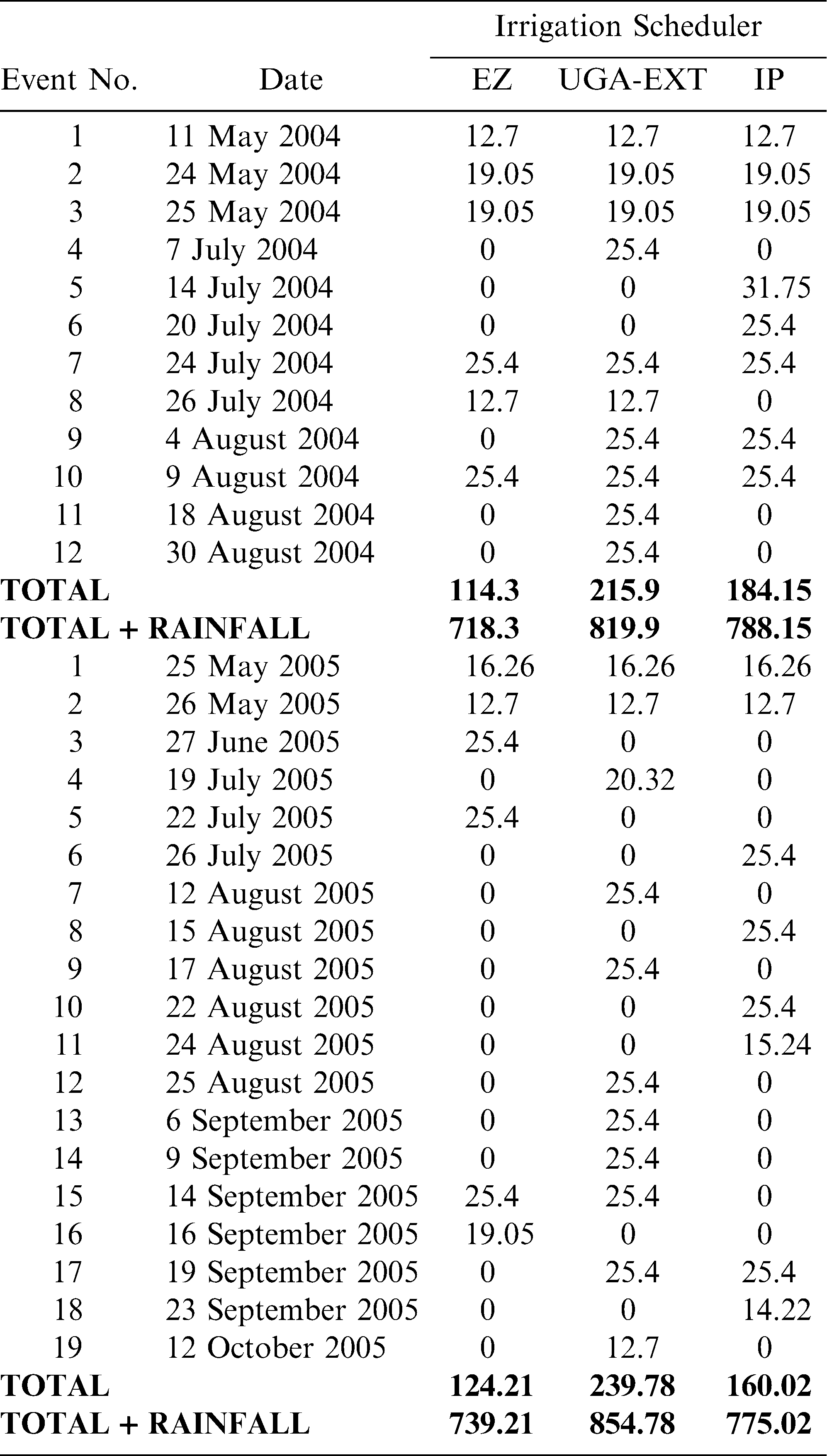Introduction
While all eyes in crop production research are typically focused on assessing, manipulating, and propagating variability in yield among crop cultivars, all too often very little is known about cultivar differences in growth habit, phenology, and ontogeny that are causal agents of that yield variability. This is certainly the case among today's cultivated peanut (Arachis hypogaea L.) genotypes where almost no information is available regarding cultivar differences in aboveground growth habit, leaf size, flowering potential, tissue nutrient contents, harvest index, nor on the differences in development of these traits through the season. On the surface it may appear that this information has relatively little value to either scientist or producer. However, in the ever-narrowing gap between economic viability and bankrupted ruin in the U.S. farm environment, these traits can have very significant implications on final peanut production and can increase the chance of economic success. Cultivar choice can be a “make or break” decision for a producer and having information about a cultivar's growth habit can be a critical component of this decision. In today's southeastern U.S. peanut production environment, there are roughly 8–10 cultivars that are available for a producer to choose from. Up front, this means that information regarding variability among cultivars in growth habit and reproduction could help a grower choose cultivars that will yield optimally under particular field conditions. Beyond these producer benefits, science can also reap the rewards of further information about cultivar variation because it represents essential input into both breeder programs and crop model development.
The existence of peanut cultivar variability in growth and development is questionable because of the increasingly narrow gene pool foundation of current peanut germplasm as a result of industry and grower preferences. The cultivars used in the southeastern U.S. peanut production are almost solely runner market types because of the preference of the processing industry towards their shelling, size, and flavor characteristics. In addition, there is an absolute necessity for the sole use of Tomato spotted wilt virus resistant runner peanut genotypes, often derived from similar parental germplasm, because of the devastating economic effects of this plant virus. Both of these conditions have significantly narrowed the genetic background and variability among peanut cultivars making them genetically quite similar in many ways (Kottapalli et al., 2007). Therefore, any variation among current cultivars is essential to document and quantify.
Of course environmental growth conditions can have an impressive influence on the expression of inherent genetic differences in peanut cultivars. One such influence is the application of supplemental irrigation during the growing season. Nearly 50% of U.S. peanut production is under irrigation and this percentage is expected to expand as the global climate becomes warmer and droughts become more frequent and severe in many of these production regions (Lamb et al., 2004). Surprisingly, within the southeast U.S., irrigation is vital to production of economically sustainable yields despite the widely presumed image of the area as receiving adequate rainfall. Although irrigation can increase yields by as much as 50% (Lamb et al., 1997; Lamb et al., 2004), skyrocketing fuel prices ($30.00 U.S. per hectare – Nathan Smith pers. comm.) necessitate that irrigation application be as efficient as possible or it will quickly lose its cost effectiveness.
Currently there are three established and utilized irrigation scheduling methods for peanut: 1) Irrigator Pro (IP), 2) the EASY Pan (Evaporation-based Accumulator for Sprinkler-enhanced Yield - EZ), and 3) the University of Georgia's Extension check book method (UGA-EXT). Irrigator Pro is based on monitoring soil temperatures and incrementing water application with crop growth stage (Lamb et al., 1993; Davidson et al., 1998). The EASY Pan method uses a galvanized washtub and float with different sized mesh screening over the top of the tub to simulate evaporation losses affected by different levels of crop cover (Thomas et al., 2004). The University of Georgia's Extension checkbook method is an incrementally increasing model linked to crop stage and has now been updated by J.P. Beasley (unpublished data). To date, no studies have actually compared the efficacy of these methods on peanut production. Variation among peanut cultivars as to how they respond to irrigation scheduling methods is most likely due to: 1) differences in growth habits; 2) variation in the time period required for fruit maturation; and 3) genetic differences in water use strategies and water-use efficiency (Rowland and Lamb, 2005). Irrigation scheduling method could have critical effects on the plasticity of these traits that translate into long-lasting effects on final crop yield.
The current study was aimed at documenting and quantifying any variability among several currently grown peanut cultivars in traits that could have significant impact on physiological functioning of the crop and thus influence yield. Secondly, the differential effects on peanut development of the three most commonly used irrigation scheduling methods (IP, EZ, and UGA-EXT) were tested. The following specific objectives were addressed: 1) is there any variation in morphology, ontogeny, and reproduction among currently grown peanut genotypes? and 2) what is the effect of irrigation scheduling method on these traits, especially on carbon isotope discrimination as a representation of seasonal water-use efficiency?
Materials and Methods
Field Preparation and Management
The experiment was carried out at the University of Georgia's C.M. Stripling Irrigation Research Park in Camilla, GA during the 2004 and 2005 growing seasons. Soil was classified as a Lucy loamy sand (loamy, kaolonitic, thermic, Arenic, Kandiudults). Soil samples were collected in the previous fall seasons and commercial lime was added according to extension service recommendations to modify soil pH by planting. Also during the previous fall seasons, the field was chiseled and cultivated prior to planting a rye cover crop (Secale cereale L. cv Wrens' Abruzzi). This cover crop was then terminated approximately 60 days prior to planting in both years using glyphosate (Roundup®, Monsanto Co., St. Louis, MO, USA). Just prior to planting peanut, the field was strip tilled using a KMC (Kelley Manufacturing Co., Tifton, GA) strip till unit with double coulters.
Peanut was planted on 11 May 2004 and 24 May 2005. Peanut rows were planted in a twin row design with an intra-row plant distance of 18 cm and seed spacing within each twin approximately 5 cm using a Monosem planter. Seed were treated with Rhizobium inoculant prior to planting and phorate insecticide was applied in the row during planting for early season thrips control. Separate peanut genotypes were planted in plots consisting of two twin rows spaced 91 cm apart and 16.8 m in length. Plots were arranged in a randomized split plot with irrigation scheduling methods as main plot factors and cultivars as subplot factors Eight peanut genotypes were tested in both 2004 and 2005: AP-3 (Gorbet, 2007), Carver (Gorbet, 2006), Tifrunner (Holbrook and Culbreath, 2007), C-99R (Gorbet and Shokes, 2002), Georgia-01R (Branch, 2002), Georgia-02C (Branch, 2003), Georgia-03L (Branch, 2004), and Georgia Green (Branch, 1996). These cultivars differed in relative maturity level: AP-3 (142–147 days after planting – DAP), Carver (135–140 DAP), Tifrunner (149–154 DAP), C-99R ( 149–154 DAP), Georgia-01R (149–154 DAP), Georgia-02C (149–154 DAP), Georgia-03L (135–140 DAP), and Georgia Green (135–140 DAP).Peanut genotypes were subjected to three different irrigation scheduling methods currently utilized in peanut production in the southeastern USA: 1) Irrigator Pro (IP); 2) the UGA EASY Pan (EZ); and 3) the UGA extension check book method (UGA-EXT). When each method called for water application, irrigation was applied through a lateral move irrigation system with drop down nozzles. The crop was maintained using recommended applications of herbicides and fungicides during the year. The crop was harvested on 17 September and 8 October in 2004 and 30 September and 24 October in 2005 based on the maturity level of the developing pods (Williams and Drexler, 1981).
