Introduction
Peanut grown in the U. S. are increasingly being produced using conservation tillage practices (Sholar et al. 1995). The traditional advantages of conservation tillage peanut production are less soil erosion and improved water infiltration compared to conventional tillage systems (Tubbs and Beasley 2009). Of practical significance to peanut growers in the southeastern U. S., conservation tillage offers savings over conventional tillage by eliminating the need for multiple tillage operations in rapid succession which can be complicated by logistics, weather delays, shortages in skilled agricultural labor, and increasing fuel costs (Tubbs and Gallaher 2005). Furthermore, conservation tillage is a critical factor that lessens incidence and severity of spotted wilt (tomato spotted wilt tospovirus) (Brown et al. 1999; Johnson et al. 2001; Marois and Wright 2003) and early leaf spot (Cercospora arachidicola Hori.) (Cantonwine et al. 2007a, 2007b) in peanut.
There are many conservation tillage variants used in the southeastern U. S. peanut producing region, with strip-tillage being the most common. Cereal cover crops are seeded the previous autumn and killed with a non-selective postemergence herbicide in early spring. Seedbeds are prepared using an implement with in-row sub-soil shanks, multiple gangs of fluted coulters to cut cover-crop debris, and ground-driven crumblers that till a band approximately 30 cm wide. Crops are seeded with planter units either tandem mounted on the strip-tillage implement or as a separate operation.
Residual control of annual grasses and small-seeded broadleaf weeds in strip-tillage peanut typically included either pendimethalin or ethalfluralin combined with a second application of a non-selective herbicide applied immediately after seeding peanut. This system has been shown to be an effective component in an integrated system to manage annual grasses (Grichar 2005; Johnson et al. 2002, 2010) and pigweed (Amaranthus spp.) (Grey and Wehtje 2005). Dinitroaniline herbicides are potentially adsorbed by cover crop debris (Helling 1976; Weber 1990) and it was speculated that this phenomenon was the primary reason for reduced efficacy in strip-tillage peanut production (Johnson et al. 2002).
Current surveys show that annual grasses continue to be among the most common and troublesome weeds of peanut (Webster 2009), with Texas millet [Urochloa texana (Buckl.) R. Webster], several species of crabgrass (Digitaria spp.), and crowfootgrass [Dactyloctenium aegyptium (L.) Willd] commonly found throughout the southeastern U. S. Despite the regular incidence of annual grasses in peanut, well documented potential yield losses (Johnson and Mullinix 2005), and need for an aggressive control strategy (Johnson et al. 2002; Prostko et al. 2001), dinitroaniline herbicides are often applied long after planting in strip-tillage production systems. Johnson et al. (2010) determined that either imazapic or paraquat must be tank-mixed with pendimethalin if applications were delayed later than peanut emergence. While no visual injury was reported from delayed applications of pendimethalin, it was beyond the scope of those studies to closely investigate the effects of delayed applications of pendimethalin on peanut growth and yield. Therefore, trials were initiated to study the effects of delayed applications of the dinitroaniline herbicides ethalfluralin and pendimethalin on peanut growth, development, and yield.
Materials and Methods
Irrigated, weed-free field studies were conducted in 2004 and 2005 at the Coastal Plain Experiment Station Ponder Farm, near Ty Ty, GA. The soil was a Tifton loamy sand; 88% sand, 6% silt, and 6% clay with 0.5% organic matter. This soil type is representative of soils in the southeastern U. S. peanut production region.
The experimental design was a randomized complete block with a factorial arrangement of treatments replicated four times. Treatments were all possible combinations of two dinitroaniline herbicides and seven application timings. Dinitroaniline herbicides evaluated were pendimethalin4 (1.1 kg ai/ha) and ethalfluralin5 (0.8 kg ai/ha), each applied at rates registered for use on peanut. Application timings for each herbicide were preemergence (PRE) immediately after seeding peanut, vegetative emergence (VE) of peanut, 1wk after VE (VE+1wk), VE+2wk, VE+3wk, VE+4wk, and a nontreated control. Herbicides were applied with a tractor-mounted CO2-pressurized plot sprayer calibrated to deliver 234 l/ha at 207 kPa using flat-flan nozzle tips. All plots were irrigated with sprinkler irrigation (1.0 to 1.8 cm) immediately after herbicide application to ensure maximum herbicide performance.
