oleic concentration since so oil extraction is necessary and samples can be processed repetitively in a matter of minutes. Genotyping with qPCR takes longer (5–6 hours) but is an extremely powerful tool for breeders since it can determine allele composition. If exact O/L ratio of a sample is needed, GC and CE are equal in accuracy, sensitivity and sample processing time (5–6 hours from extraction to result).
In summary, the results reported here validate CE as an accurate and sensitive method for determining the O/L ratio of individual peanut seed. Use of this method and genotyping methods such as qPCR will enable breeders to determine the genetics and oil composition of early generation breeding material and target specific O/L ratios for further development and release.
Acknowledgements
The authors would like to express their gratitude to Lisa Myers, BreAnn Lindsey and Joren Whitley for technical assistance. Appreciation should also be given to Dr. Tom Isleib of NC State University supplying Brantley seed for analysis. Mention of trade names or commercial products in this article is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture.
Literature Cited
Bannore Y.C Chenault K.D Melouk H.A and El Rassi Z 2008 . Capillary electrophoresis of some free fatty acids using partially aqueous electrolyte systems and indirect UV detection . Application to the analysis of oleic and linoleic acids in peanut breeding lines. J. Sep. Sci. 31 : 2667 – 2676 .
Banks D.J Kirby J.S and Sholar J.R 1989 . Registration of ‘Okrun’ peanut . Crop Sci. 29 : 1574 .
Baring M.R Simpson C.E Burow M.D Black M.C Cason J. Ayers J Lopez Y and Melouk H.A 2006 . Registration of ‘Tamnut OL07’ peanut . Crop Sci. 46 : 2721 – 2722 .
Barkley N.A Chamberlin K.D.C Wang M.L and Pittman R.N 2010 . Development of a real-time PCR genotyping assay to identify high oleic acid peanuts (Arachis hypogaea L.) . Mol. Breed. 25 : 541 – 548 .
Barkley N.A Chamberlin K.D.C Wang M.L and Pittman R.N 2011 . Genotyping and fatty acid composition analysis in segregating peanut (Arachis hypogaea L) populations . Peanut Sci. 38 : 11 – 19 .
Chen Z Wang M.L Barkley N.A and Pittman R.N 2010 . A simple allele-specific PCR assay for detecting FAD2 alleles in both A and B genomes of the cultivated peanut for high-oleate trait selection . Plant Mol. Biol. Rep 28 : 542 – 548 .
Chu Y Ramos L Holbrook C.C and Ozais-Akins P 2007 . Frequency of a loss-of-function mutation in oleoyl-PC desaturase (ahFAD2A) in the mini-core of the U.S. peanut germplasm collection . Crop Sci. 47 : 2372 – 2378 .
Chu Y Holbrook C.C and Ozais-Akins P 2009 . Two alleles of ahFAD2B control the high oleic acid trait in cultivated peanut . Crop Sci. 49 : 2029 – 2036 .
Dermaux A Sandra P and Ferraz V 1999 . Analysis of free fatty acids and fatty acid phenacyl esters in vegetable oils and margarine by capillary electrochromatography . Electrophoresis 20 : 74 – 79 .
Isleib T.G Young C.T and Knauft D.A 1996 . Fatty acid genotypes of five Virginia-type peanut cultivars . Crop Sci. 36 : 556 – 558 .
Isleib T.G Mozingo R.W Copeland S.C Graeber J.B Novitzky W.P Pattee H.E Sanders T.H Mozingo R.W and Coker D.L 2006a . Registration of ‘Brantley’ peanut . Crop Sci. 46 : 2309 – 2311 .
Isleib T.G Wilson R.F and Novitzky W.P 2006b . Partial dominance, pleiotropism, and epistasis in the inheritance of the high-oleate trait in peanut . Crop Sci. 46 : 1331 – 1335 .
Jung S Swift D Sengoku E Patel M Teule F Powell G Moore K and Abbott A 2000a . The high oleate trait in the cultivated peanut [Arachis hypogaea L.]. I. Isolation and characterization of two genes encoding microsomal oleoyl-PC desaturases . Mol. Gen. Genet. 263 : 796 – 805 .
