As demand for organic products has increased in the U.S. in recent years, the demand for organic peanuts (Arachis hypogaea L.) has subsequently followed a similar upward trend (Dimitri and Greene, 2002; Lamb et al. 2007; Puppala, 2007). Demand for organic peanuts is generally regarded as the fastest growing sector of the entire U.S. peanut industry (Lamb et al., 2007; Parker, 2006; Puppala, 2007). Heightened demand for organic peanuts has translated to higher price premiums for certified organic versus conventional peanut. At the time of this writing, certified organic runner-type peanut prices can reach $1100/Mg, based on contracts, compared to approximately $390/Mg for conventional runner peanut (N.B. Smith, personal communication, 2010). Therefore, economic incentives exist for peanut growers interested in pursuing organic production. However, these economic incentives are sometimes negated by lower yields and additional costs associated with organic production such as additional trips through the field for cultivation, hand-weeding, and land certification costs.
Disease and weed pressure are significant limiting factors to organic peanut production, especially in the southeastern U.S. (Branch and Culbreath, 2008; Guerena and Adam, 2008; Johnson et al., 2008). However, numerous runner-type cultivars have been released in recent years that have displayed excellent disease resistance and productive capacity in organic production scenarios (Branch and Culbreath, 2008; Holbrook and Culbreath, 2008; Holbrook et al., 2008). Therefore, identifying effective, affordable weed control regimes for organic peanut is paramount for the development of commercial production.
Weed management in conventional peanut cropping systems is typically achieved via a combination of crop rotation and various cultural practices, as well as cultivation and herbicide use for control (Ferrell, et al. 2009; Hauser et al., 1973). In certified organic production, application of synthetically-derived herbicides is forbidden (USDA, 2005). There are herbicide options available for organically-grown crops that are derived from natural sources. However, these herbicides are limited in scope and efficacy and cost-prohibitive for most commercial production (Johnson and Mullinix, 2008; Johnson et al., 2008). Therefore, focus has shifted to non-chemical methods for weed control in organic peanut. Numerous non-chemical weed control techniques are available for organic systems, including mechanical, thermal, mulching, and biological methods (Bond and Grundy, 2001), although these techniques have shown mixed results. Johnson and Mullinix (2008) reported that clove oil, citric acid + acetic acid, and broadcast propane flaming used for remedial weed control, combined with sweep cultivation, were ineffective against annual grasses and dicot weeds and resulted in decreased peanut yields in an organic production scenario.
Sweep cultivation can be an effective method of controlling both mature weeds and weeds between peanut rows (Böhrnsen, 1993; Bowman, 1997; Wilcut et al., 1987). However, sweep blades are unable to remove weeds in or near the actual peanut row, where the effects of weed competition are greatest on crop plants (Bowman, 1997; Johnson and Mullinix, 2008; Smith et al., 2000). Tine cultivation, utilizing a tine cultivator with rigid or spring-loaded tines, is an effective method for reducing in-row weed populations with minimal damage to the crop. The metal tines vibrate vigorously, providing weed control by disrupting or burying germinating weed seedlings within the top 3 cm of soil. Most importantly, the tines are able to cultivate much closer to the crop rows than sweep blades, thus improving weed control closest to the actual crops (Bowman, 1997; Reddiex et al., 2001). Therefore, integration of tine cultivation into the organic weed control program could be an effective method of mechanical in-row weed control for organic peanut. The objectives of this study were to (1) evaluate the efficacy and economics of tine cultivation + sweep cultivation + hand-weeding on weed control compared to no cultivation or weeding and (2) assess the effects of various frequencies and durations of tine cultivation on weed control and overall productivity of peanut grown under organic management.
Materials and Methods
Irrigated field trials were conducted at the University of Georgia (UGA) Horticulture Hill and Lang-Rigdon Farms near Tifton, GA in 2008 and 2009, respectively. The trials were conducted on a Tifton loamy sand (fine-loamy, Kaolinitic, thermic Plinthic Kandiudults), with 88% sand, 10% silt, 2% clay, and 0.56% organic matter (2008 site) and 90% sand, 6% silt, 4% clay, and 0.88% organic matter (2009 site). All plots were planted 6 June 2008 and 5 June 2009 using a 2-row Monosem precision air planter (Monosem, Inc., Edwardsville, KS) at a depth of 6 cm and a seeding rate of 20 seed/m. Production practices included conventional tillage prior to planting and irrigation as needed during the season, according to UGA recommendations for peanut (UGA, 2010). No herbicides, fungicides (including seed treatments), or insecticides were applied to the plots at any point during the season in order to follow approved USDA-certified organic production practices.
The experiment utilized a strip-plot design with a 2 × 3 factorial arrangement of tine cultivation treatments including two frequencies (once per week [1×] or twice per week [2×]) for three durations (3 wk, 4 wk, or 5 wk). A non-cultivated, non-weeded control treatment was also included for comparison, to evaluate whether or not cultivated treatments provided any benefit over no cultivation/weeding. Including a non-weeded control resulted in seven total cultivation treatments and all treatments were replicated four times across the field. The seven cultivation treatments were overlaid in strips across two peanut cultivars to create the strip-plot design. A strip-plot design was utilized to allow sufficient plot length to facilitate equipment operation at an optimum speed. Individual plot sizes were 1.8 m × 9.2 m, with a 0.9-m standard row spacing. Cultivars planted were ‘Georganic’ (Holbrook and Culbreath, 2008) and ‘Tifguard’ (Holbrook et al., 2008), which have displayed strong host plant resistance to foliar diseases. Additionally, Tifguard has displayed significant resistance to the peanut root-knot nematode (Meloidogyne arenaria [Neal] Chitwood), making it a strong option for organic production (Holbrook et al., 2008). Tine cultivations were conducted using a tine cultivator (Aerostar, Einböck GmbH & CoKG, Austria) (Fig. 1) and were initiated 10 days after planting (DAP) in 2008 and 9 DAP in 2009. Cultivation with flat sweeps was conducted once on all cultivated plots at the 3 wk tine cultivation (24 DAP in 2008 and 23 DAP in 2009) to control inter-row weeds. All cultivated plots were hand-weeded twice in 2008 (23 June and 14 July) and once in 2009 (28 July) to remove escaped weeds. Hand-weeding times were recorded for each plot. Sweep cultivation and supplemental hand-weeding were employed to ensure measurable yields for each treatment at the end of the season and for evaluation of each system as a whole for potential implementation by growers.