Plant Collection and Analysis
Leaf Area Index (LAI) measurements and plant collections were conducted on the following dates in 2004: 15 June, 12 July, and 17 August; and in 2005: 23 June, 1 August, and 6 September. LAI measurements were taken using an LAI-2000 meter (LI-COR Biosciences, Lincoln, NE, USA) across a single row in each plot, using one above canopy measurement and four below canopy measurements spaced in line with the row and every 23 cm away from that point for a total area coverage of 91 cm. Within each genotype X irrigation scheduling method plot, one second nodal position leaf was collected on the main apex stem. Just after excision, chlorophyll content was measured using the Minolta SPAD (Soil-Plant Analyses Development Unit, Minolta Corp., Ramsey, N.J., U.S.A.) chlorophyll meter directly after removal from the plant. The SPAD chlorophyll meter measures absorbance by plant tissues of wavelengths in the visible spectrum and serves as a measure of the relative internal concentration of chlorophylls a and b. One SPAD chlorophyll reading was taken on each of the four leaflets, avoiding the midrib, and then averaged for one chlorophyll reading per plant to correct for possible non-homogeneous distribution of chlorophyll throughout the leaf (Monje and Bugbee, 1992). After collection of the second nodal leaf, the entire plant was dug up leaving the upper root system intact. Tetrafoliate leaves and whole plants were then placed on ice and refrigerated at 4 C until further analysis. A second intact plant was collected and analyzed for: percent N, P, K, Mg, Ca, S, and ppm of B, Zn, Mn, Fe, and Cu (Waters Laboratory, Camilla, GA).
In the laboratory, whole plants were examined and measured for: total number of live flowers, total number of pegs, total number of pods, and internode length on the apex stem. The plants were then divided into leaves, internodes, and pods. Sampled second nodal leaves were hydrated in distilled water for at least three hours prior to leaf area measurement in order to bring them all to a standardized turgor level (Nageswara Rao et al., 2001). Leaflets were removed from each petiole and the leaf area of the four leaflets was measured with an LI-3000A leaf area meter (LI-COR Inc., Lincoln, NE, U.S.A.) and summed to give total leaf area. Whole plant biomass components and second nodal leaves were oven dried at 60 C for 72 hours and weighed. Leaves were then fine ground using a Braun ® (model KSM2) coffee grinder and analyzed for carbon isotope composition (δ13C), δ15N, %C, and %N. Specific leaf area (SLA) was calculated as the ratio of leaf area to leaf dry weight. Harvest index on whole plants was calculated as the ratio of pod weight to the sum of pod, internode, and leaf weights.
Isotope Analysis
In both years leaf samples from each peanut genotype and irrigation scheduling method were collected for analysis of the following traits: δ13C, δ15N, percent carbon (%C), percent nitrogen (%N) and C/N ratio from four replications in 2004 and three in 2005. One second nodal apex leaf was collected from each plot. Sampling was completed in a single day and within the morning hours (800–1200 EDT). In both years, peanut leaf tissue was collected approximately 90 days after planting. This phenological period is associated with the highest ribulose bisphosphate carboxylase (rubisco) levels and concomitantly the highest photosynthetic levels of the season thereby ensuring isotopic differences among genotypes would be most evident (Nageswara Rao and Wright, 1994; Nageswara Rao et al., 1995). Tissue collection was standardized to the second nodal apex position on leaves that had relatively no insect or disease damage. The tetrafoliate second nodal leaf was excised, placed on ice, and refrigerated at 4 C until further analysis.
Isotopic composition of the leaf samples was analyzed at the Colorado Plateau Stable Isotope Laboratory, Department of Biological Sciences, Northern Arizona University. Samples of the ground leaves (2 mg, +/− 0.2 mg) were weighed, sealed in capsules and, along with standards, loaded into the elemental analyzer autosampler (a “Zero Blank” autosampler from Costech Analytical Technologies in Valencia, CA). Samples and standards were combusted in the elemental analyzer (Carlo Erba NC2500 elemental analyzer coupled with a Thermoquest Finnigan Delta plus isotope ratio mass spectrometer). Lab standards, which were calibrated against internationally distributed isotope standards, were analyzed at regular intervals throughout the sample runs. The resulting N2 and CO2 gases (along with isotopic reference gases for N2 and CO2) were admitted to the mass spectrometer via Finnigan's Conflo II interface. Data were collected and processed by Finnigan's Isodat software. Sample results are based on one analysis per sample (δ13C, δ15N, %N and %C were all determined with the same analysis). Isotope results are reported in delta notation vs. Air (for nitrogen) and vs. PDB (for carbon) in permil. Stable carbon isotope composition was expressed as δ13C where δ13C (‰) = [(R sample/R standard)-1] × 1000, and where R is the 13C/12C ratio. Composition of 13C/12C (δ13C) rather than discrimination of 13C (Δ) is reported due to the possible differences in atmospheric components linked to natural or man-made C emissions between seasons.
Data Analysis
Statistical analyses were performed using JMP SAS (SAS 1997). Factorial analysis of variance (ANOVA) was used to determine the effect of year, period within the season (early, mid, and late), cultivar, irrigation scheduling method, and all possible two-way interactions on growth, tissue nutrient, and reproductive traits. Factorial ANOVA was also used to determine the effect of year, cultivar, irrigation scheduling method, and all two-way interactions on isotope, C, and N composition of leaf tissue collected at the optimal physiological time period. Differences among multiple levels of a given factor were determined using a Tukey's HSD multiple comparisons test. Pearson product-moment correlations were used to determine the relationship between late season pod number and pod weight with growth, nutrient, and reproductive characteristics. For those traits showing significant correlation with either late season pod number of pod weight, a stepwise regression analysis with forward selection was performed individually to determine the predictive value of those correlated traits.
Results
Environmental Conditions
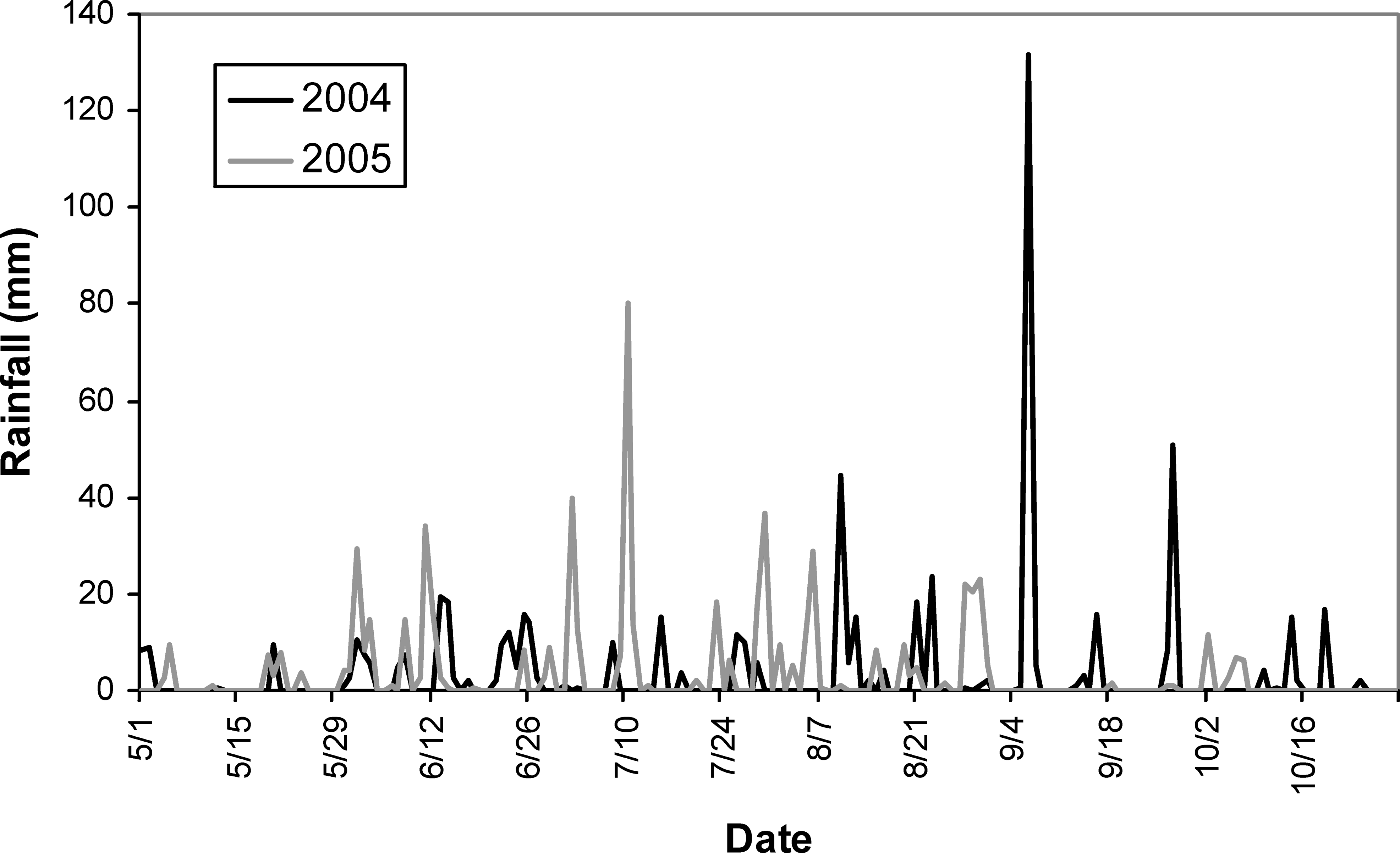
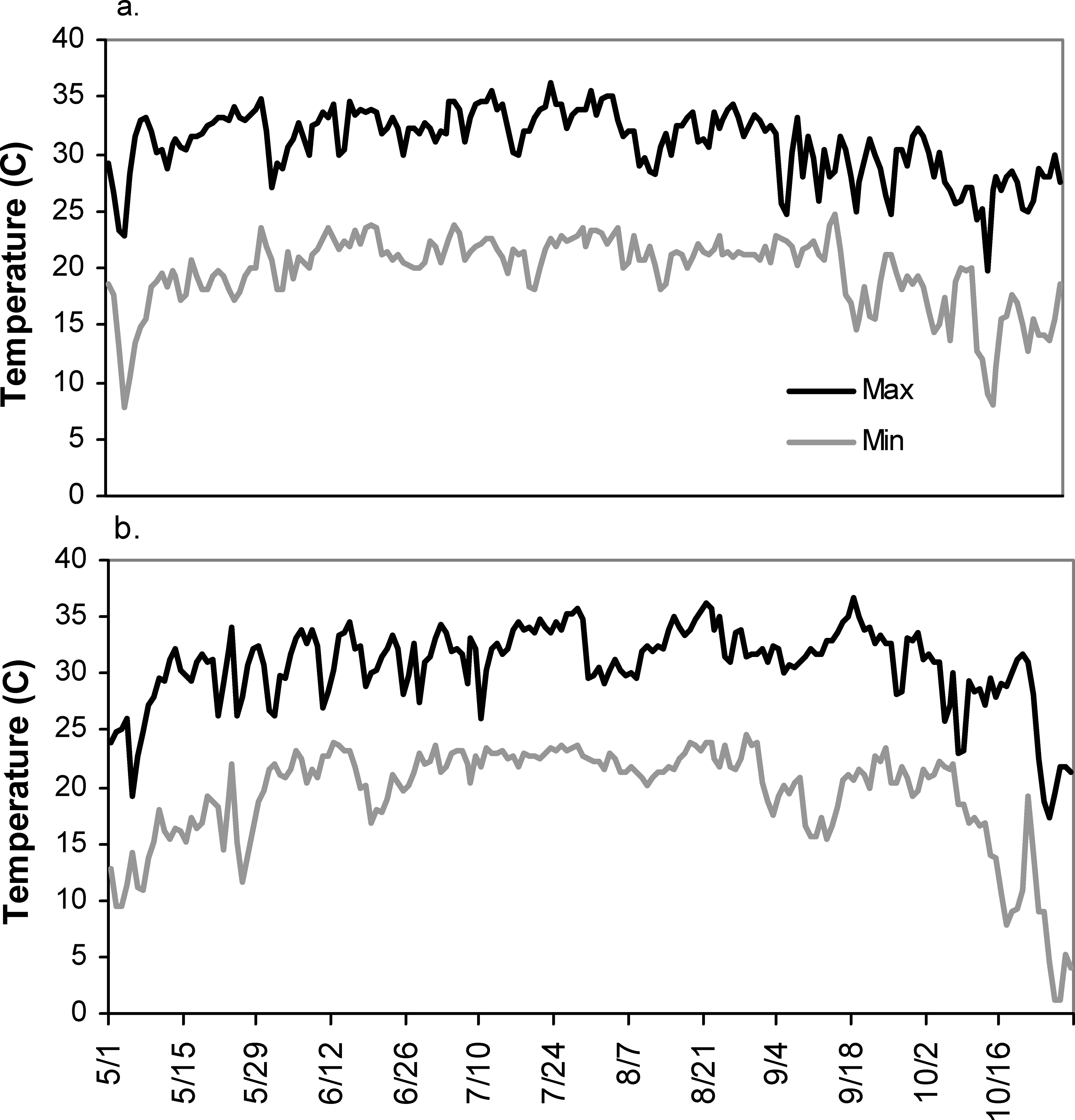
Total irrigation applied in each irrigation scheduling method differed among methods with the greatest water amounts applied for the UGA Extension method in both 2004 and 2005 respectively (216 mm, 240 mm), followed by the Irrigator Pro method (184 mm, 160 mm), and least for the EZ Pan method (114 mm, 124 mm) (Table 1). Differences in total rainfall during the growing season between years were minimal (604 mm in 2004 and 615 mm in 2005). However, seasonal patterns of rainfall for the growing seasons of 2004 and 2005 did differ (Figure 1; Georgia Weather Net; http://www.georgiaweather.net/). Most notably, the rainfall received during the critical periods of peak flowering and fruit set/maturation (June–August) was 315 and 541 mm in 2004 and 2005, respectively. Maximum ambient temperatures were similar in 2004 and 2005 across the growing season and predominantly hovered between 30 and 35 C with lower maximums both early (1 May–15 May) and late in the season (1 October - harvest) (Figure 2; Georgia Weather Net; http://www.georgiaweather.net/). Minimum temperatures were also similar for both years with the predominant range between 17 and 23 C.