Jung S Powell G Moore K and Abbott A 2000b . The high oleate trait in the cultivated peanut [Arachis hypogaea L.]. II. Molecular basis and genetics of the trait . Mol. Gen. Genet. 263 : 806 – 811 .
Kronholm J Revilla-Ruiz P Porras S.P Hartonen K Carabias-Martinez R and Riekkola M 2004 . Comparison of gas chromatography-mass spectrometry and capillary electrophoresis in analysis of phenolic compounds extracted from solid matrices with pressurized hot water . J. Chromatogr. A. 1022 : 9 – 16 .
Lopez Y Smith O.D Senseman S.A and Rooney W.L 2001 . Genetic factors influencing high oleic acid content in Spanish market-type peanut cultivars . Crop Sci. 41 : 51 – 56 .
Moore K.M and Knauft D.A 1989 . The inheritance of high oleic acid in peanut . J. Hered. 80 : 252 – 253 .
Norden A.J Gorbet D.W Knauft D.A and Young C.T 1987 . Variability in oil quality among peanut genotypes in the Florida breeding program . Peanut Sci. 14 : 7 – 11 .
O'Byrne D.J Knauft D.A and Shireman R.B 1997 . Low fat-monosaturated rich diets containing high-oleic peanuts improve serum lipoprotein profiles . Lipids 32 : 687 – 695 .
Patel M Jung S Moore K Powell G Ainsworth C and Abbott A 2004 . High-oleate peanut mutants result from a MITE insertion into the FAD2 gene . Theor. Appl. Gen. 108 : 1492 – 1502 .
Phillips R.J and Singleton B 1978 . The determination of specific free fatty acids in peanut oil by gas chromatography . J. Amer. Oil Chem. Soc. 55 : 225 – 227 .
SAS Inst . 2008 . SAS/STAT Guide for personal computers . Ver.9.0 Ed. SAS Int , Cary, N.C .
Shipley M.M Dillwith J.W Bowman A.S Essenberg R.C and Sauer J.R 1993 . Changes in lipids of the salivary glands of the lone star tick, Amblyomma americanum, during feeding . J. Parasit. 79 : 834 – 842 .
Simpson C.E Baring M.R Schubert A.M Melouk H.A Lopez Y and Kirby J.S 2003 . Registration of ‘Olin’ peanut . Crop Sci. 43 : 1880 – 1881 .
Singleton J.A and Pattee H.E 1987 . Characterization of peanut oil triglycerols by HPLC, GLC and EIMS . J. Amer. Oil Chem. Soc. 64 : 534 – 538 .
Smith O.D Simpson C.E Grichar W.J and Melouk H.A 1991 . Registration of ‘Tamspan 90’ peanut . Crop Sci. 31 : 1711 .
Teres S Barcelo-Coblijn G Benet M Alvarez R Bressani R Halver J.E and Escriba P.V 2008 . Oleic acid content is responsible for the reduction in blood pressure induced by olive oil . Proc. Natl. Acad. Sci. USA 105 : 13811 – 13816 .
Tillman B.L Gorbet D.W and Person G 2006 . Predicting oleic and linoleic acid content of single peanut seeds using near-infrared reflectance spectroscopy . Crop Sci. 46 : 2121 – 2126 .
Vassiliou E.K Gonzalez A Garcia C Tadros J.H Chakraborty G and Toney J.H 2009 . Oleic acid and peanut oil high in oleic acid reverse the inhibitory effect of insulin production of the inflammatory cytokine TNF-α in both in vitro and in vivo systems . Lipids in Health and Disease . doi: 10.1186/1476-511X-8-25.
Wang M Liu J Chen H Lin J and Lin C 2008 . Comparison of the separation of nine tryptamine standards based on gas chromatography, high performance liquid chromatography and capillary electrophoresis methods . J. Chromatogr. A 1181 : 131 – 136 .
Notes
Author Affiliations
1.Corresponding Author, Kelly D. Chamberlin, USDA-ARS, Wheat, Peanut and other Field Crops Research Unit, 1301 N. Western, Stillwater, OK 74075; Kelly.chamberlin@ars.usda.gov.