Late-season weed density estimates were conducted on 28 Oct. 2008 and 22 Oct. 2009 using a 0.5-m2 quadrat, counting individual plants within the quadrat. Weed estimates were conducted at the end of the season, immediately prior to peanut inversion, to assess season-long effects of each treatment on weed control and to allow later-germinating weeds to be present for accurate identification and counting. Despite different maturity ranges for Tifguard (approx. 135 d) and Georganic (approx. 150 d), all plots in 2008 were inverted using a 2-row KMC digger-shaker-inverter (Kelley Manufacturing Co., Tifton, GA) on 30 Oct. due to cool temperatures (four consecutive days of minimum temperatures below 8° C) causing continued development to cease, and harvested 6 Nov. using a 2-row Lilliston small plot peanut combine. In 2009, Tifguard plots were inverted on 23 Oct. and Georganic plots on 28 Oct. (cool October temperatures again ceased additional development of the Georganic cultivar) and all plots were harvested 4 Nov. 2009. Final plant stand, hand-weeding time, final pod yield, and weed density data were collected for each treatment. Pod yields were adjusted to 7% moisture. Final plant stand counts were conducted after peanut inversion on 6 Nov. 2008 and 2 Nov. 2009, covering 3 m of row per plot. Stand counts were conducted by placing a 1.5-m-long pole between two peanut rows and counting the number of plants that fell within the 1.5-m distance in each row.
Economic analysis was also conducted for each treatment. Input costs for organic production included hand labor, seed, equipment, and fuel costs. Revenue data were generated based on an $1100/Mg premium for organic peanut and a $9.12/hr wage for hand labor (NASS-GA, 2010). Adjusted net revenue (ANR) was determined as ANR = Y * (P − M) − (S − R − D), where Y is yield (kg/ha), P is price ($/ha), M is marketing costs (e.g. checkoff funding) ($/ha), S is seed cost ($/ha), R is costs for repair, equipment, labor, and fuel ($/ha), and D is drying costs ($/ha) for each treatment.
Statistical analysis was performed with SAS 9.2 software (SAS Institute, 2009). Data were statistically analyzed by analysis of variance, using PROC GLIMMIX. Means were separated in PROC GLIMMIX via multiple pairwise t-tests at P ≤ 0.05. Two separate statistical analyses were conducted for the data, similar to the analyses conducted by Arnold et al. (2002). The first analysis included the non-cultivated control treatment, in order to statistically compare each cultivated treatment to the control. The second analysis excluded the control to test the significance of frequency and duration main effects on each variable.
Results and Discussion
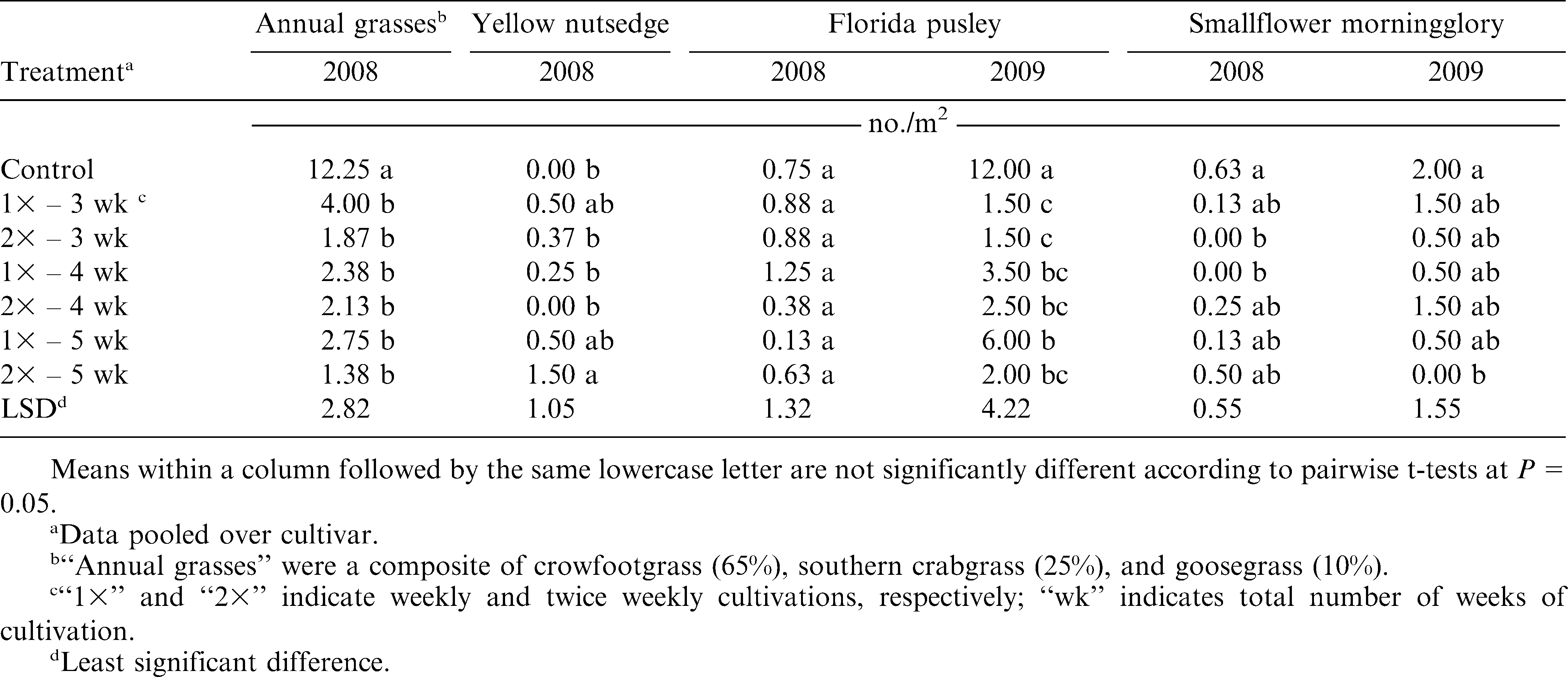
Prevalent weed species differed significantly between the two years. Pressure from annual grass species and perennial nutsedges was greater at the 2008 site than the 2009 site. Therefore, weed population estimates were performed on crowfootgrass (Dactyloctenium aegyptium [L.] Willd.), goosegrass (Eleusine indica [L.] Gaertn.), southern crabgrass (Digitaria ciliaris [Retz.] Koeler), and yellow nutsedge (Cyperus esculentus L.) in 2008 but not in 2009. Populations of these weed species were too sparse to legitimately estimate in 2009. Estimates of Florida pusley (Richardia scabra L.) and smallflower morningglory (Jacquemontia tamnifolia [L.] Griseb.) were performed both years.