Growth, Nutrient, and Reproductive Traits
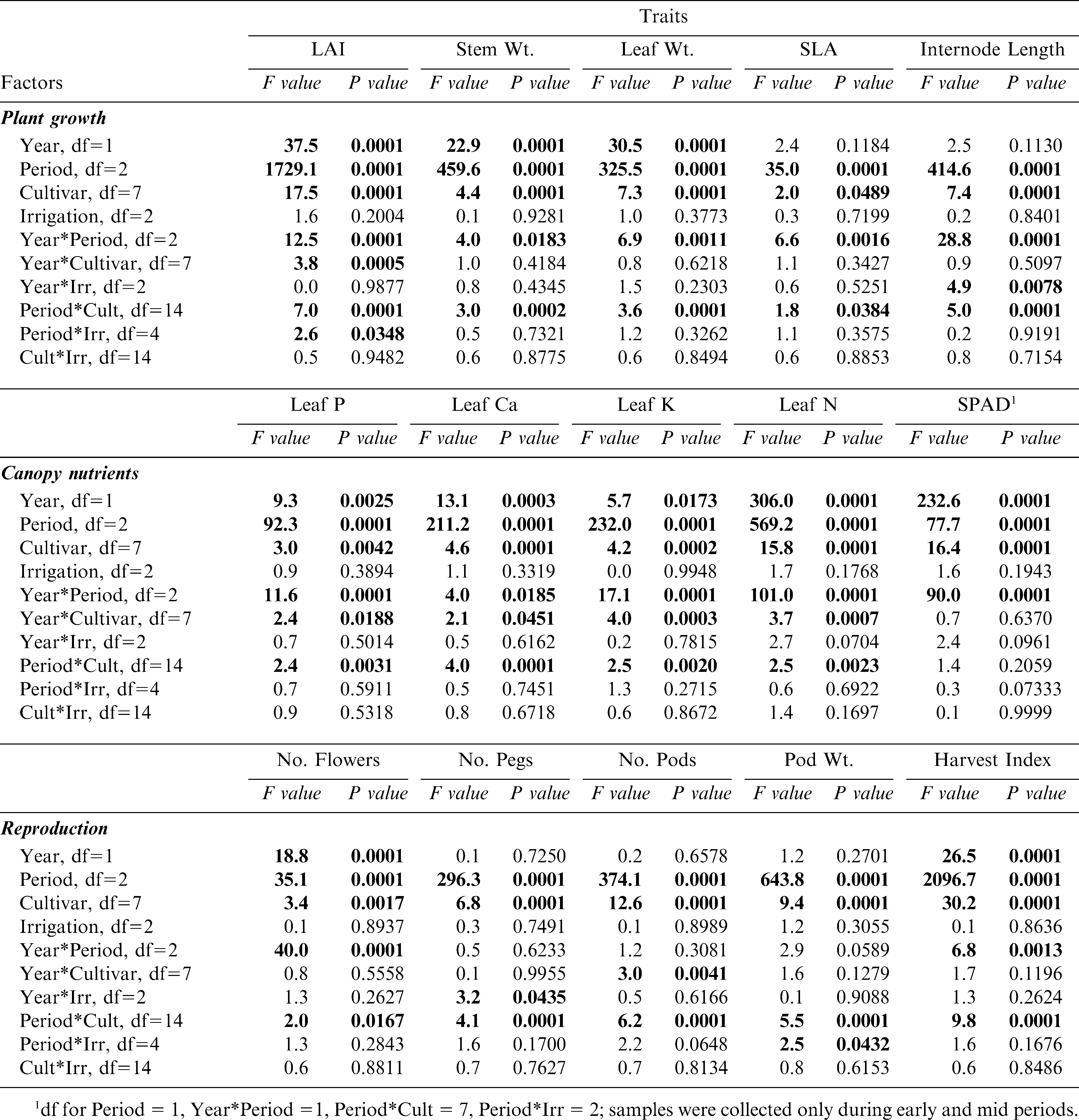
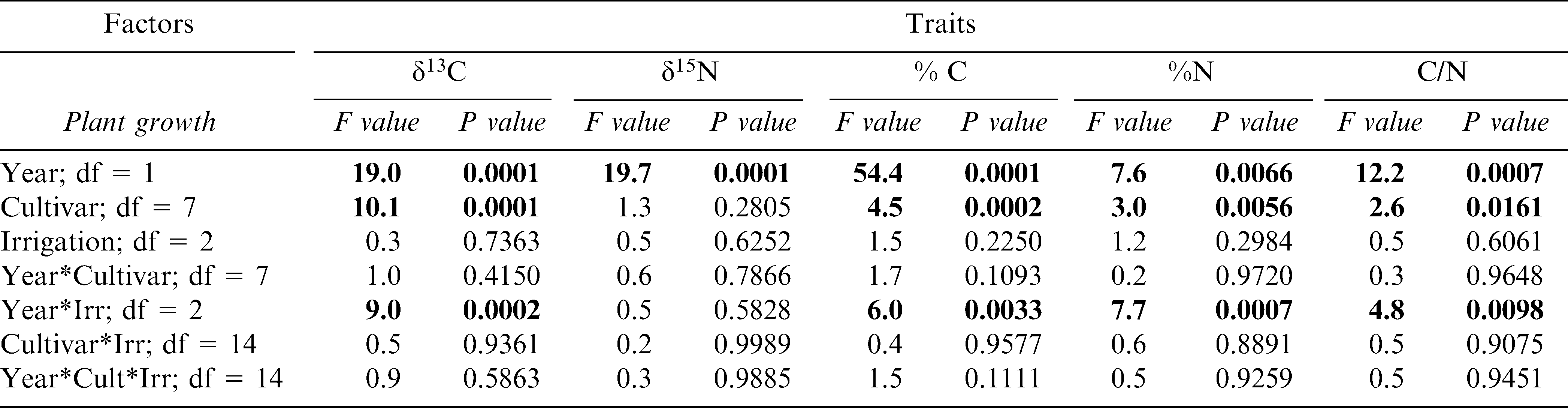
ANOVA results for growth, tissue nutrient, and reproductive traits showed significant differences in most traits between 2004 and 2005 and period within the growing season during these years. (Table 2). Significant year differences illustrate possible annual differences in climatic patterns (likely rainfall during fruit initiation and maturation), while period differences indicate the obligate changes in phenology as the crop develops. Less predictable were the significant differences among peanut cultivars in all of the measured traits, indicating the presence of inherent genetic differences in growth, nutrient, and reproductive characteristics despite the often assumed lack of variability among cultivated peanut germplasm. The lack of significant interactions between year and cultivar for most of the plant growth (except LAI) and reproductive (except number of pods) traits indicated that the environment may have had minimal impact on changing these inherently genetic traits. Irrigation scheduling method showed a lack of effect across the board among all measured traits, including no significant interactions between irrigation scheduling method and cultivar (Table 2).
ANOVA for the growth traits of 8 peanut genotypes that were grown in both 2004 and 2005 at the Stripling Irrigation Research Park; F-values and P-values given. Factors include year (2004, 2005), period during the growth season (early, mid, and late), peanut cultivar, and irrigation scheduling method (EZ, UGA-EXT, and IP). Statistically significant effects are listed in bold type.
