Introduction
Weed control in peanut (Arachis hypogaea L.) production relies heavily on herbicides and multiple applications are routine. Intensive herbicide use is due in part to the diversity of troublesome weeds in peanut producing regions of the U. S., slow early season peanut vegetative growth (Cardina and Brecke, 1991), and subterranean fruiting that increases harvest losses from weeds beyond yield reduction from weed competition (Hauser et al., 1973).
Tall dicot weeds present unique challenges to peanut production. One of the dicot weeds that is a perpetual challenge in the southeastern U. S. is Florida beggarweed [Desmodium tortuosum (Sw.) DC]. Florida beggarweed is documented to be among the most common and troublesome weeds in each of the southeastern peanut producing states (Webster, 2005). Florida beggarweed seedlings are often shaded by peanut leaflets and inconspicuous early in the growing season (Hauser et al., 1973). As the season progresses, Florida beggarweed emerges thorough the peanut canopy and some ecotypes eventually reach a height of 3.5 m (Cardina and Brecke, 1989; 1991; Webster and Cardina, 2004). Tall weeds such as Florida beggarweed, sicklepod [Senna obtusifolia (L.) H.S. Irwin & Barneby], bristly starbur (Acanthospermum hispidum DC.), and Amaranthus spp. which escape earlier control tactics intercept fungicides which can lead to foliar disease epidemics (Royal et al., 1997). The woody stems of tall weeds also make peanut digging and vine inversion difficult (Hauser et al., 1973) and are often mowed before harvest to aid in the digging operation. Considering these possible forms of loss, including losses from competition alone, Florida beggarweed at a density of 6 plants/m2 reduces peanut yield by 25% (Hauser et al., 1975).
Florida beggarweed can be managed season-long in peanut with individual herbicides and combinations that include metolachlor, paraquat, imazapic, flumioxazin, diclosulam, and chlorimuron (Grey et al., 2003). Chlorimuron is the sole option to control escaped Florida beggarweed during the mid-season, with applications from 60 DAE until 45 days prior to harvest (Anonymous, 2009a). Chlorimuron applications are restricted to this time interval since early-season applications are frequently injurious to peanut (Sims et al., 1987). During the registered mid-season application interval, less chlorimuron is absorbed and translocated by peanut, and more is metabolized to a non-phytotoxic form compared to earlier applications (Wilcut et al., 1989). Even when applied at the appropriate time and rate, chlorimuron temporarily stunts peanut, although the phytotoxic effect has repeatedly been shown to not affect peanut yield (Colvin and Brecke, 1988; Johnson et al., 1992; Prostko et al., 2008; Wehtje and Grey, 2004; Wilcut et al., 1989).
The complex subtleness of peanut response to chlorimuron is also shown by increased incidence of spotted wilt caused by the tomato spotted wilt Tospovirus (TSWV) in chlorimuron treated peanut (Prostko et al., 2008). This phenomenon is reflected in current recommendations for managing spotted wilt that consider the use of chlorimuron as a minor risk factor that can increase incidence of spotted wilt (Brown et al., 2005). Peanut response to chlorimuron is not ignored by growers, who tend to be reluctant to use the herbicide unless escaped Florida beggarweed are numerous.
Florida beggarweed must be controlled the first 4 to 6 weeks (28 to 42 DAE) of the growing season to protect the crop from any yield reduction caused by weed interference (Hauser et al., 1975). It is interesting to note that the chlorimuron application interval (Anonymous, 2009a) is later than the critical period of weed control, meaning that a non-recoverable yield reduction is expected, even if chlorimuron controls the weed. Furthermore, chlorimuron will not consistently control Florida beggarweed taller than 25 cm (Anonymous, 2009a). Cardina and Brecke (1991) monitored Florida beggarweed growth in peanut and determined that it took approximately 45 d for Florida beggarweed height to reach 25 cm. Similarly, by 60 DAE (the earliest permissible time to apply chlorimuron), Florida beggarweed height was greater than 60 cm (Cardina and Brecke, 1991), a size beyond which chlorimuron cannot provide consistent control. In practice, peanut growers frequently apply chlorimuron to Florida beggarweed that are too large for consistent control.
Based on these circumstances, it would be useful for peanut growers to have the option of applying chlorimuron earlier than 60 DAE and presumably to younger and smaller Florida beggarweed. Wehtje and Grey (2004) evaluated the sensitivity of several peanut cultivars to early applications of chlorimuron in weed-free trials. Across all cultivars, they reported no yield response in three of four trials from early applications of chlorimuron, with the earliest application made five weeks after planting (approximately 28 DAE). While these results show that peanut may have acceptable tolerance to chlorimuron applied earlier than what is registered, the earliest labeled application timing remains 60 DAE. Therefore, research trials were initiated to determine whether the benefits of controlling smaller weeds earlier in the season compensate for the potential risk of significant peanut injury from chlorimuron.