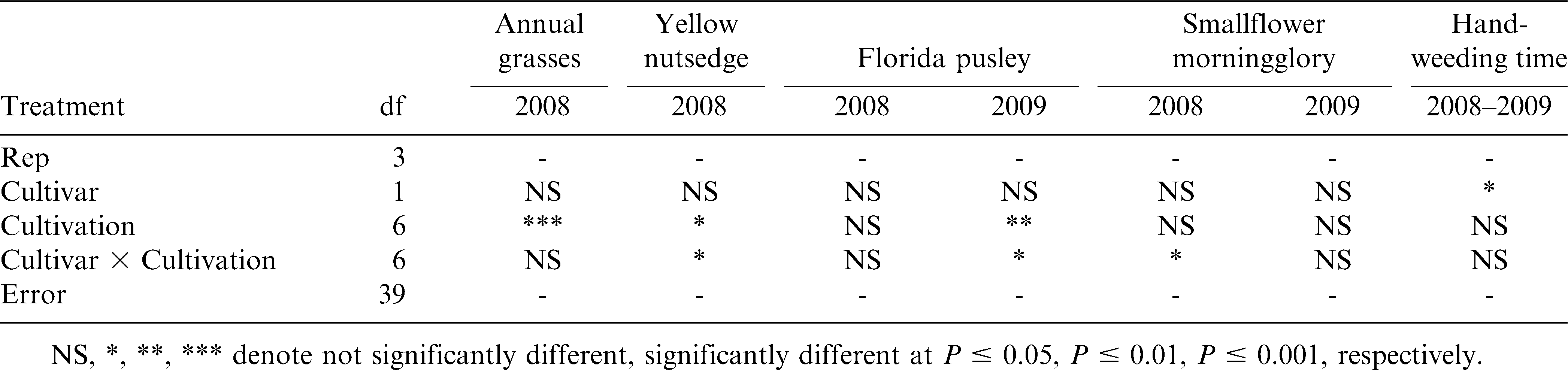
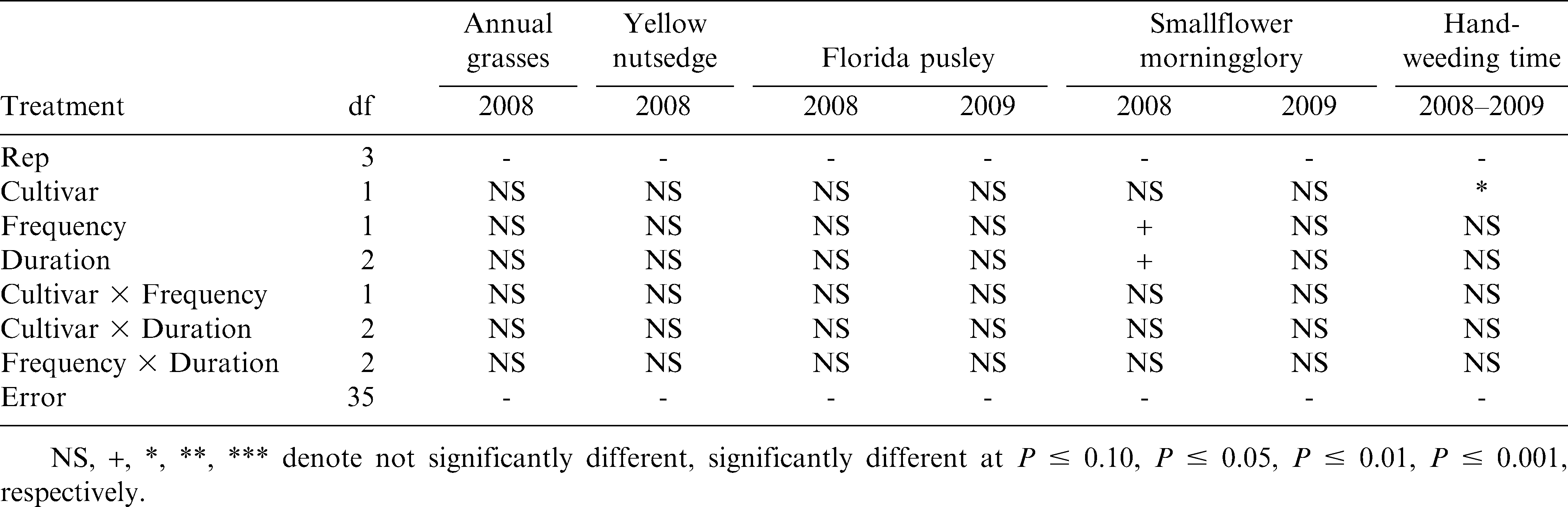
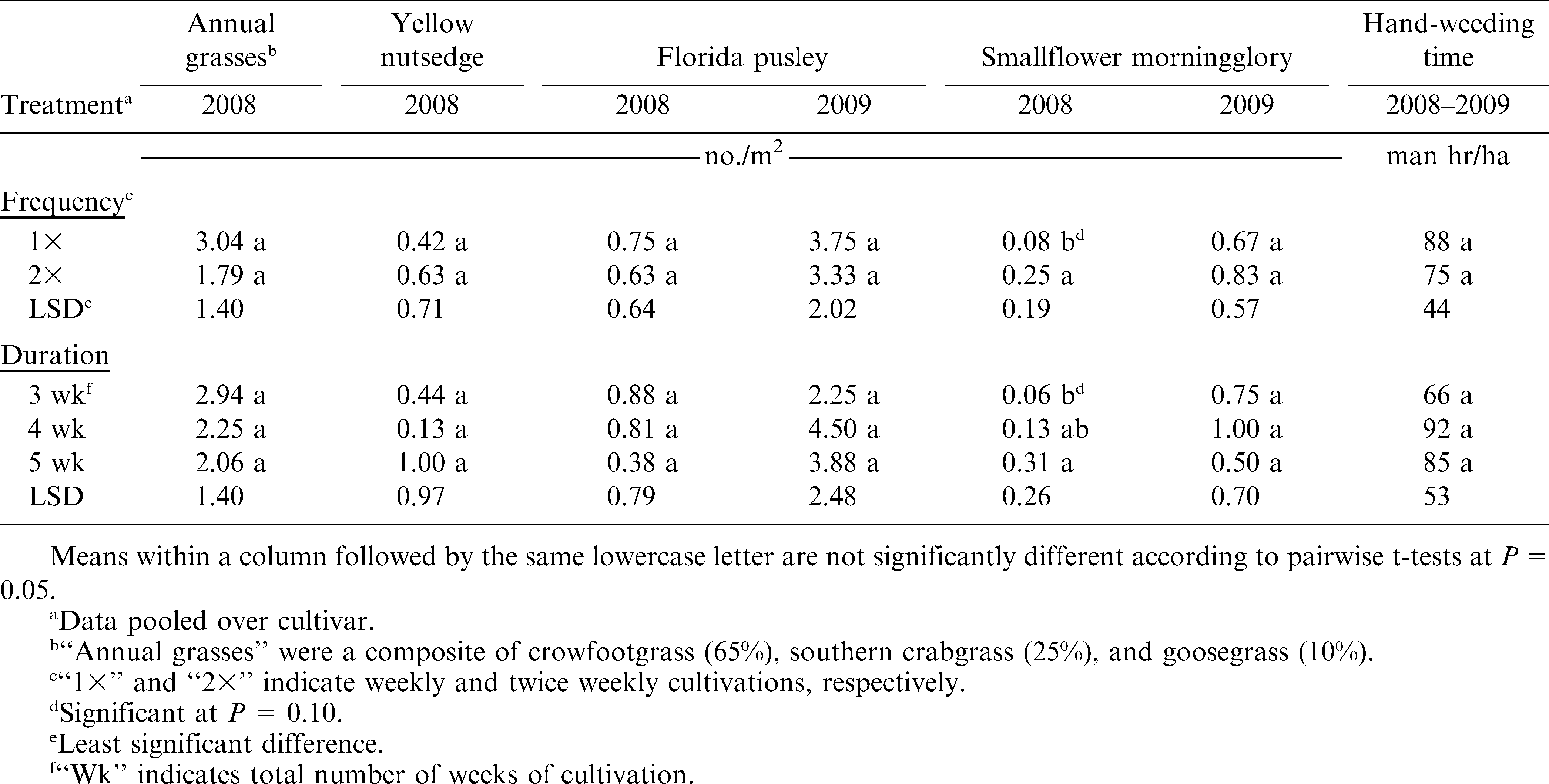
In the analyses that included the non-weeded control, there were significant Cultivar × Cultivation interactions (P ≤ 0.05) for yellow nutsedge data in 2008, Florida pusley data in 2009, smallflower morningglory data in 2008 (Table 1), and peanut grade data in 2008 (Table 2). However, analysis of each treatment, separated by cultivar, provided little additional information, especially since the yellow nutsedge, smallflower morningglory, and grade treatment data showed little or no difference from the control when pooled over cultivar or analyzed separately. Similarly, every treatment in 2009 reduced Florida pusley density compared to the control except for one Tifguard treatment. Therefore, for the sake of brevity, the results for each of these response variables were pooled over cultivar and are presented as such below. In the analyses that excluded the non-weeded control, there were no significant Cultivar × Frequency, Cultivar × Duration, or Frequency × Duration interactions (P ≤ 0.05) for any response variable (Tables 3 & 4). These results were therefore also pooled over cultivar. Cultivar data were pooled over cultivation treatment, to present cultivar-only effects on select response variables.
Annual Grass Control
The three grass species counted in 2008 were combined for analysis and are subsequently expressed as “annual grasses,” consisting of approximately 65% crowfootgrass, 25% southern crabgrass, and 10% goosegrass. All cultivation treatments reduced annual grass weed populations over the control (P ≤ 0.05), but this effect was not observed for yellow nutsedge populations (Table 5). Cultivation was mostly ineffectual against nutsedge, with the 2× – 5 wk treatment actually resulting in a greater density than the control (P ≤ 0.05). This may either have been a result of inconsistent nutsedge densities at the 2008 site or a potentially synergistic effect of cultivation, which may have increased nutsedge densities. Frequency and duration of cultivation did not have a significant effect on either annual grass or yellow nutsedge density (P ≤ 0.05) (Table 6). Overall, any level of tine cultivation in this experiment, combined with sweep cultivation and hand-weeding, proved effective in reducing annual grass density compared to the non-cultivated group, but not yellow nutsedge.