Plots were seeded with rye at 63 kg/ha using a grain drill (18 cm spacing) in the autumn after the preceding crop harvest. In early April, the rye cover was killed with glyphosate at 1.1 kg ai/ha. Seedbeds were formed with a two-row strip-tillage implement6 that prepared a tilled seedbed 30 cm wide and planted to peanut with vacuum planters7 in a separate operation. ‘C99R’ peanut (Gorbet and Shokes 2002) were seeded in early May each year at a rate of 112 kg/ha. After seeding peanut, the entire experimental area was treated with paraquat (0.6 kg ai/ha) alone to control emerged weeds. Plots were maintained weed-free throughout the season with one application of bentazon (1.1 kg ai/ha) plus 2,4-DB (0.3 kg ai/ha). An additional treatment of clethodim (0.14 kg ai/ha) was used to control escaped annual grasses. Other than herbicide treatments, crop and pest management practices were based on recommendations by the University of Georgia Cooperative Extension Service (Prostko 2009).
Parameters measured were visual estimates of crop stunting, vegetative biomass, pod biomass, and yield. Visual estimates of crop stunting were made 78 and 85 days after emergence (DAE) in 2004 and 2005, respectively. Vegetative and pod biomass samples were collected from 1 m of row, partitioned into pods and foliage, dried at 38°C for 72 hours, and weighed. Mid-season biomass samples were collected 68 and 71 DAE while late-season biomass samples were collected at 120 and 110 DAE in 2004 and 2005, respectively. Peanut yields were collected from the remainder of the plot by digging, inverting, and combining peanut using commercial two-row equipment. Yield samples were mechanically cleaned to remove foreign material. Yields are reported as cleaned, farmer-stock peanut.
Data were analyzed using a mixed-model analysis. Degrees of freedom were partitioned to test singularly and in combination the effects of dinitroaniline herbicides and time of application on peanut growth parameters and yield. Means were separated using Fisher's Protected LSD (P≤0.05).
Results and Discussion
Analysis of variance indicated a nonsignificant year by treatment interaction for all parameters. Therefore, data are combined across years. The same analysis also showed nonsignificant interactions between dinitroaniline herbicides and application timings for all parameters. Therefore, all data are presented as main effects at P≤0.05.