Materials and Methods
Irrigated field trials were conducted from 2006 to 2008 near Attapulgus, GA. Soil at the Attapulgus Research Farm was a Lucy loamy sand (loamy, kaolinitic, thermic Arenic Kandiudults); 88% sand, 8% silt, 4% clay, and 0.4% organic matter. Soils at this location are representative of the southeastern U. S. peanut production region.
The experimental design was a split-plot with four replications. Main plots were chlorimuron (9 g ai/ha) applied at 21, 35, 49, 63, 77, and 91 DAE, a treated control of flumioxazin (105 g ai/ha) applied PRE (immediately after seeding peanut), and a nontreated control. A non-ionic surfactant4 (0.25% by vol.) was included with all chlorimuron treatments. Main plots were four rows (91 cm spacing) wide by 6.1 m long. Sub-plots were weeds present and weed-free with handweeding. Herbicides were applied with a tractor-mounted CO2-pressurized plot sprayer or backpack sprayer, calibrated to deliver 234 L/ha at 207 kPa using low-drift Turbo TeeJet® spray tips. Sub-plots were two rows wide and 6.1 m long.
Experimental sites were harrowed in March to terminate small grain cover crops, moldboard plowed in mid-April, seedbeds formed and shaped in late-April, and planted to peanut in early May each year. The entire experimental area was treated with pendimethalin applied preplant plant incorporated at 1.1 kg ai/ha to control annual grasses. ‘C99R’ (Gorbet and Shokes, 2002) peanut were planted in 2006 and 2007, with ‘Georgia Green’ (Branch, 1996) planted in 2008. Compared to other cultivars, both of these are considered relatively tolerant of chlorimuron (Wehtje and Grey, 2004). Maintenance weed control common to all plots the remainder of the season included one cultivation, clethodim at 0.18 kg ai/ha, and one application of bentazon at 1.1 kg ai/ha plus 2,4-DB at 0.3 kg ai/ha. A crop oil concentrate adjuvant5 (1.0% by vol.) was included with both postemergence maintenance herbicide applications. Excluding weed control, cultural practices and pest management decisions for peanut were based on recommendations from the Georgia Cooperative Extension Service (Beasley et al., 1997).
Visual estimates of percent weed control were made one week prior to harvest using a scale of 0 to 100 where 0 = no control and 100 = complete control. Weeds present at mid-season during the study were Florida beggarweed, tropic croton (Croton glandulosus var. septentrionalis Muell.-Arg.), sicklepod, and bristly starbur originating from natural infestations. Florida beggarweed were present at densities ≤5 plants/m2, tropic croton at ≤5 plants/m2, sicklepod at ≤2 plants/m2, and bristly starbur at ≤2 plants/m2.
Immediately prior to harvest, plots were mowed to remove the upper portion of tall weeds to facilitate more efficient digging and vine inversion. Peanut were dug at optimum maturity, inverted, and later combined with commercial-scale equipment, modified for small plot use. Pod samples were cleaned to remove foreign material contaminants, including weeds. A 500g sub-sample was collected for each plot to determine the percentage of total sound mature kernels (TSMK).
Data for percent weed control and peanut yield were subjected to analysis of variance with partitioning appropriate for the split-plot experimental design. Means for significant main effects and interactions were separated using Fisher's Protected LSD at P ≤ 0.05. Arcsine transformations of weed control ratings did not change the results of the analysis of variance; therefore, non-transformed data were used for analysis and presentation.
Results and Discussion
Analysis of variance indicated nonsignificant year by treatment interaction for weed control, peanut yield, and grade. Therefore, data were combined over years for presentation. There was a significant interaction between herbicide treatments and weediness for peanut yield and grade, and these data are presented as interactive means.
Flumioxazin controlled Florida beggarweed more effectively than chlorimuron regardless of application (Table 1). Among chlorimuron treatments, herbicide applications at either 21 or 35 DAE controlled Florida beggarweed 66 to 67% and were more effective than later applications of chlorimuron (38 to 51% control). Florida beggarweed height ranged from 90 to 120 cm at the earliest labeled application timing (60 DAE), which is considerably larger than the 25 cm maximum height allowance on the chlorimuron label (Anonymous, 2009a). Florida beggarweed were greater than 2 m tall when chlorimuron was applied 91 DAE and the applicators had to hold the spray boom at full extension above their head to achieve adequate spray coverage of the weed.