Dicot Weed Control and Hand-Weeding Times
The effects of cultivation on Florida pusley varied between 2008 and 2009 (Tables 5 & 6), due primarily to the differing baseline pressure between the two test sites. The greatest weed pressure at the 2008 site was from annual grass species, with relatively low baseline Florida pusley populations. However, Florida pusley was the dominant weed species at the 2009 site. Therefore, treatment differences in Florida pusley control were non-existent in 2008, but were very pronounced in 2009. Similar to the trends in annual grass control, all cultivated treatments reduced Florida pusley populations compared to the control (P ≤ 0.05) in 2009 (Table 5). Based on these results, it appears that when Florida pusley is predominant, these cultivation regimes can reduce Florida pusley densities. However, when baseline Florida pusley densities are relatively low, these regimes are mostly ineffective at further reducing weed densities. It is also possible that in 2008, when grass pressure was heavier, the more upright growth habit of the annual grass weeds allowed them to out-compete Florida pusley, which grows more prostrate. Thus, the surviving number of Florida pusley plants could have been equalized by this effect in all treatments, including the non-cultivated plots. Frequency and duration effects were insignificant on Florida pusley density (P ≤ 0.05) (Table 6).
Cultivation appeared to have a minor effect on smallflower morningglory populations, but was inconsistent between the two years of this experiment (Table 5). All but two cultivated treatments in 2008 and one cultivated treatment in 2009 were ineffective at reducing smallflower morningglory densities over the control. In fact, the 2× – 3 wk and 1× – 4 wk treatments actually increased morningglory densities compared to the control in 2008. Weekly cultivations reduced morningglory density compared to bi-weekly cultivations (P ≤ 0.10) in 2008, but cultivation frequency effects were insignificant in 2009 (Table 6). In 2008, increased duration of tine cultivation appeared to actually increase smallflower morningglory densities. The 5-wk duration in 2008 significantly increased morningglory density compared to the 3-wk duration (P ≤ 0.10). Although baseline morningglory densities were greater in 2009, duration effects were insignificant. Based on these results, increased numbers of tine cultivation appear to have potential for increasing smallflower morningglory density, although cultivation effects were inconsistent between both years of this experiment. More research is needed to evaluate the potential of cultivation for smallflower morningglory control in organic peanut.