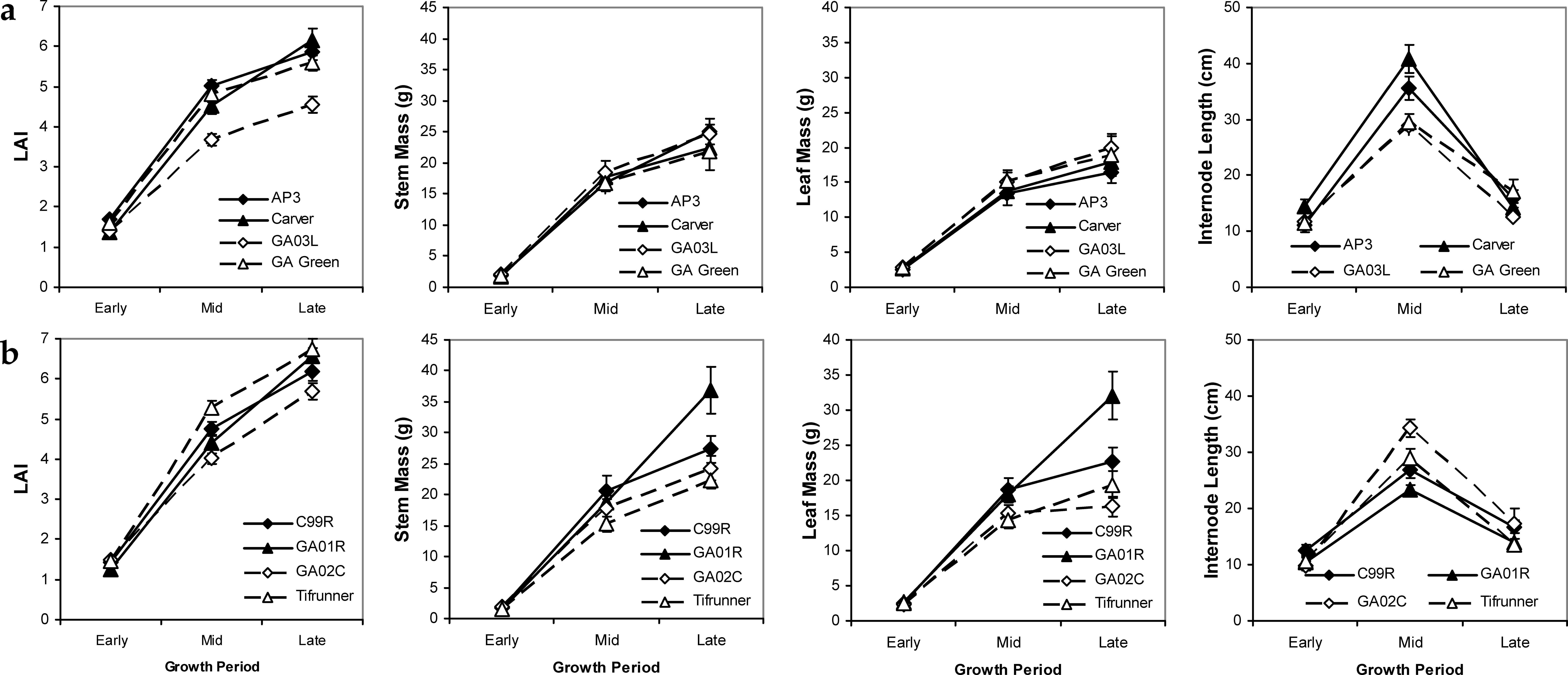
Patterns in growth trait means across the season in 2004 and 2005 indicate some differences in growth and reproduction between mid- and late-maturing cultivars. The late-maturing cultivars (C99R, GA01R, GA02C, Tifrunner) on average had higher maximum values of LAI, stem mass, and leaf mass measured in the late growth period, with the Georgia-01R cultivar having the highest values overall (Figure 3). The larger LAI of late-maturing cultivars was due in part to the larger late season production of stems and leaves over mid-maturing cultivars. The overall pattern across cultivars showed a customary linear increasing pattern for LAI, stem mass, and leaf mass as the season progressed and as plant size increased. Contrastingly, internode length peaked in mid-season and declined into late season, likely indicating a shift in allocation patterns from stems to leaves and reproductive structures. Trade-offs between allocation patterns to different tissue types revealed interesting variability in growth strategies among cultivars. For example, the cultivar Georgia-01R had a large biomass allotment to stems and leaves, but maintained short internode lengths, thereby maintaining a mid-level LAI. In contrast, the cultivar Carver had high LAI values probably attributed to its large internode lengths; while the relatively small LAI values for Georgia-03L were linked to its short internode lengths (Figure 3).
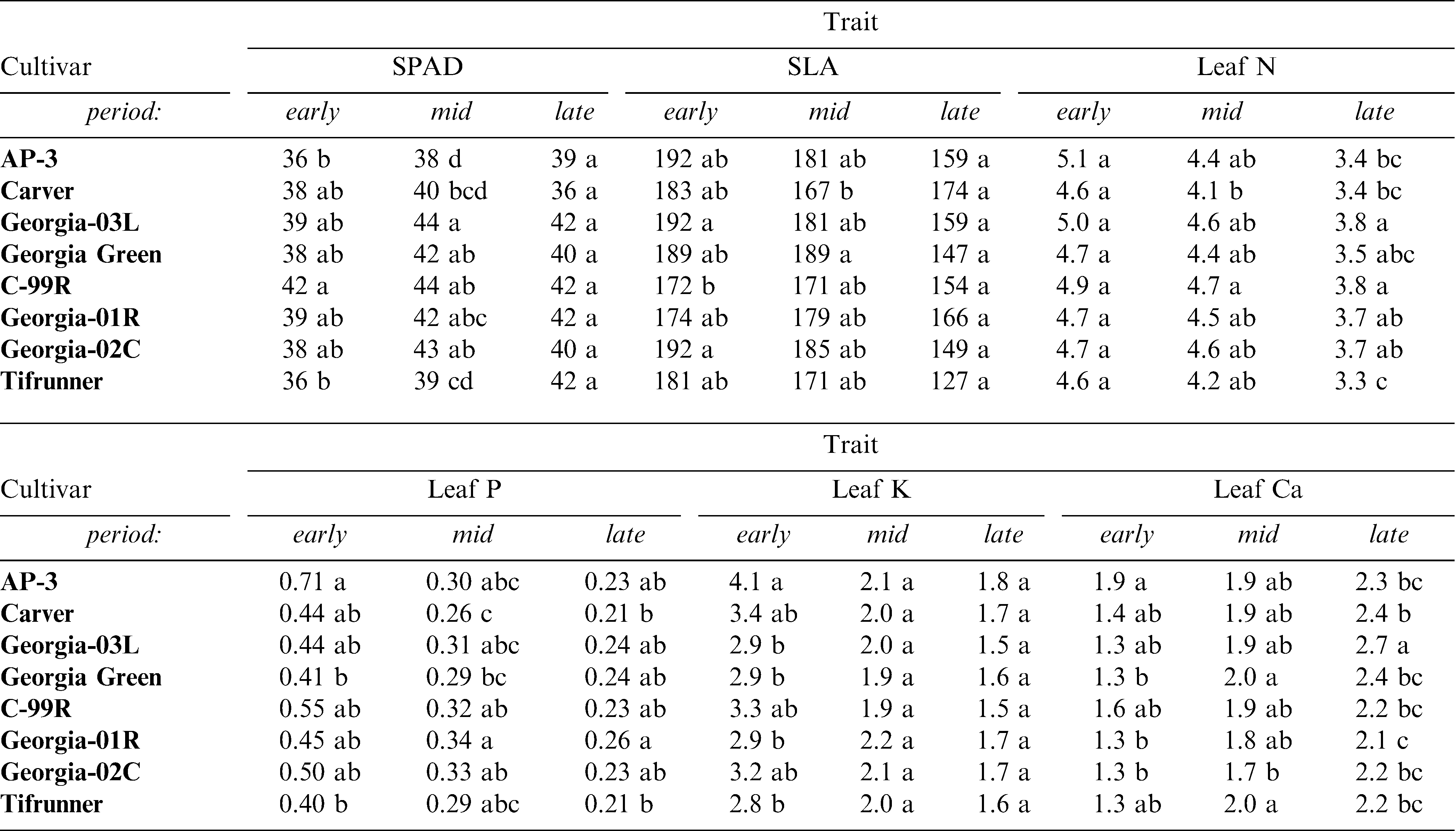
Tissue nutrient levels showed minimal variability among cultivars with higher differences occurring among periods during the growing season (Table 3). SPAD chlorophyll content showed an increasing accumulation as the season progressed. Late-maturing cultivars appeared to accumulate chlorophyll somewhat faster than mid-maturing genotypes and variation in SPAD chlorophyll was maximal at mid-season indicating a difference among cultivars in their ability to accumulate chlorophyll during the early and peak reproductive periods. The patterns in SPAD levels did not reflect leaf nitrogen as would be expected. Across all cultivars, mean N was highest in the early season and lowest during the late season (Table 3).
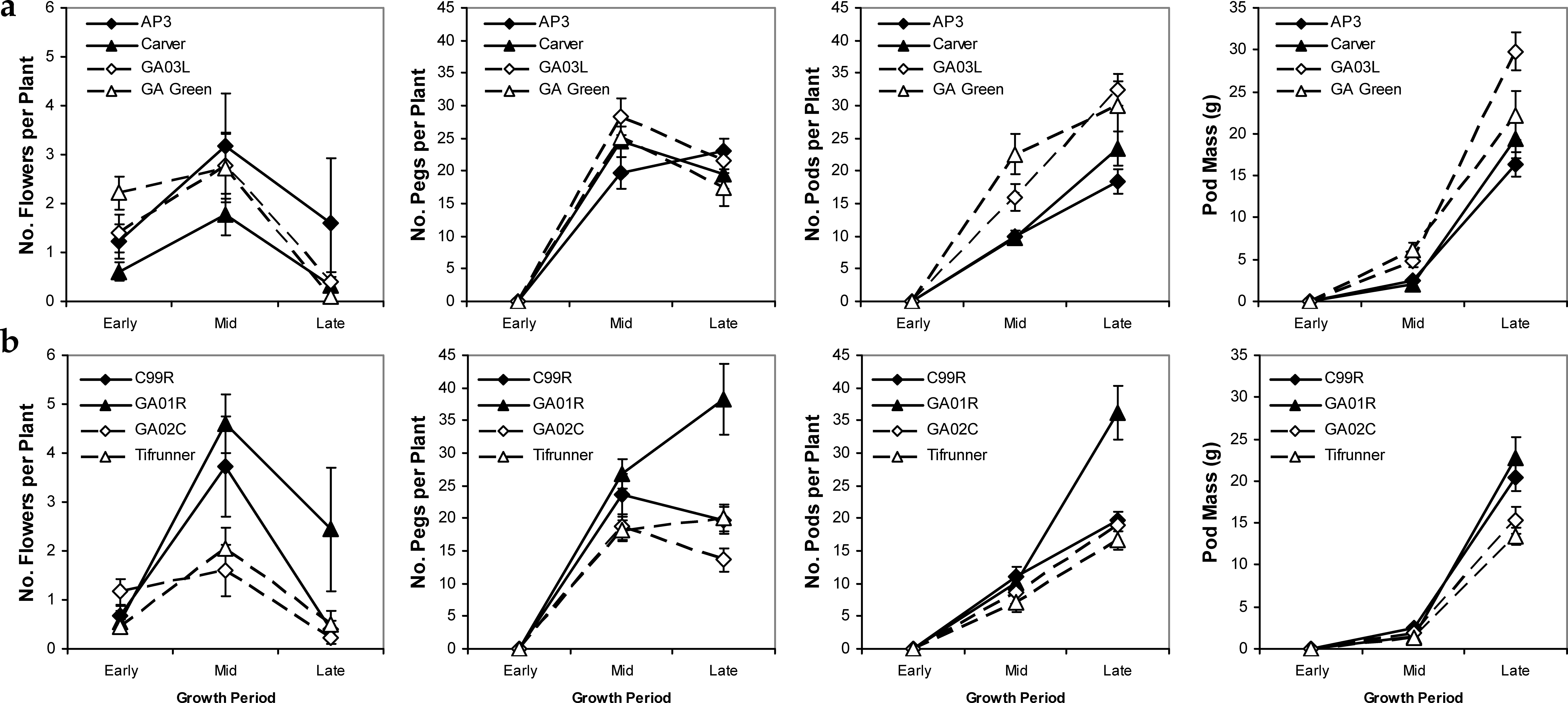
Reproductive characters showed variability among cultivars that was likely genetically based because patterns of variation were similar across years (data not shown) and irrigation scheduling method had no significant effect within years. Flower and peg production peaked in mid season for most cultivars except for AP-3 (mid-maturing) and Georgia-01R (late-maturing) which showed maximum peg number per plant at the late season period, while number of pods per plant showed a positive linear pattern and pod mass an exponentially increasing pattern across cultivars as the season progressed (Figure 4). Similarly, Georgia-01R had a high number of flowers and a higher number of pegs and pods per plant than all of the other cultivars except Georgia-03L for pegs and Georgia Green, Georgia-03L, and Carver for pods. Pod weight showed slightly different patterns of variability among cultivars: Georgia-03L had the heaviest pods, while the high peg and pod producer, Georgia-01R had only medium weight pods significantly lighter than Georgia-03L but significantly heavier than Tifrunner only (Figure 4). Patterns of conversion from flowers to pegs and eventually pods can be ascertained by examining the relative numbers of each consecutive tissue type. High “efficiency” of conversion of flowers into pegs and pegs into pods could be seen in the cultivar Carver which had low numerical flower production but mid numbers of pegs and pods in the late season. Alternatively, low “efficiency” of tissue conversion could be seen in the cultivar AP-3 which had the highest numerical flower counts per plant at mid-season among mid-maturing cultivars but the lowest numerical pod count per plot in the late season indicating that many of its flowers were never converted to pegs and pods. Overall high numbers of flowers, pegs, and pods throughout the season were evident in the cultivars Georgia-01R and Georgia-03L cultivars, with overall low counts of these tissues evident in Tifrunner (Figure 4).