Visual injury
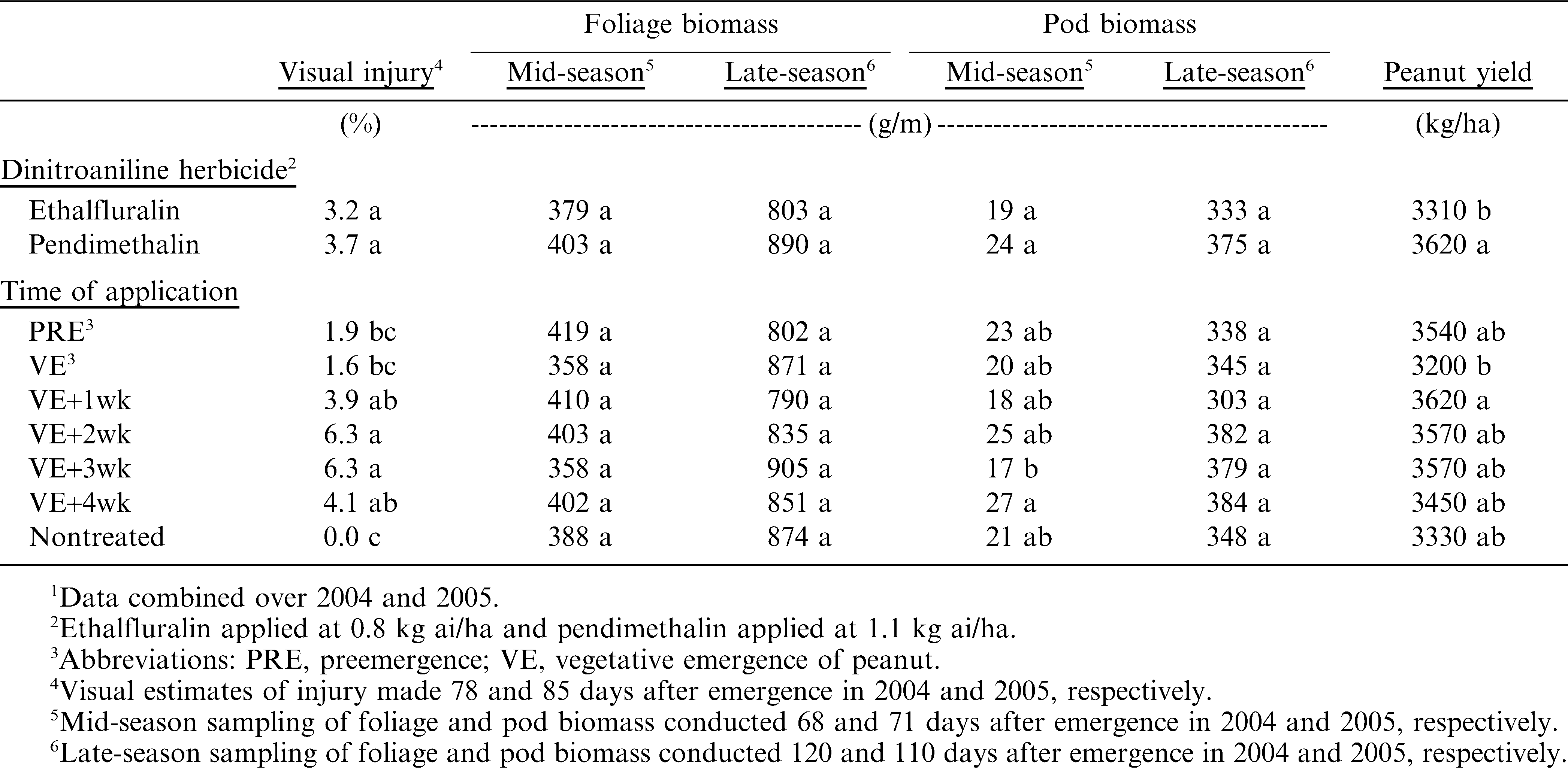
Symptoms of injury were overall stunting of peanut growth compared to the nontreated controls. There was no difference in visual estimates of peanut injury between plots treated with ethalfluralin or pendimethalin, respectively (Table 1). Visual estimates of peanut injury were low, ranging from 3.2% to 3.7% for ethalfluralin and pendimethalin, respectively. Visual injury from dinitroaniline herbicides was significant when herbicides were applied VE+1wk or later, with visual estimates of injury ranging from 1.6% to 6.3% among the application timings (Table 1). Compared to the nontreated control, peanut treated with dinitroaniline herbicides applied at PRE or VE were not affected.
Foliage biomass
Across all times of herbicide application, neither ethalfluralin nor pendimethalin affected peanut foliage biomass when measured mid- or late-season (Table 1). This is consistent with estimates of visual injury that showed no difference between peanut treated with ethalfluralin and pendimethalin. Similarly, mid- and late-season foliage biomass was not affected by herbicide application timings, which was not consistent with estimates of visual injury. A possible explanation is that the estimates of visual injury were generally low (1.6% to 6.3%) and statistical differences among treatments may be of minimal practical significance. Furthermore, this discrepancy may also be due to accuracy differences between directly measuring peanut vegetative growth using foliage biomass and subjectively estimating visual injury.
Pod biomass
When averaged across all possible application timings, peanut pod biomass measured mid- and late-season was not affected by the dinitroaniline herbicides evaluated in these trials (Table 1). However, times of herbicide application affected pod biomass when measured mid-season. Dinitroaniline herbicides were most inhibitory of mid-season pod biomass when applied at VE+3wk. However, pod bimass of peanut treated with dinitroaniline herbicides at VE+3wk was not different from the nontreated control and from peanut treated from PRE through VE+2wk. There is no obvious explanation for this inconsistency in mid-season pod biomass response to delayed applications of dinitroaniline herbicides. Furthermore, late-season pod biomass was not affected by times of dinitroaniline herbicide application.