Hand-weeding times did not differ among cultivation frequency or duration both years (P ≤ 0.05) (Table 6). These results indicate that increasing the number of cultivations did not significantly reduce the amount of time spent on hand-weeding. This is an important point to consider, as hand-weeding represents one of the costliest inputs to any organic cropping system. Additional research is needed to evaluate the interaction of hand-weeding times with varying levels of tine and sweep cultivation to determine the optimum weed control strategies for agronomic and economic productivity.
Final Plant Stand, Pod Yield, and Grade
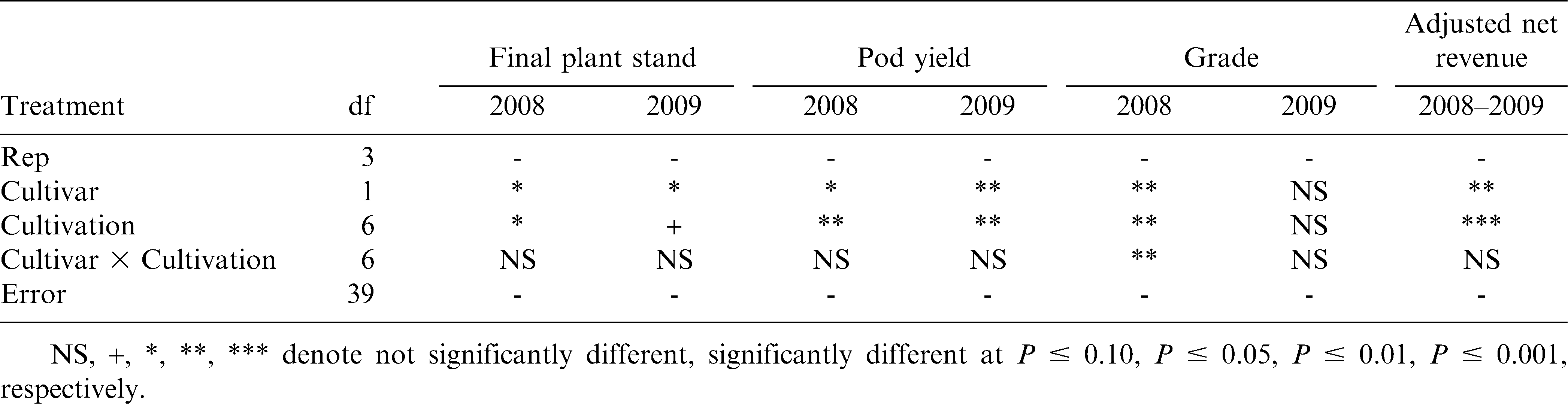
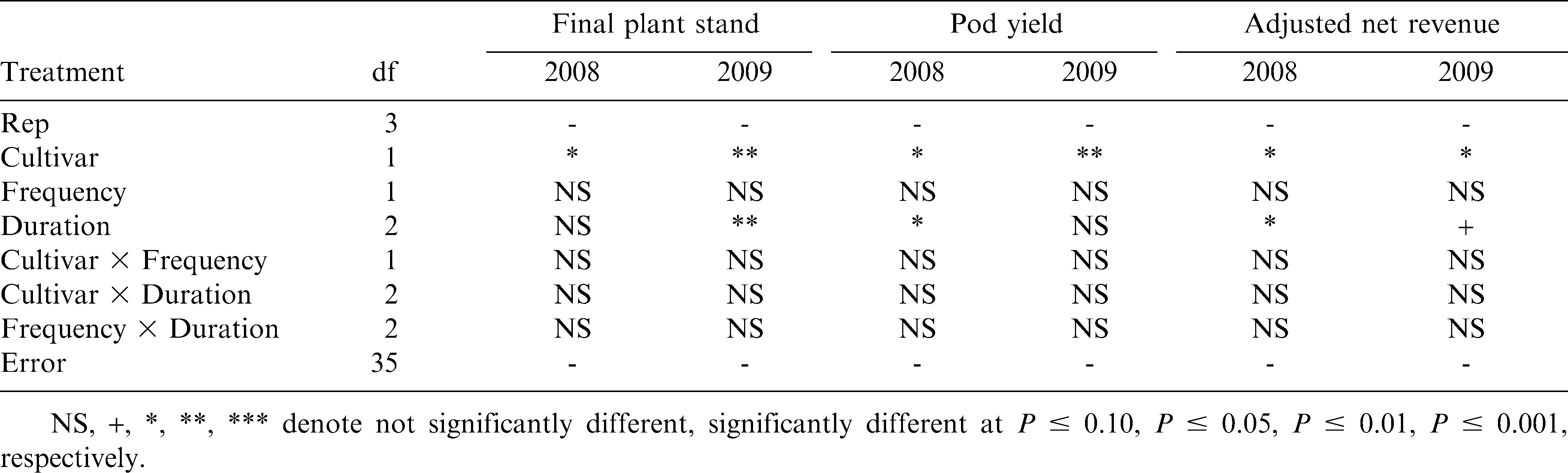
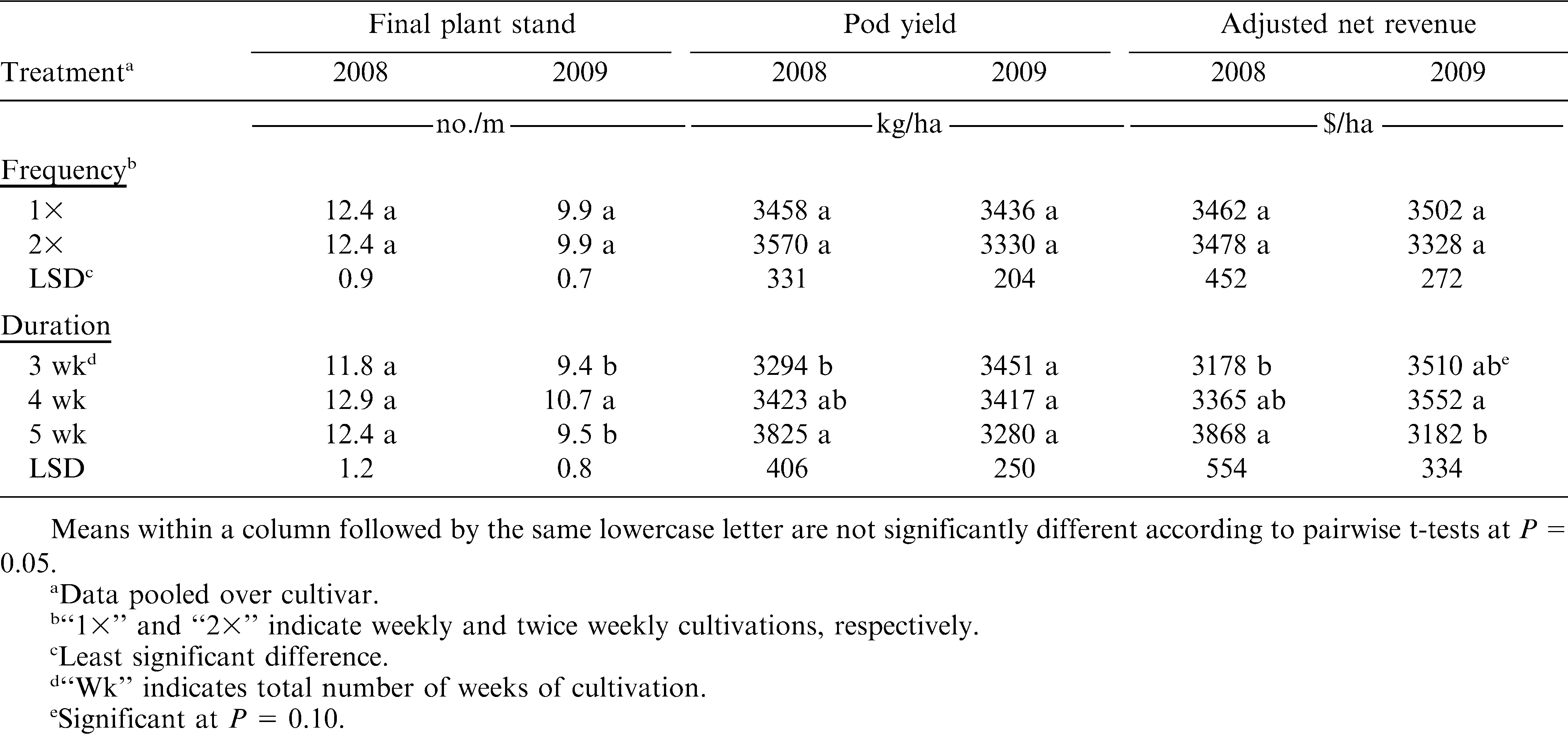
In 2008, all cultivated treatments resulted in greater peanut stands compared to the control (P ≤ 0.05) (Table 7). However, in 2009, only two of the cultivation treatments resulted in higher plant densities than the control (P ≤ 0.05). These results could have been influenced by the varying weed pressure between the site-year locations. The greater baseline annual grass densities in 2008 may have had a greater impact on peanut stands than in 2009, when baseline pressure was almost non-existent. The harmful effects of annual grass weeds on peanut have been documented (Johnson and Mullinix, 2005); therefore, reducing annual grasses densities in 2008 using the weed control regimes in this experiment likely resulted in the similar plant stand trends that year. This is an important consideration in organic peanut systems, where proper stand establishment is crucial for ensuring minimized disease impacts and adequate ground coverage for maximum peanut productivity. Both cultivation frequency and duration effects in 2008, however, were insignificant for plant stand (P ≤ 0.05) (Table 8). Frequency effects in 2009 were insignificant, but the 4-wk duration resulted in the greatest final peanut stands that year (P ≤ 0.05). It is unclear as to why the 4-wk duration of tine cultivation improved peanut stands in 2009, since it did not have a significant effect on weed control that year.