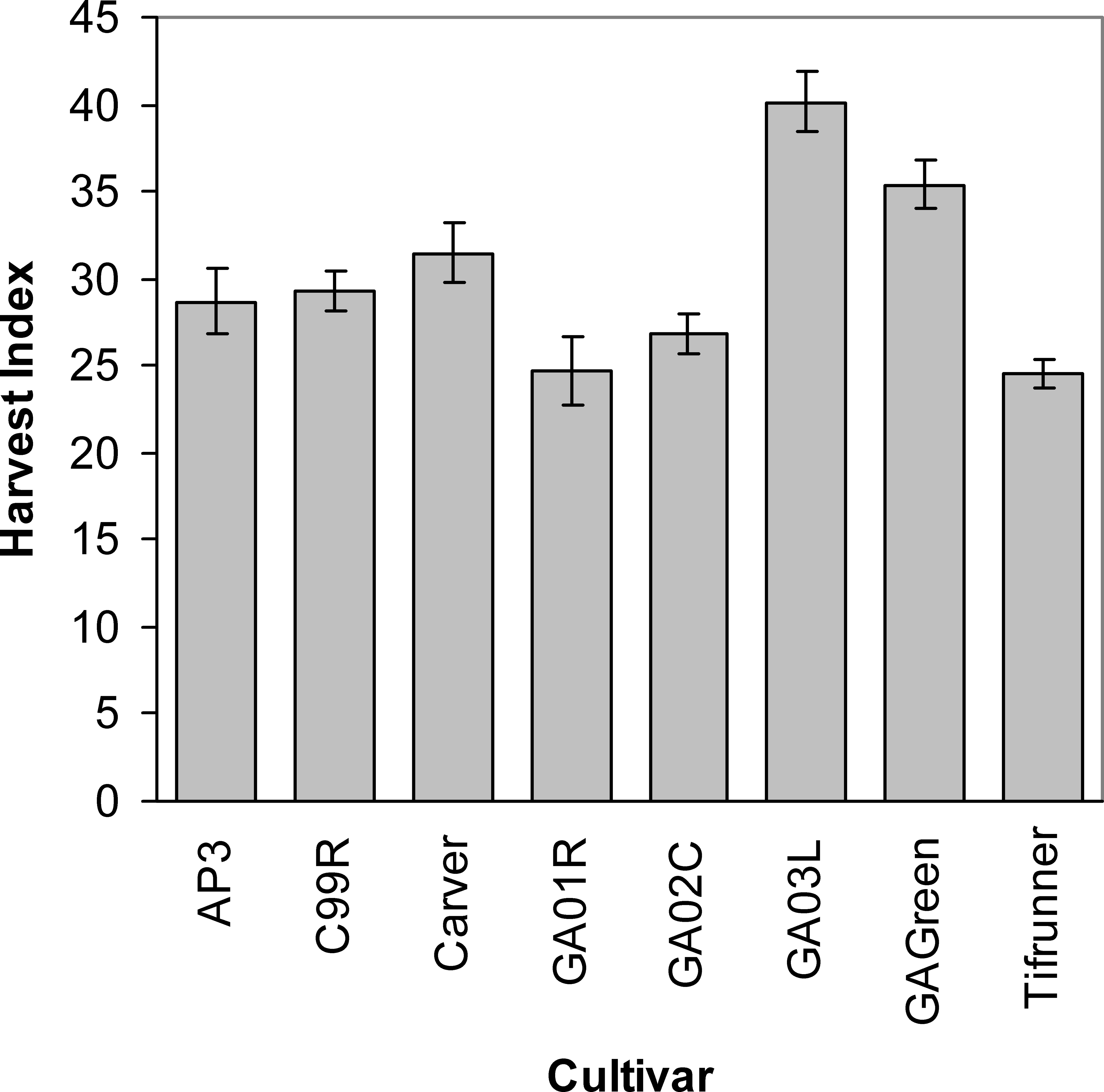
Differences among cultivars in the efficiency of tissue assimilation and conversion to yield were illustrated in the variability in harvest index at the end of the season (Figure 5). The cultivar Georgia-03L had the highest harvest index value of any of the cultivars except Georgia Green. Interestingly, it appears that the low harvest indices seen in Tifrunner and Georgia-01R resulted from two different partitioning strategies: Tifunner had overall low leaf and stem mass coupled with low pod mass while Georgia-01R had overall high leaf and stem mass coupled with high pod mass. The resulting ratios produced the same relative low harvest index. In contrast, the high harvest index of Georgia-03L was a consequence of modest leaf and stem production coupled with high pod mass in relation to the other cultivars.
Isotopes
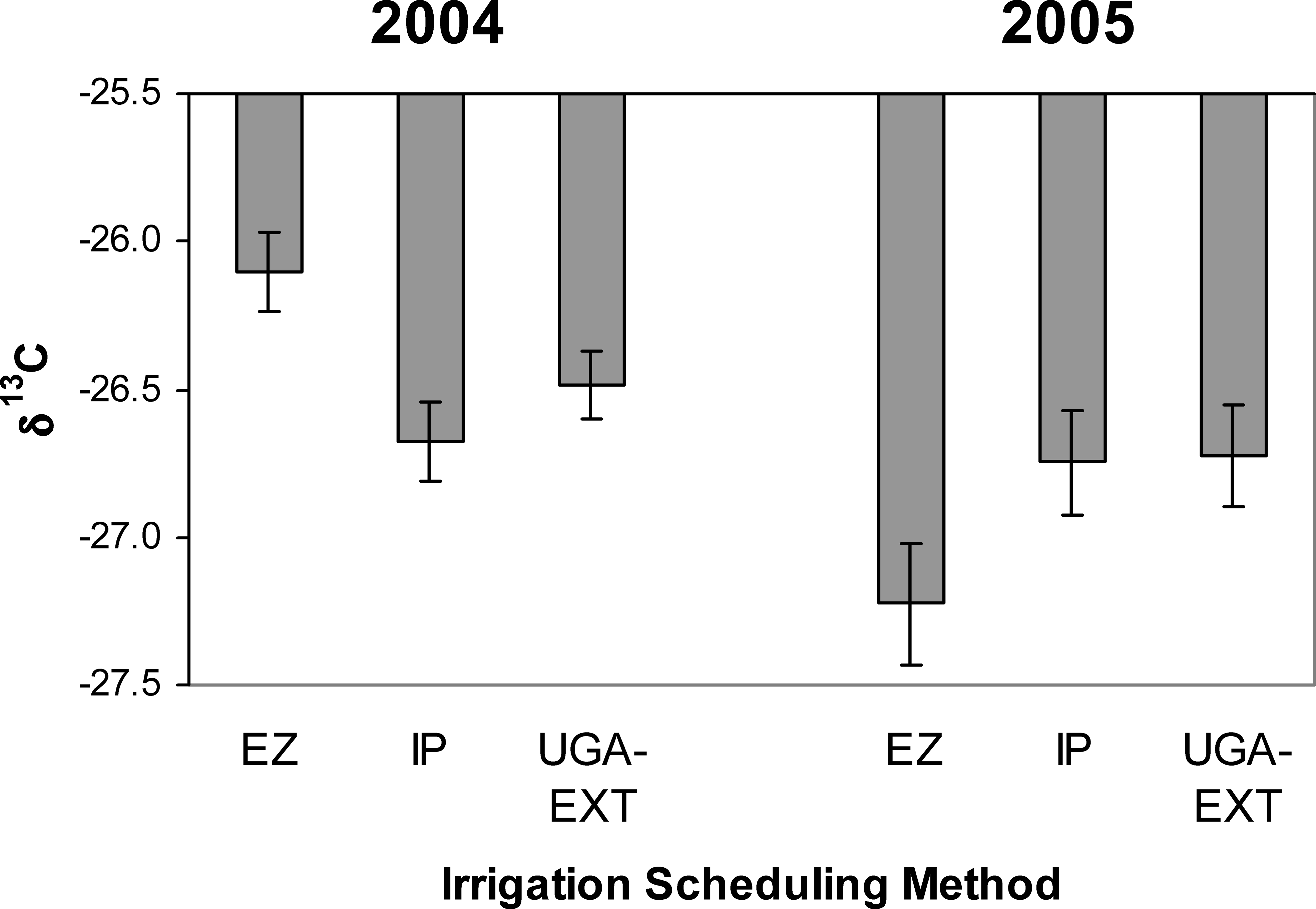
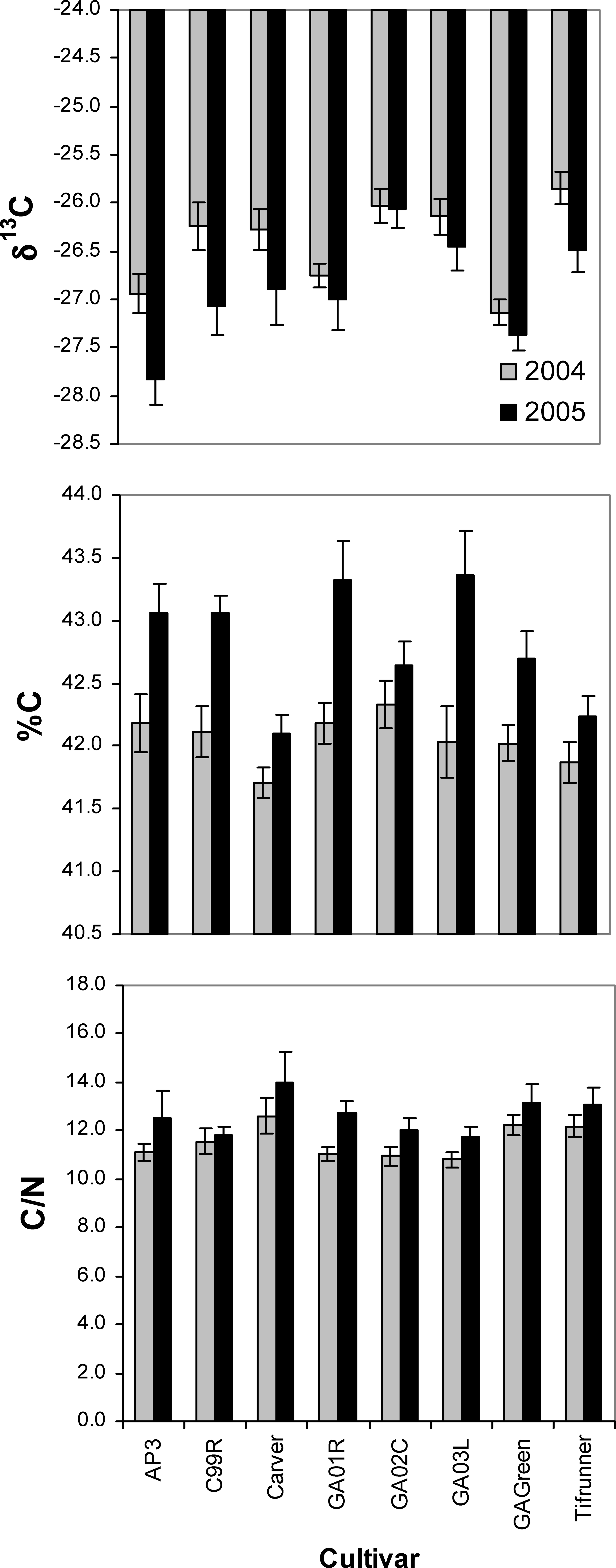
Isotope analysis measured at the time period when physiological assimilation is maximal showed a strong year effect similar to that experienced by the growth, reproductive, and tissue traits (Table 4). Also similar to these other traits, isotope composition was not affected by irrigation scheduling method. However, year did have a significant interaction with irrigation scheduling method for all the traits except δ15N. For δ13C, this interaction was likely significant because the isotopic composition in the EZ treatment responded differently among years; EZ plants were more water-use efficient in 2004 but showed the lowest efficiency among the scheduling treatments in 2005 (Figure 6). For %C, %N, and C/N ratio the significant interaction between year and irrigation scheduling method was driven by different directional responses between years in the IP treatment (data not shown). Among cultivars, Georgia-02C and Tifrunner had significantly higher δ13C (and thus water-use efficiency) than Georgia-01R, GA Green, and AP-3 across years; AP-3 had the lowest levels of leaf δ13C than any of the cultivars except Georgia-01R and Georgia Green (Figure 7).