Peanut yield
When combined across all possible application timings, peanut treated with ethalfluralin had lower yields than those treated with pendimethalin (Table 1). This is not consistent with earlier studies that showed no phytotoxicity from ethalfluralin (Johnson and Mullinix 1999; Johnson et al. 1997). Those trials were in conventional tillage peanut production systems, with comparisons between preplant incorporated and PRE applications at increasing rates. When averaged across dinitroaniline herbicides, peanut yields also differed among application timings. The lowest peanut yields were in plots treated with dinitroaniline herbicides applied at VE; however those yields did not differ from the nontreated control. The yield response is also inconsistent when compared to mid-season pod biomass. Mid-season pod biomass was reduced when peanut were treated at VE+3wk (Table 1). However, yield of peanut treated VE+3wk with dinitroaniline herbicides was among the greatest of all possible times of herbicide application. We do not have an explanation for the inconsistent results between mid-season pod biomass and peanut yield.
Peanut yield response to ethalfluralin was not expected. Earlier trials showed peanut to be very tolerant of ethalfluralin applied PPI or PRE at increasing rates (Johnson and Mullinix 1999; Johnson et al. 1997). Those trials were in conventional tillage systems and at recommended application timings. In the present trials, the lack of significant interaction between dinitroaniline herbicides and application timings eliminates the possibility that ethalfluralin was unusually injurious to peanut compared to pendimethalin when applied later than PRE. Perhaps in strip-tillage systems the presence cover crop debris differentially influences the availability of ethalfluralin and pendimethalin causing ethalfluralin to be more injurious to peanut. That theory will require subsequent research.
Delayed applications of dinitroaniline herbicides do not appear to be overly phytotoxic to peanut based solely on visual estimates of stunting or other measurements of vegetative growth response. When pod formation and development are considered, the potential exists for subtle losses when dinitroaniline herbicides applications are delayed. The slight discrepancies between mid-season pod biomass response and final peanut yield make it difficult to predict incidence of this phenomenon and identify the risky application timings. However, all data in these trials clearly show that there are no phytotoxicity concerns if dinitroaniline herbicides are applied PRE in strip-tillage production systems (Table 1). A prudent interpretation of these data is that any delayed application of either ethalfluralin or pendimethalin has the potential to inhibit pod formation and reduce peanut yield. Earlier companion studies that investigated the effects of delayed applications of pendimethalin on weed control (Johnson et al. 2010) showed that delayed applications required the addition of either paraquat or imazapic to achieve adequate weed control. It is evident that delayed applications of dinitroaniline herbicides in strip-tillage peanut are not a wise production decision and every effort must be made to apply these herbicides PRE, immediately after seeding the crop.
Acknowledgements
We acknowledge the technical contributions of Daniel R. Evarts and Andy M. Hornbuckle toward the success of these experiments.
Literature Cited
Brown S. , Todd J. , Culbreath A. , Baldwin J. , and Beasley J. 1999 Tomato spotted wilt virus of peanut: Identifying and avoiding high risk situations. Univ. of Georgia. Coop. Ext. Serv. Bull 1165 .
Cantonwine E. G. , Culbreath A. K. , and Stevenson K. L. 2007a Effects of cover crop residue and preplant herbicide on early leaf spot of peanut. Plant Dis 91 : 822 – 827 .
Cantonwine E. G. , Culbreath A. K. , and Stevenson K. L. 2007b Characterization of early leaf spot suppression by strip tillage in peanut. Phytopathology 97 : 187 – 194 .
Gorbet D. W. and Shokes F. M. 2002 Registration of ‘C-99R’ peanut. Crop Sci 42 : 2207 .
Grey T. L. and Wehtje G. R. 2005 Residual herbicide weed control systems in peanut. Weed Technol 19 : 560 – 567 .
Grichar W. J. 2005 Using herbicides in a peanut strip-tillage production system. Crop Management. http://www.plantmanagementnetwork.org/pub/cm/research/2005/peanut/. (last accessed 12 August 2010).