All cultivation treatments significantly improved pod yields over the control (P ≤ 0.05) in both years of the experiment (Table 7). Notably, cultivation in 2008 resulted in a 250% yield increase over the non-cultivated treatment, compared to only a 60% yield increase in 2009. Johnson and Mullinix (2005) reported that Texas panicum (Panicum texanum Buckl.) densities of 2.2 plants/m2 reduced pod yields by 25% and other annual grasses have a similar impact on peanut yields. The greater density of annual grasses in 2008 likely caused heavy yield reductions in the control, where annual grasses occurred at approximately 12.3 plants/m2 by the end of the season. Cultivation frequency effects on yield were insignificant both years (P ≤ 0.05) (Table 8). Duration of cultivation, however, significantly affected pod yield in 2008, where the 5-wk duration significantly improved pod yield over the 3-wk duration. Duration effects in 2009 were insignificant. These results indicate that any level of tine cultivation along with sweep cultivation and hand-weeding can result in significant yield improvement. Although cultivation frequency effects were insignificant, increasing the duration of tine cultivation has the potential for improving peanut yields in areas of significant annual grass pressure. Peanut grades in cultivated treatments were not different from the control in either year of this experiment (Table 7).
Economic Results
Net revenues were significantly affected by cultivation in both years of this experiment. All levels of tine cultivation combined with sweep cultivation and hand-weeding improved net revenues over the control (P ≤ 0.05) both years (Table 7). Revenues from cultivated treatments were 85–100% greater than the control, despite numerous passes across the field and hours of supplemental hand-weeding. Cultivation frequencies had no effect on net revenues either year (P ≤ 0.05); duration effects, however, were significant in both years of the experiment (Table 8). In 2008, similar to pod yield and grade, the 5-wk duration improved net revenue over the 3-wk duration, despite a greater number of total passes across the field. Yet, in 2009, the 4-wk duration improved net revenue over 5 wk (P ≤ 0.10). The additional pass(es) through the field at the 5-wk duration, coupled with numerically suppressed yields that may have been a result from minor vine damage close to bloom and peg formation while gaining no added weed control benefit, likely caused revenues to be statistically lower with the 5-wk duration. These results indicate that tine cultivations at least once weekly for either 4 or 5 wk (depending on species and severity of weeds), combined with sweep cultivation and hand-weeding, can significantly improve net revenues over a non-cultivated, non-weeded control. However, it should be understood that considerable time and effort in weed management to get weed seedbank populations to controllable levels is paramount prior to growing organic peanut. Therefore, economic incentives exist for peanut growers who are interested in organic production, but those higher revenues are needed in order to offset low revenues associated with the transitional period before organic certification, when organic management must be followed but organic premiums are not yet offered.
Cultivar Comparison
Differences in plant stand, hand-weeding time, pod yield, and net revenue were also significant between Georganic and Tifguard in both 2008 and 2009 (Table 9). Tifguard plots produced denser plant stands than Georganic and resulted in subsequently lower hand-weeding times. Tifguard also generated greater yield and net revenue than Georganic both years. The greater stand establishment of Tifguard, combined with its rapid, lateral growth habit, likely provided quicker coverage of the soil between rows, aiding in suppressing weed growth and reducing the amount of hand-weeding for the cultivar. The lower hand-weeding times in Tifguard treatments, combined with greater peanut stands and yields, resulted in a more profitable overall system than did the Georganic treatments. It should be noted, however, that Georganic is a later-maturing cultivar and was not able to reach full maturity in either year of this experiment, due to cold weather shutdown. This could have had a significant impact on Georganic yields and subsequent net revenues both years. However, with a shorter growing season than Georganic and unique host plant nematode resistance (Holbrook et al., 2008), Tifguard appears to be a strong option for maximizing both yield and profitability in an organic peanut system.
Summary and Conclusions
These results suggest that tine cultivation, combined with flat-sweep cultivation and supplemental hand-weeding, is a viable option for managing weeds and optimizing production and profitability of Georganic and Tifguard peanut cultivars in an organic management scenario. In this experiment, cultivated treatments significantly reduced densities of annual grasses and Florida pusley over the control. They did not, however, effectively control yellow nutsedge or smallflower morningglory, although both species had relatively low baseline populations in this experiment. These weed control regimes also resulted in significant plant stand and yield improvements. Yields in cultivated treatments were 60–250% greater than those in the non-cultivated control. There was also evidence that initial weed species composition played a significant role in the effectiveness of cultivation on peanut stand and pod yield. Annual grasses are very competitive with peanut. However, the combination of timely tine cultivating, sweep cultivation, and supplemental hand-weeding minimized annual grasses and maximized pod yields. Varying cultivation frequency had almost no significant effect on weed control or productivity of peanut. Cultivating at least once a week for a duration of either 4 or 5 wk after planting, however, displayed potential for improving peanut stands, yields, and net revenues in an organic production system, especially when annual grass weeds were predominant. More research is needed to assess various combinations of tine and sweep cultivations that may be even more effective and profitable. Tifguard appears to be a strong cultivar option for organic production, having resulted in lower hand-weeding times and producing greater plant stands, yields, and net revenues than Georganic.
With the high price premiums available today for certified organic peanut, there is significant potential for the profitability of utilizing tine cultivation regimes for weed control in these systems. From our observations, it was also apparent that supplemental hand-weeding is an unavoidable necessity in any organic weed management program and represents one of the costliest inputs to any organic cropping system. A limitation of this data is that we did not have the ability to include a cultivated control with no hand-weeding to quantify differences of cultivation versus hand-weeding. Additional research is needed to address such differences.