Flumioxazin was the only treatment that effectively controlled tropic croton in peanut (Table 1). Overall, chlorimuron did not effectively control tropic croton in these trials. Of the application timings evaluated, chlorimuron applied at 35 DAE was the most effective in controlling tropic croton (62%), which was less than control with flumioxazin (86%). Chlorimuron is not registered for tropic croton control (Anonymous, 2009a).
None of the treatments effectively controlled sicklepod (Table 1). Of the treatments evaluated, flumioxazin and chlorimuron applied from 21 through 49 DAE were the most effective (52 to 70% control). Lack of effective sicklepod control with flumioxazin is not surprising since the herbicide is not registered for sicklepod control in peanut (Anonymous, 2009b). Chlorimuron is used for sicklepod control in soybean [Glycine max (L.) Merr.] (Anonymous, 2009a). However, applications are made early-season to seedling sicklepod and often at rates greater than those applied to peanut.
Flumioxazin controlled bristly starbur better (78%) than chlorimuron, regardless of application timing (Table 1). Bristly starbur control by chlorimuron varied from 20 to 50%, with control generally less with later chlorimuron applications. While flumioxazin is not registered for bristly starbur control, suppression is often obtained (Anonymous, 2009b) and this is generally consistent with our results. Chlorimuron is also registered to suppress bristly starbur in peanut (Anonymous, 2009a). However, in our trials bristly starbur control by chlorimuron was poor, with control ranging from 20 to 30% at the registered application timings.
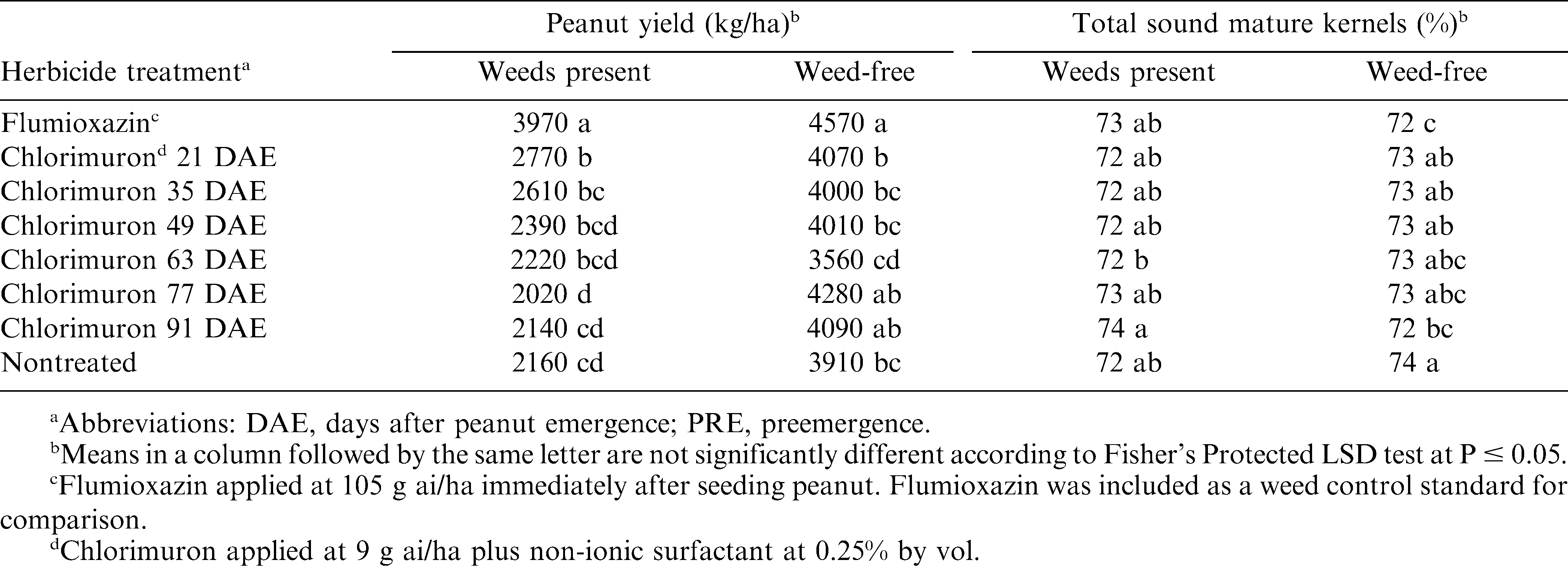
Greatest peanut yield occurred with flumioxazin in both weedy and weed-free plots (Table 2). In weedy plots, peanut treated with chlorimuron at 21 DAE yielded more than the nontreated control, while peanut treated with chlorimuron from 35 to 91 DAE yielded the same as the nontreated weedy control. Based on peanut yield response in the presence of weeds, chlorimuron applied at 21 DAE controlled dicot weeds and protected peanut yields better than later applications. However, none of the chlorimuron treatments protected peanut yield as effectively as flumioxazin.