ANOVA for the leaf carbon and isotope composition of 8 peanut genotypes that were grown in both 2004 and 2005 at the Stripling Irrigation Research Park; F-values and P-values given. Factors include year (2004, 2005), peanut cultivar, and irrigation scheduling method (EZ, UGA-EXT, and IP). Statistically significant effects are listed in bold type.
Correlation and Regression Analyses
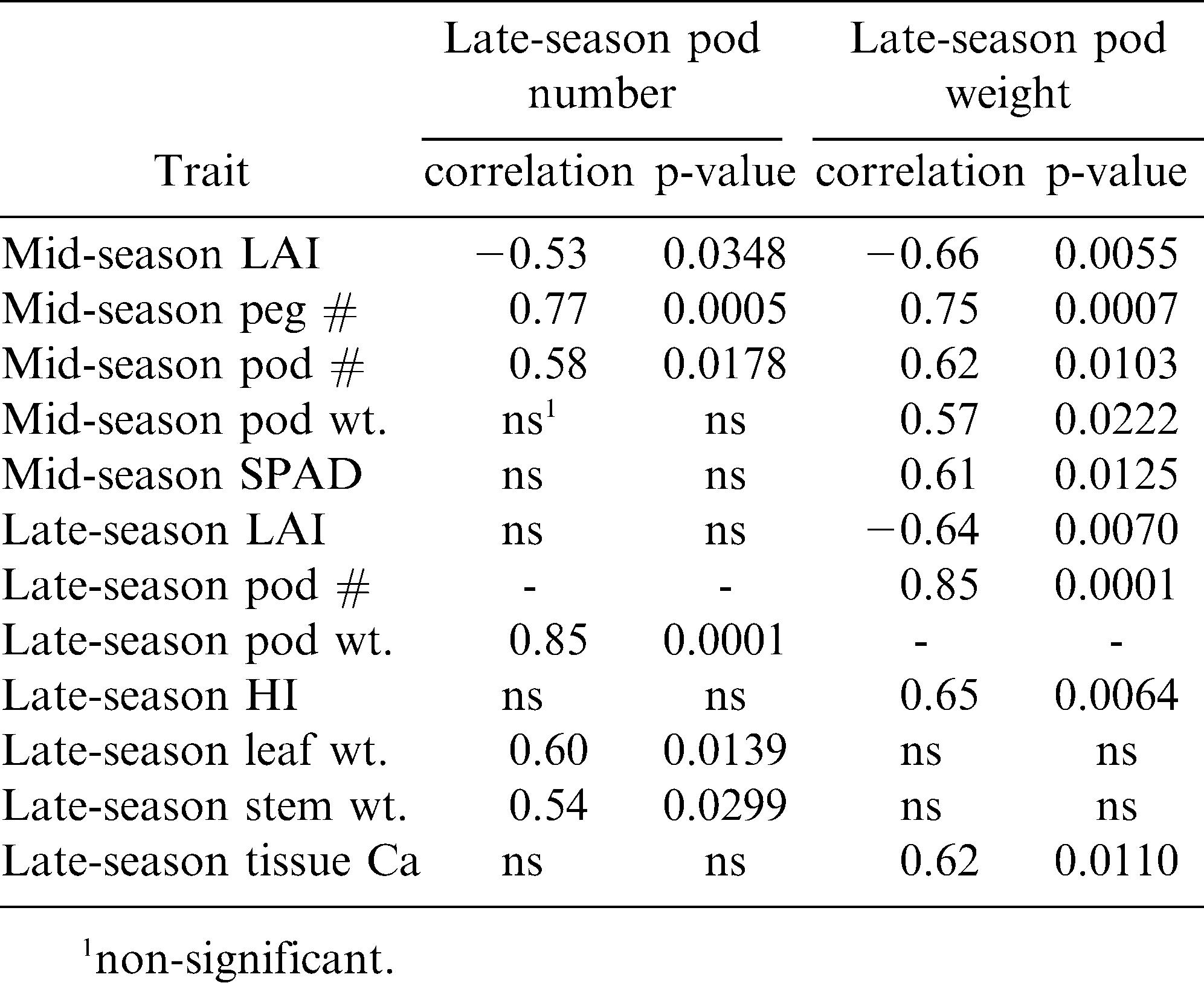
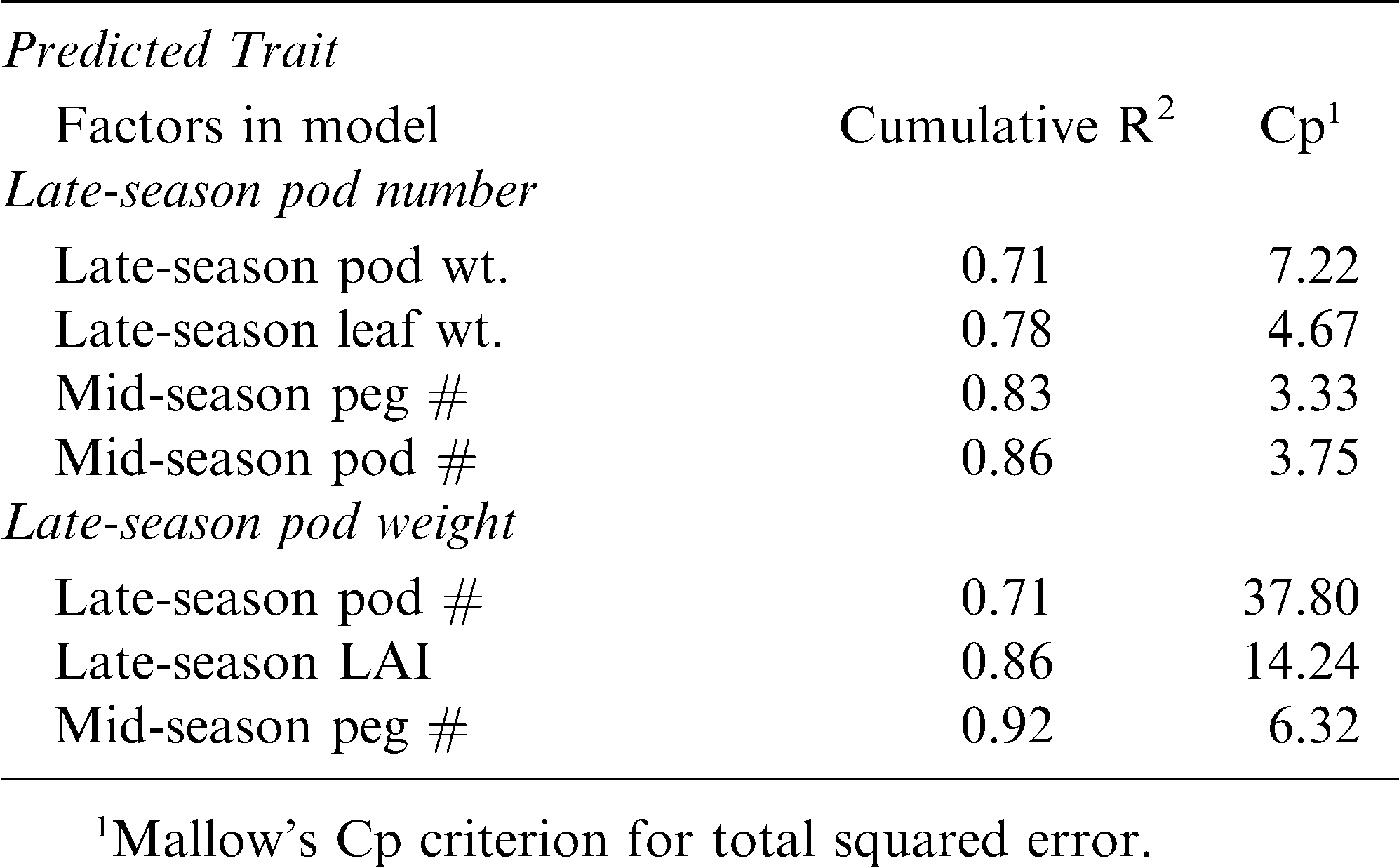
The correlation between pod number and pod weight in the late season was examined for all the growth, reproductive, and nutrient analyses in all three seasonal periods (early, mid, and late) across years and cultivars to determine the possible causal and predictive strength of these measured traits. Across all these traits, late-season pod number was significantly correlated only with mid-season LAI, peg number per plant, and pod number per plant, and late-season pod weight per plant, leaf weight per plant, and stem weight per plant (Table 5). Likewise, late-season pod weight per plant was correlated with mid-season LAI, peg number, pod number, pod weight, and SPAD, and late-season LAI, pod number, harvest index, and tissue Ca. When the respective significant traits were added into stepwise regression models for late-season pod number and for late-season pod weight separately, there were even fewer traits that were predictive of reproductive output . For late-season pod number, the following traits were significant in the model and resulted in a cumulative R2 value of 0.86: late-season pod weight and leaf weight, and mid-season peg number and pod number (Table 6). Similarly, the following traits were significantly predictive of late-season pod weight with a cumulative R2 value of 0.92 for the model: late-season pod number and LAI, and mid-season peg number.