Acknowledgements
The technical efforts of Paige Adams, Katie Davis, Corey Thompson, and Will Vance were essential to the success of this research project and are supremely appreciated. We are also grateful to Calcium Products, Inc. for their donation of OMRI-approved gypsum and the National Peanut Board and Georgia Peanut Commission for providing financial assistance for the high labor requirements of this experiment.
Literature Cited
Arnold M. D Rummel D. R Bordovsky J. P Slosser J. E and Carroll S. C Use of the center pivot irrigation system for reduction of cotton aphid sugars on cotton lint Southwest. Entomol. 2002 27 ( 1 ): 11 – 19 .
Böhrnsen A Several years' results about mechanical weeding in cereals In Communications 4th International Conference IFOAM, Non-chemical Weed Control , Dijon, France 1993 95 – 101 .
Bond W and Grundy A. C Non-chemical weed management in organic farming systems Weed Res. 2001 41 : 383 – 405 .
Bowman G Steel in the Field: a Farmer's Guide to Weed Management Tools. Burlington, VT: Sustainable Agriculture Publications Univ. of Vermont , Burlington, VT 1997 .
Branch W. D and Culbreath A. K Disease and insect assessment of candidate cultivars for potential use in organic production Peanut Sci. 2008 35 : 61 – 66 .
Dimitri C and Greene C Recent growth patterns in the U.S. organic foods market [Online] 2002 USDA-Econ. Res. Ser. Agric. Info. Bull. No. 777. Available at http://www.ers.usda.gov/publications/aib777/aib777c.pdf (verified 6 Oct. 2011) .
Ferrell J. A MacDonald G. E and Brecke B. J Weed management in peanuts. Spec. Ser. SS-AGR-03 Florida Coop. Ext. Serv., IFAS, Univ. of Florida , Gainesville Available at http://edis.ifas.ufl.edu/wg008 (verified 6 Oct. 2011). 2009 .
Guerena M and Adam K National Sustainable Agriculture Information Service 2008 Peanuts: organic production. NCAT: Butte, MT .
Hauser E. W Cecil S. R and Dowler C. C Systems of weed control for peanut Weed Sci. 1973 21 ( 3 ): 176 – 180 .
Holbrook C. C and Culbreath A. K Registration of ‘Georganic’ peanut J. Plant Reg. 2008 2 : 17.
Holbrook C. C Timper P Culbreath A. K and Kvien C. K Registration of ‘Tifguard’ peanut J. Plant Reg. 2008 2 : 92 – 94 .
Johnson W. C and Mullinix B. G Texas panicum (Panicum texanum) interference in peanut (Arachis hypogaea) and implications for treatment decisions Peanut Sci. 2005 32 : 68 – 72 .
Johnson W. C and Mullinix B. G Potential weed management systems for organic peanut production Peanut Sci. 2008 35 : 67 – 72 .
Johnson W. C Mullinix B. G and Boudreau M. A Peanut response to naturally-derived herbicides used in organic crop production Peanut Sci. 2008 35 : 73 – 75 .
Lamb M. C Cantonwine E Sorensen R. B Nuti R. C Tillman G and Smith N. B Economics of organic versus conventional peanut and cotton Proc. Am. Peanut Res. Educ. Soc. 2007 39 : 73 (abstr.) .
Parker B Organic peanut production in the US: the sheller's perspective Proc. Am. Peanut Res. Educ. Soc. 2006 38 : 92 (abstr.) .
Puppala N Effects of biofungicides and botanical extracts on yield and quality of Valencia peanut Proc. Am. Peanut Res. Educ. Soc. 2007 39 : 70 (abstr.) .
[NASS-GA] National Agricultural Statistics Service in cooperation with the Georgia Department of Agriculture , Farm labor news release: hired workers down 1 percent, wage rates up 2 percent from a year ago NASS, U.S. Dept. of Agriculture, Georgia Field Office , Athens, GA 2010 .
Reddiex S. J Wratten S. D Hill G. D Bourdôt G. W and Frampton C. M Evaluation of mechanical weed management techniques on weed and crop populations New Zealand Plant Prot. 2001 54 : 174 – 178 .
SAS Institute , The SAS system for Windows. v. 9.2 SAS Inst. , Cary, NC 2009 .
Smith R Lanini W. T Gaskell M Mitchell J Koike S. T and Fouche C Weed management for organic crops 2000 Davis: University of California. ANR Publication 7250 .
[UGA] University of Georgia , 2009 Peanut Update [Online] 2010 UGA College of Agricultural and Environmental Sciences, Athens, GA. Available at http://www.caes.uga.edu/commodities/fieldcrops/peanuts/2009peanutupdate/index.html (verified 6 Oct. 2011) .
[USDA] United States Department of Agriculture , Organic foods production act of 1990 [Online] 2005 Title XXI of the Food, Agric., Conservation, and Trade Act of 1990 (Public Law 101–624). USDA, Washington, D.C. Available at http://www.ams.usda.gov/AMSv1.0/getfile?dDocName=STELPRDC5060370&acct=nopgeninfo (verified 6 Oct. 2011) .
Wilcut J. W Wehtje G. R and Walker R. H Economics of weed control in peanuts (Arachis hypogaea) with herbicides and cultivations Weed Sci. 1987 35 : 711 – 715 .
Notes
- Dept. of Crop and Soil Sciences, University of Georgia, Coastal Plain Experiment, Station, Tifton, GA 31793-0748
- USDA - ARS, Coastal Plain Experiment Station, Tifton, GA 31793-0748
- Dept. of Agricultural and Applied Economics, University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793-0748
- Dept. of Plant Pathology, University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793-0748
- Experimental Statistics, University of Georgia, Griffin, GA 30223-1797 *Corresponding author: D.Q. Wann (E-mail: dylan.wann@ttu.edu)
Author Affiliations