In weed-free plots, there were no differences in peanut yield among peanut treated with chlorimuron at any application timing, excluding 63 DAE, which we consider an anomaly in the data. In absence of weeds, peanut treated with chlorimuron earlier than permitted generally yielded the same as those treated with chlorimuron at the allowed time interval. This indicates that peanut has acceptable tolerance to chlorimuron applied earlier than permissible.
Chlorimuron did not affect TSMK, regardless of the presence of weeds (Table 2). Factors that severely stunt peanut can delay maturity and reduce grade if harvested too soon. This possible effect on peanut grade was not observed and is further evidence that early applications of chlorimuron are not injurious enough to significantly affect peanut growth. Weed-free peanut treated with flumioxazin and chlorimuron at 91 DAE had lower grade than nontreated peanut. There was neither significant visual injury (data not shown) nor yield reduction from these treatments, thus no obvious explanation why grades were reduced from these treatments.
These results indicate there is little reason for peanut growers to apply chlorimuron for weed control in peanut, regardless of the time of application, but especially as the label presently permits. This is in agreement with Wehtje et al. (2000) who showed reduced net returns from peanut production when chlorimuron was applied late-season and integrated into an overall weed management system. They attributed these results to weed control too late in the season to protect yields and subtle peanut injury from which there was incomplete recovery. Chlorimuron is registered for control of one weed species in peanut, Florida beggarweed, which further limits the utility of this herbicide. Grey and Bridges (2005) applied chlorimuron 49 or 63 DAE and neither application consistently controlled Florida beggarweed or increased peanut yield over the weed-free control. In our trials, chlorimuron applied 21 DAE was more efficacious than chlorimuron applied at later applications, but Florida beggarweed control was still less than flumioxazin. Furthermore, flumioxazin controlled Florida beggarweed, tropic croton, and bristly starbur more effectively than chlorimuron at any application timing.
Our trials were structured such that there was minimal dicot weed control other than from the experimental treatments. In practice, chlorimuron is used to control Florida beggarweed that escaped control from earlier treatments. Perhaps early season herbicides would control, stunt, or weaken Florida beggarweed and give the impression of better performance of chlorimuron compared to chlorimuron used as the sole means of Florida beggarweed control. However, this scenario was not created in our trials and is purely speculative.
It is clear in our trials that the risk of early applications of chlorimuron are minor compared to the potential benefits of controlling younger and smaller Florida beggarweed. Chlorimuron applied to weed-free peanut did not injure peanut enough to reduce yield compared to the nontreated control, regardless of the time of application. However, other weed control options such as flumioxazin are more efficacious and beneficial than chlorimuron at any time of application and improve peanut yield.
Acknowledgements
We acknowledge the superior technical contributions of Robert W. Giddens and Daniel R. Evarts to these studies. Appreciation is also expressed to William R. Mills, III, Superintendent of the Attapulgus Research and Extension Center for research support.
Literature Cited
Anonymous 2009a DuPont Classic Herbicide label. http://msds.dupont.com/msds/pdfs/EN/PEN_09004a35804a989d.pdf (Accessed 2 February 2009).
Anonymous 2009b Valor SX Herbicide label. http://www.valent.com/agriculture/products/valor/label-msds.cfm (Accessed 25 February 2009).
Beasley J. , Bader M. , Baldwin J. , Harris G. , Padgett B. , Brown S. L. , and MacDonald G. 1997 Peanut Production Field Guide. Georgia Coop. Ext. Ser. Bull 1146 .
Branch W. D. 1996 Registration of ‘Georgia Green’ peanut. Crop Sci 36 : 806 .
Brown S. L. , Culbreath A. K. , Todd J. W. , Gorbet D. W. , Baldwin J. A. , and Beasley J. P. 2005 Development of a method of risk assessment to facilitate integrated management of spotted wilt of peanut. Plant Dis 89 : 348 – 356 .