Discussion
There is true value in studies that examine growth and developmental differences among crop cultivars because they describe genetic variability that can either be utilized in breeding programs or crop models (Bell et al., 1991b; Bell et al., 1993a; Craufurd et al., 2002; Baterng et al., 2003; Kiniry et al., 2005). For breeding programs, a description of the differences among cultivars is essential for exploiting available genetic variability during the development of new germplasm. For crop modelers, knowledge of how cultivars can differ in their growth and reproductive responses can be essential to the success of the model's predictive ability. For example, the crop model PNUTGRO requires over 40 cultivar-specific inputs for successful simulation of growth and yield in peanut (Hammer et al., 1995). Previous work has shown that there is variability in growth and reproduction in peanut but comparisons have typically been made among widely different peanut species and botanical types (Bell et al., 1991a,b; Bell et al., 1993a,b; Bell et al., 1994; Craufurd et al., 2002; Banterng et al., 2003). But the existence of variability among current peanut cultivars whose genetic pedigrees have become increasingly similar due to the type of traits that have been favored for several years is questionable at best. However, this study has documented and quantified several important growth and reproductive characteristics that differ among currently cultivated peanut genotypes. It also appears that there are differences among cultivars in resource allocation strategies for canopy development that can translate into effects on pod production.
Despite expectations of a kind of genetic dilution to phenotype variability among cultivars, genotype differences were quite strong for growth traits. Previously documented cultivar differences in leaf weight and harvest index do exist, but again these were in widely diverse botanical types (Bell et al., 1993a; Craufurd et al., 2002; Banterng et al., 2003), making the current study's documentation of growth variability among closely related cultivars unique. Although year had a significant effect on these traits indicating an effect of climate on peanut physiology, there were largely no interactions between year and genotype indicating that the changes caused by climate were relatively low in magnitude and did not alter the overall genetic pattern of development for each cultivar. A lack of an effect of irrigation scheduling method or interaction between irrigation scheduling method and cultivar for growth traits also illustrated the fairly strong genetic control over aboveground biomass development among the cultivars in this study.
Overall, the pattern and magnitude of LAI measurements across the season in this study were typical for other peanut cultivars (Banterng et al., 2003; Kiniry et al., 2005), but some studies have documented a more parabolic pattern for LAI development (Kar and Kumar, 2007) as well. Patterns of growth in the current study indicated that late-maturing cultivars had higher late-season growth than mid-maturing cultivars. Trade-offs in allocation patterns among LAI, stem, and leaf growth indicated cultivars had different strategies for canopy development. For example, although the late-maturing cultivar Georgia-01R had high partitioning to leaves and stems, its short internode lengths maintained a mid-level LAI. Two mid-maturing cultivars alternated between large internode lengths and thus high LAI (Carver) and short internode lengths and low LAI (Georgia-03L). Variability in partitioning strategies was also illustrated by similar harvest indices that were arrived at by coupling low below- and aboveground biomass in Tifrunner, while Georgia-01R coupled high below- and aboveground biomass. Biomass partitioning especially between leaves and stems or between vegetative and reproductive traits can vary among cultivars in other legumes such as cowpea (Vigna unguiculata (L.) Walp.) and chickpea (Cicer arietinum L.), and some diversely related peanut cultivars (Bell et al., 1993b; Bell et al., 1994; Krishnamurthy et al., 1999; Banterng et al., 2003; San José et al., 2004).
Unlike growth traits, the separation among cultivars in reproductive traits was not related to maturity class. On average, mid- and late-maturing cultivars had similar flower, peg, and pod production. With elevated canopy growth but equal reproductive rates, late-maturing cultivars were expected to have lower harvest indices than mid-maturing genotypes. This was certainly the case, especially for the later maturing cultivars Georgia-01R, Georgia-02C, and Tifrunner which had the lowest harvest indices among all eight cultivars. This pattern of lowered harvest indices in late-maturing cultivars indicates a somewhat lower efficiency of yield return for the amount of energy invested in aboveground canopy structures. These results concur with differences in harvest index that have previously been found between peanut cultivars of different maturity classes with late maturing cultivars having low relative harvest indices (Bell et al., 1991b; Bennett et al., 1993). This is true across all grain/seed producing food crops, legumes, or cereals.
Aside from the contribution of genetics, harvest index can be significantly affected by various environmental factors that affect peanut growth and reproduction including: temperature (Vara Prasad et al., 2000); heat stress (Vara Prasad et al., 1999); irradiance and photoperiod (Bagnall and King, 1991a,b); ozone stress (Booker et al., 2007; Burkey et al. 2007); and even planting pattern (Lanier et al., 2004) Of course one of the greatest influences on harvest index can be irrigation amount and timing (Reddy et al., 2003). That is why perhaps the most surprising result from this study was the lack of impact of irrigation scheduling method on peanut growth and reproduction. Even though there was over a 100 mm difference in the total water applied in both years between the top and bottom irrigation scheduling methods, the crop performed well under all systems and was not significantly affected. This is likely due to the fact that all three irrigation scheduling methods in both years provided more than enough water to meet maximal cultivar yield potential. Based on sap flow water-use measurements in southeastern peanut, seasonal water-use can be approximately 710–735 mm (unpublished data). For both 2004 and 2005, water receipt (from both irrigation and precipitation combined) topped these levels indicating that irrigation application could have been reduced while still meeting crop demand.
Although the irrigation environment did not affect growth and reproduction to an appreciable degree, yearly variation did. Looking at the climatic data, the most likely factor was the timing and amount of rainfall, with 2005 having the highest rainfall total during the period of flowering and fruit maturation. However, the crop did not utilize this higher water availability in reproduction (as evidenced by the lack of significance for year in peg number and pod number and weight). The effect of this difference in water application between years was in the growth characteristics; namely stem and leaf weight (Table 2) which were actually lower across cultivars in 2005 (data not shown). Additional water “availability” coupled with lower leaf and stem production might indicate some degree of water over-saturation in the soil. Tissue nitrogen was also lower in 2005 than in 2004 (data not shown) which indicate deleterious effects on nitrogen fixation under hypoxic soil conditions. Either way, water availability appeared to be more than adequate for the crop in both years.
Harvest index measured in these eight cultivars showed relatively low values across years in comparison to 14 studies examining harvest index in peanut reviewed by Kiniry et al. (2005). The low harvest indices in the current study are even more surprising given all treatments were irrigated because harvest index is predicted to be as high as 0.58 under efficient irrigation (Kiniry et al., 2005). This result also provides evidence that all three irrigation scheduling methods used in this test were likely applying too much water to the crop, thereby increasing canopy growth without a concomitant increase in yield and thus leading to low harvest indices. Based on these results, it appears that irrigation scheduling methods for southeastern U.S. peanut have a lot of room to lower total water application during the season and increase efficiency of crop water use.
Variation among cultivars in δ13C showed there is room to develop peanut germplasm with increased seasonal water-use efficiency as well. The larger canopies of late-maturing cultivars necessarily spell a larger area for transpirational water loss. For this reason, it might be expected that water-use would be higher in late- than mid-maturing cultivars and thus lead to decreased water-use efficiency. However, patterns of δ13C did not reflect lower seasonal water-use efficiency for late-maturing cultivars. In fact two of the cultivars exhibiting the highest seasonal water-use efficiencies (Georgia-02C and Tifrunner) were late-maturing cultivars. Seasonal water-use efficiency appears to be under tight genetic control in U.S. peanut cultivars because irrigation scheduling method had no effect on δ13C composition. Isotopes are often affected by irrigation regime, but the current results agree with previous results that found very little effect of irrigation on the isotope composition of southeastern cultivars (Rowland and Lamb, 2005). The only slight indication of irrigation scheduling changing the pattern of peanut water-use was the differing δ13C levels in the EZ method between years. In 2004, the EZ treatment showed much higher water-use efficiency in the crop while in 2005, this trend was opposite. Therefore, irrigation scheduling appears to have the potential to affect crop water-use efficiency but is highly dependent on prevailing climatic conditions.
The existence of varying partitioning strategies among cultivars in this study necessitates the assumption that reproduction might be predicted from patterns in certain growth traits throughout the season. Although an early study indicated that peanut yield was determined more by genetic pedigree than growth habit (Norden and Lipscomb, 1974) more recent studies show a strong link between growth and reproduction for other legumes (Krishnamurthy et al., 1999) and other peanut culitvars (Bennett et al., 1993; Banterng et al., 2003). In the current study, traits relating to canopy development (and thus assimilation capacity) as well as mid-season reproductive potential appeared to be causally linked to final reproductive output in the late season. LAI and leaf weight had some correlative relationships with the yield-determining traits of pod number and pod weight. As expected, pod number and weight were highly correlated with one another; a result that has recently been supported in different peanut cultivars (Haro et al., 2007). This relationship between pod number and weight indicates there is no real trade-off between pod number and pod size likely due to the indeterminate nature of peanut. Aside from the relationships between pod number and weight, the strongest predictors of reproductive output were late-season traits including leaf weight and LAI. However, the correlation pattern was different for these two canopy traits; there was a negative relationship between LAI and pod weight, but for leaf weight, there was a positive relationship with pod number. This disparity may reflect an allocation trade-off where the more energy put into an overall larger canopy (LAI) had no real pay-off in production, while increased leaf production alone does have benefits because assimilation potential would be expected to increase. Mid-season traits that were important predictors of pod production were peg and pod numbers. These relationships really illustrate that the reproductive potential of the crop is determined at that mid-season point. Therefore, although the overall growth habit of peanut is indeterminate, any energy allocated to reproductive structures by the crop later in the season goes to waste because the plant cannot adequately mature these fruits.
This study documented no large differences among cultivars in nutrient accumulation or allocation of a magnitude that would have biological relevance. This result has importance to the application of the nutrient predictor model that uses the nutrient levels of leaf tissue in the late-season to predict optimum peanut harvest maturity (Rowland et al., 2008). Because genotype appears to have little effect on nutrient levels in leaf tissue across the season, application of this maturity model across different peanut genotypes is likely to give reasonable predictions of peanut maturity. The overall parabolic pattern of tissue nutrient levels confirms what Rowland et al. (2008) found for the cultivar Georgia Green. A decline in vegetative nitrogen late in the season found in these eight peanut cultivars shows a possible remobilization of nitrogen from vegetative parts to developing fruits which consequently drops nutrient levels in the late season as the crop matures (Bell et al., 1994).