Helling C. S. 1976 Dinitroaniline herbicides in soils. J. Environ. Qual 5 : 1 – 15 .
Johnson W. C. and Mullinix B. G. 2005 Texas panicum (Panicum texanum) interference in peanut (Arachis hypogaea) and implications for treatment decisions. Peanut Sci 32 : 68 – 72 .
Johnson W. C. and Mullinix B. G. 1999 Peanut seedling response to dinitroaniline herbicides applied preplant incorporated and preemergence. Peanut Sci 26 : 18 – 23 .
Johnson W. C. , Brenneman T. B. , Baker S. H. , Johnson A. W. , Sumner D. R. , and Mullinix B. G. 2001 Tillage and pest management considerations in a peanut-cotton rotation in the southeastern coastal plain. Agron. J 93 : 570 – 576 .
Johnson W. C. , Colvin D. L. , Wehtje G. R. , Littlefield T. A. , and Mullinix B. G. 1997 Peanut response to ethalfluralin: rates and methods of application. Peanut Sci 24 : 101 – 104 .
Johnson W. C. , Prostko E. P. , and Mullinix B. G. 2010 Annual grass control in strip-tillage peanut production with delayed applications of pendimethalin. Weed Technol 24 : 1 – 5 .
Johnson W. C. , Prostko E. P. , and Mullinix B. G. 2002 Texas panicum (Panicum texanum) control in strip-tillage peanut (Arachis hypogaea) production. Peanut Sci 29 : 141 – 145 .
Marois J. J. and Wright D. L. 2003 Effect of tillage system, phorate, and cultivar on tomato spotted wilt of peanut. Agron. J 95 : 386 – 389 .
Prostko E. P. 2009 2009 peanut update. Univ. of Georgia Coop. Ext. Serv. http://www.caes.uga.edu/commodities/fieldcrops/peanuts/2009peanutupdate/index.html (last accessed 12 August 2010).
Prostko E. P. , Johnson W. C. , and Mullinix B. G. 2001 Annual grass control with preplant incorporated and preemergence applications of ethalfluralin and pendimethalin in peanut (Arachis hypogaea). Weed Technol 15 : 36 – 41 .
Sholar J. R. , Mozingo R. W. , and Beasley J. P. 1995 Peanut cultural practices. pp. 354 – 382 In. Pattee H. E. and Stalker H. T. (eds.) Advances in Peanut Science Amer. Peanut Res. Educ. Soc Stillwater, OK .
Tubbs R. S. and Beasley J. P. 2009 2009 peanut update - tillage and row pattern update. Univ. of Georgia Coop. Ext. Serv. http://www.caes.uga.edu/commodities/fieldcrops/peanuts/2009peanutupdate/tillage.html (last accessed 12 August 2010).
Tubbs R. S. and Gallaher R. N. 2005 Conservation tillage and herbicide management for two peanut cultivars. Agron. J 97 : 500 – 504 .
Weber J. B. 1990 Behavior of dinitroaniline herbicides in soils. Weed Technol 4 : 394 – 406 .
Webster T. M. 2009 Weed survey - southern states. Proc. So. Weed Sci. Soc 62 : 509 – 524 .
Notes
- Research Agronomist, USDA-ARS, Coastal Plain Experiment Station, P. O. Box 748, Tifton, GA 31793. [^]
- Professor, Univ. of Georgia, Dep. Crop and Soil Sci., Univ. of Georgia, P. O. Box 748, Tifton, GA 31793. [^]
- Agricultural Research Statistician, Univ. of Georgia, Georgia Experiment Station, Griffin, GA 30224. [^] *Corresponding author's email: Carroll.Johnson@ars.usda.gov
- Prowl® 3.3EC; BASF Corporation; 26 Davis Drive; Research Triangle Park, NC 27709. [^]
- Sonalan® HFP; Dow AgroSciences LLC; Indianapolis, IN 46268. [^]
- Two row strip-tillage implement, Kelley Manufacturing Company, 80 Vernon Drive, Tifton, GA 31793. [^]
- Vacuum planter, ATI, Inc. 17135 West 116th St., Lenexa, KS 66219. [^]
Author Affiliations
Footnotes