Cardina J. and Brecke B. J. 1991 Florida beggarweed (Desmodium tortuosum) growth and development in peanuts (Arachis hypogaea). Weed Technol 5 : 147 – 153 .
Cardina J. and Brecke B. J. 1989 Growth and development of Florida beggarweed (Desmodium tortuosum) selections. Weed Sci 37 : 207 – 210 .
Colvin D. L. and Brecke B. J. 1988 Peanut yield and weed control as affected by timing and application rate of chlorimuron. Proc. South. Weed Sci. Soc 41 : 60 .
Gorbet D. W. and Shokes F. M. 2002 Registration of ‘C-99R’ peanut. Crop Sci 42 : 2207 .
Grey T. L. and Bridges D. C. 2005 Control method and time of emergence effects on Florida beggarweed (Desmodium tortuosum) and competition in peanut (Arachis hypogaea). Peanut Sci 32 : 73 – 80 .
Grey T. L. , Bridges D. C. , Eastin E. F. , Prostko E. P. , Vencill W. K. , Johnson W. C. , Brecke B. J. , MacDonald G. E. , Ducar J. A. , Everest J. W. , Wehtje G. R. , and Wilcut J. W. 2003 Residual weed control with imazapic, diclosulam, and flumioxazin in southeastern peanut (Arachis hypogaea). Peanut Sci 30 : 23 – 28 .
Hauser E. W. , Buchanan G. A. , and Ethredge W. J. 1975 Competition of Florida beggarweed and sicklepod with peanuts. I. Effects of periods of weed-free maintenance or weed competition. Weed Sci 23 : 368 – 372 .
Hauser E. W. , Santelmann P. W. , Buchanan G. A. , and Rud O. E. 1973 Controlling weeds in peanuts. 327 – 360 In Peanuts - Culture and Uses Amer. Peanut Res. Educ. Assoc., Inc Stillwater, OK .
Johnson W. C. , Mullinix B. G. , and Brown S. M. 1992 Phytotoxicity of chlorimuron and tank mixtures on peanut (Arachis hypogaea). Weed Technol 6 : 404 – 408 .
Prostko E. P. , Kemerait R. C. , Jost P. H. , Johnson W. C. , Brown S. N. , and Webster T. M. 2008 The influence of cultivar and chlorimuron on spotted wilt disease of peanut. Peanut Sci (in press).
Royal S. S. , Brecke B. J. , Shokes F. M. , and Colvin D. L. 1997 Influence of broadleaf weeds on chlorothalonil deposition, foliar disease incidence, and peanut (Arachis hypogaea) yield. Weed Technol 11 : 51 – 58 .
Sims G. R. , Wehtje G. , McGuire J. A. , and Hicks T. V. 1987 Weed control and response of peanut (Arachis hypogaea) to chlorimuron. Peanut Sci 14 : 42 – 45 .
Webster T. M. and Cardina J. 2004 A review of the biology and ecology of Florida beggarweed (Desmodium tortuosum). Weed Sci 52 : 185 – 200 .
Webster T. M. 2005 Weed survey: southern states - broadleaf crops subsection. Proc. So. Weed Sci. Soc 58 : 291 – 306 .
Wehtje G. and Grey T. L. 2004 Response of new cultivars to early postemergence chlorimuron applications. Peanut Sci 31 : 119 – 123 .
Wehtje G. , Brecke B. J. , and Martin N. R. 2000 Performance and economic benefit of herbicides used for broadleaf weed control in peanut. Peanut Sci 27 : 11 – 16 .
Wilcut J. W. , Wehtje G. R. , Patterson M. G. , Cole T. A. , and Hicks T. V. 1989 Absorption, translocation, and metabolism of foliar applied chlorimuron in soybeans (Glycine max), peanuts (Arachis hypogaea), and selected weeds. Weed Sci 37 : 175 – 180 .
Notes
Author Affiliations
1 Research Agronomist, USDA-ARS, Coastal Plain Experiment Station, P. O. Box 748, Tifton, GA 31793.
2 Assoc. Professor, Univ. of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793.
3 Agricultural Research Statistician, Georgia Experiment Station, Griffin, GA 30223. *Corresponding author's email: Carroll.Johnson@ars.usda.gov.
4 Chem Nut 80-20® non-ionic surfactant-adjuvant, Chem Nut Inc., P. O. Box 3706, Albany, GA 31707.
5 Chem Nut Agri-Oil® crop oil concentrate adjuvant, Chem Nut Inc., P. O. Box 3706, Albany, GA 31707.