While yield is the basis on which genotypes are compared and evaluated in peanut breeding trials, much less information is known about differences in growth and developmental traits because many of these characteristics are largely ignored (Banterng et al., 2003). By selecting for yield alone and not examining other traits that are causally related to yield may be a less successful strategy in breeding programs because these traits can be important predictors of yield (Wallace et al., 1993). Information on the physiological basis to yield variation among cultivars can be used to explain the mechanisms behind such variation and even determine alternate management strategies in the field that can optimize a particular genotype's performance (Baterng et al., 2003). Therefore, knowing the causes behind yield variability can ultimately aid in maximizing it.
Acknowledgements
We thank Kathy Gray for her supervision and implementation of plant sampling and analysis; without her attention to data quality, this work would not have been possible. We thank John Paulk, Rad Yager, and Ivey Griner for plot establishment and maintenance and their general support. We thank William Pearce for his assistance in field and laboratory sample processing.
Literature Cited
Bagnall D. J. and King R. W. 1991a Response of peanut (Arachis hypogaea) to temperature, photoperiod and irradiance. 1. Effect on flowering. Field Crops Res 26 : 263 – 277 .
Bagnall D. J. and King R. W. 1991b Response of peanut (Arachis hypogaea) to temperature, photoperiod and irradiance. 2. Effect on peg and pod development. Field Crops Res 26 : 279 – 293 .
Banterng , Patanothai P. A. , Pannangpetch K. , Jogloy S. , and Hoogenboom G. 2003 Seasonal variation in the dynamic growth and development traits of peanut lines. J. of Agric. Sci 141 : 51 – 62 .
Bell M. J. , Shorter R. , and Mayer R. 1991a Cultivar and environmental effects on growth and development of peanuts (Arachis hypogaea L.). I. Emergence and flowering. Field Crops Res 27 : 17 – 33 .
Bell M. J. , Shorter R. , and Mayer R. 1991b Cultivar and environmental effects on growth and development of peanuts (Arachis hypogaea L.). II. Reproductive development. Field Crops Res 27 : 35 – 49 .
Bell M. J. , Wright G. C. , and Harch G. R. 1993a Environmental and agronomic effects on the growth of four peanut cultivars in a sub-tropical environment. I. Dry matter accumulation and radiation use efficiency. Expl. Agric 29 : 473 – 490 .
Bell M. J. , Wright G. C. , and Harch G. R. 1993b Environmental and agronomic effects on the growth of four peanut cultivars in a sub-tropical environment. II. Dry matter partitioning. Expl. Agric 29 : 491 – 501 .
Bell M. J. , Wright G. C. , Suryantini , and Peoples M. B. 1994 The N2-fixing capacity of peanut cultivars with differing assimilate partitioning characteristics. Aust. J. Agric. Res 45 : 1455 – 1468 .
Bennett J. M. , Sinclair T. R. , Ma L. , and Boote K. J. 1993 Single leaf carbon exchange and canopy radiation use efficiency of four peanut cultivars. Peanut Sci 20 : 1 – 5 .
Booker F. L. , Burkey K. O. , Pursley W. A. , and Heagle A. S. 2007 Elevated carbon dioxide and ozone effects on peanut: I. Gas-exchange, biomass, and leaf chemistry. Crop Sci 47 : 1475 – 1487 .
Branch W. D. 1996 Registration of ‘Georgia Green’ peanut. Crop Sci 36 : 806 .
Branch W. D. 2002 Registration of ‘Georgia-01R’ peanut. Crop Sci 42 : 1750 – 1751 .
Branch W. D. 2003 Registration of ‘Georgia-02C’ peanut. Crop Sci 43 : 1883 – 1884 .
Branch W. D. 2004 Registration of ‘Georgia-03L’ peanut. Crop Sci 44 : 1485 – 1486 .
Burkey K. O. , Booker F. L. , Pursley W. A. , and Heagle A. S. 2007 Elevated carbon dioxide and ozone effects on peanut: II. Seed yield and quality. Crop Sci 47 : 1488 – 1497 .
Craufurd P. Q. , Vara Prasad P. V. , and Summerfield R. J. 2002 Dry matter production and rate of change of harvest index at high temperature in peanut. Crop Sci 42 : 146 – 151 .
Davidson J. I. , Griffin W. J. , Lamb M. C. , Williams R. G. , and Sullivan G. 1998 Validation of EXNUT for scheduling peanut irrigation in North Carolina. Peanut Sci 25 : 50 – 58 .
Gorbet D. W. 2006 Registration of ‘Carver’ peanut. Crop Sci 46 : 2713 .
Gorbet D. W. 2007 Registration of ‘AP-3’ peanut. J. of Plant Registrations 1 : 126 – 127 .
Gorbet D. W. and Shokes R. M. 2002 Registration of ‘C99R’ peanut. Crop Sci 42 : 2207 .
Hammer G. L. , Sinclair T. R. , Boote K. J. , Wright G. C. , Meinke H. , and Bell M. J. 1995 A peanut simulation model: I. Model development and testing. Agron. J 87 : 1085 – 1093 .
Haro R. J. , Otegui M. E. , Collino D. J. , and Dardanelli J. L. 2007 Environmental effects on seed yield determination of irrigated peanut crops: links with radiation use efficiency and crop growth rate. Field Crops Res 103 : 217 – 228 .
Holbrook C. C. and Culbreath A. K. 2007 Registration of ‘Tifrunner’ peanut. J. of Plant Registrations 1 : 124 .
Kar G. and Kumar A. 2007 Surface energy fluxes and crop water stress index in groundnut under irrigated ecosystem. Agric. Forest Met 146 : 94 – 106 .
Kiniry J. R. , Simpson C. E. , Schubert A. M. , and Reed J. D. 2005 Peanut leaf area index, light interception, radiation use efficiency, and harvest index at three sites in Texas. Field Crops Res 91 : 297 – 306 .
Kottapalli K. R. , Burow M. D. , Burow G. , Burke J. , and Puppala N. 2007 Molecular characterization of the U.S. peanut mini core collection using microsatellite markers. Crop Sci 47 : 1718 – 1727 .
Krishnamurthy L. , Johansen C. , and Sethi S. C. 1999 Investigation of factors determining genotypic differences in seed yield of non-irrigated and irrigated chickpeas using a physiological model of yield determination. J. Agron. and Crop Sci 183 : 9 – 17 .
Lamb M. C. , Davidson J. I. , and Butts C. L. 1993 Peanut yield decline in the southeast and economically feasible solutions. Peanut Sci 20 : 36 – 40 .
Lamb M. C. , Davidson J. I. , Childre J. W. , and Martin N. R. 1997 Comparison of peanut yield, quality, and net returns between non-irrigated and irrigated production. Peanut Sci 24 : 97 – 101 .
Lamb M. C. , Masters M. H. , Rowland D. , Sorensen R. B. , Zhu H. , Blankenship P. D. , and Butts C. L. 2004 Impact of sprinkler irrigation amount and rotation on peanut yield. Peanut Sci 31 : 108 – 113 .
Lanier J. E. , Jordan D. L. , Spears J. F. , Wells R. , Johnson P. D. , Barnes J. S. , Hurt C. A. , Brandenburg R. L. , and Bailey J. E. 2004 Peanut response to planting pattern, row spacing, and irrigation. Agron. J 96 : 1066 – 1072 .
Monje O. A. and Bugbee B. 1992 Inherent limitations of nondestructive chlorophyll meters: a comparison of two types of meters. Hortsci 27 : 69 – 71 .
Nageswara Rao R. C. , Talwar H. S. , and Wright G. C. 2001 Rapid assessment of specific leaf area and leaf nitrogen in peanut (Arachis hypgaea L.) using a chlorophyll meter. J. Agronomy and Crop Science 186 : 175 – 182 .
Nageswara Rao R. C. , Udaykumar M. , Farquhar G. D. , Talwar H. S. , and Prasad T. G. 1995 Variation in carbon isotope discrimination and its relationship to specific leaf area and ribulose-1,5-bisphosphate carboxylase content in groundnut genotypes. Australian Journal of Plant Physiology 22 : 545 – 551 .
Nageswara Rao R. C. and Wright G. C. 1994 Stability of the relationship between specific leaf area and carbon isotope discrimination across environments in peanut. Crop Sci 34 : 98 – 103 .
Norden A. J. and Lipscomb R. W. 1974 Influence of plant growth habit on peanut production in narrow rows. Crop Sci 14 : 454 – 457 .
Reddy T. Y. , Reddy V. R. , and Anbumozhi V. 2003 Physiological responses of groundnut (Arachis hypogea L.) to drought stress and its amelioration: a critical review. Plant Growth Reg 41 : 75 – 88 .
Rowland D. L. and Lamb M. C. 2005 The effect of irrigation and genotype on carbon and nitrogen isotope composition in peanut (Arachis hypogaea L.) leaf tissue. Peanut Sci 32 : 48 – 56 .
Rowland D. L. , Sorensen R. B. , Butts C. L. , Faircloth W. H. , and Sullivan D. G. 2008 Canopy characteristics and their ability to predict peanut maturity. Peanut Sci 35 : 43 – 54 .
Saleh G. B. and Masiron N. A. 1994 Performance and heritability studies on some peanut lines and cultivars. J. of Islamic Acad. Sci 7 : 39 – 43 .
San José J. J. , Montes R. A. , Nikonova N. , Valladares N. , Buendia C. , Malave V. , and Bracho R. 2004 Dry-matter partitioning and radiation-use efficiency in cowpea cultivars (Vigna unguiculata (L.) Walp. cvs TC-9-6 and M-28-6-6) during consecutive seasonal courses in the Orinoco llanos. J. of Agric. Sci 142 : 163 – 175 .
SAS 1997 JMP Statistical Discovery Software Cary, NC, USA SAS Institute Inc .
Thomas D. L. , Harrison K. A. , and Hook J. E. 2004 Sprinkler irrigation scheduling with the UGA EASY pan: performance characteristics. Appl. Eng. Agric 20 : 439 – 445 .
Vara Prasad P. V. , Craufurd P. Q. , and Summerfield R. J. 1999 Sensitivity of peanut to timing of heat stress during reproductive development. Crop Sci 39 : 1352 – 1357 .
Vara Prasad P. V. , Craufurd P. Q. , and Summerfield R. J. 2000 Effect of high air and soil temperature on dry matter production, pod yield and yield components of groundnut. Plant and Soil 222 : 231 – 239 .
Wallace D. H. , Baudoin J. P. , Beaver J. S. , Coyne D. P. , Halseth D. E. , Masaya P. N. , Munger H. M. , Myers J. R. , Silbernagel M. , Yourstone K. S. , and Zobel R. W. 1993 Improving efficiency of breeding for higher crop yield. Theor. Appl. Genet 86 : 27 – 40 .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
Notes
- Plant Physiologist, USDA-ARS, National Peanut Research Laboratory, PO Box 509, 1011 Forrester Dr. SE, Dawson, GA 39842; current address: Texas AgriLife Research, 1619 Garner Field Rd, Uvalde, TX 78801. [^]
- Professor and Extension Peanut Agronomist, University of Georgia, Crop and Soil Sciences Dept., 104 Research Way, Tifton, GA, 31793. [^]
- Research Agronomist, USDA-ARS, National Peanut Research Laboratory, PO Box 509, 1011 Forrester Dr. SE, Dawson, GA 39842. [^] *Corresponding author (email: dlrowland@ag.tamu.edu).
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the USDA.
Author Affiliations