Introduction
In response to the elimination of a quota requirement in the 2002 Farm Bill, production of peanut (Arachis hypogaea L.) in South Carolina expanded from 4,400 ha (11,000 ac) prior to 2003, to 29,000 ha (72,000 ac) in 2008. Approximately 80% of this production is in virginia market-type cultivars, with the remainder in runner types. The complex of foliar and soil-borne diseases in the South Carolina coastal plain is significantly different from that of the traditional virginia-type production area in Virginia and North Carolina. In South Carolina the primary foliar disease threat is late leaf spot caused by Cercosporidium personatum (Berk. and Curt.) Deighton. However, resistance evaluation for virginia-type cultivars has logically focused on the less aggressive early leaf spot disease caused by Cercospora arachidicola Hori because this is the predominant pathogen in the traditional virginia-type production area (Shew et al., 1995). The primary soil-borne disease for South Carolina producers is stem rot caused by Sclerotium rolfsii Sacc.; rather than those of primary concern to growers in Virginia and North Carolina, Sclerotinia blight caused by Sclerotinia minor Jagger, and Cylindrocladium black rot (CBR) caused by Cylindrocladium parasiticum Crous, Wingfield, & Alfenas. Although resistance to both late leaf spot and stem rot has been identified in the virginia-type cultivar Georgia-05E (Branch and Culbreath, 2008), all of the current commercially acceptable virginia-type cultivars are highly susceptible to both stem rot and late leaf spot. As a result, all virginia-type cultivars produced in South Carolina must be protected with an intensive preventative fungicide program of 5-7 treatments for late leaf spot from 30 to 120 days after planting (DAP); with four of these treatments (at 60, 75, 90, and 105 DAP) also including a fungicide with stem rot activity (Chapin et al., 2008). This is expensive, and under the predominantly rain-fed production conditions, stem rot suppression is often erratic due to the limited opportunity to wash fungicide into the soil at critical application times. An additional risk of continually exposing highly susceptible cultivars to intense late leaf spot selection pressure is that it may shorten the useful lifespan of fungicides upon which current production systems depend. For example, tebuconazole, which was highly effective against late leaf spot prior to 2004, no longer controls this foliar disease in South Carolina (Chapin and Thomas, 2005).
In addition to the above fungal diseases, tomato spotted wilt tospovirus (TSW), vectored by thrips [Frankliniella fusca (Hinds), Thysanoptera: Thripidae], is an economically significant disease throughout the southeastern peanut producing states, including severe economic loss in the virginia-type production area (Herbert et al. 2007). All commercially available virginia-type cultivars are susceptible to economic injury from TSW, although cultivars NC-V 11 (Wynne et al. 1991), Gregory (Isleib et al. 1999), and CHAMPS (Mozingo et al. 2006) have moderate resistance (Culbreath et al. 2000).
At the 2005 National Peanut Board Grower Summit (12 July, 2005; Portsmouth, VA) a panel of grower, industry, and research leaders came to the consensus that the most critical production research need of the U.S. peanut farmer was the development of locally adapted, reduced-cost production systems which incorporate advantages found in new cultivars. This study was conducted toward meeting that need. The objective was to field screen advanced virginia-type breeding lines for resistance to spotted wilt disease, late leaf spot, and stem rot. Doing so will enable us to select for and advance cultivars better adapted to South Carolina conditions as well as avoiding the advancement of cultivars shown to be highly disease susceptible.
Materials and Methods
Experimental Design & Crop Production
Tests were conducted over three growing seasons (2006–2008) at the Edisto Research and Education Center in Barnwell County, SC. In each year, the soil type was Dothan loamy sand (fine, loamy, siliceous, thermic, Plinthic Paleudult), the experimental design was a randomized complete block with four replicates of each cultivar or breeding line, and the experimental unit was a two-row plot (0.96-m row spacing) by 9.1m length. Four experimental rows (two cultivars or breeding lines) were alternated with four traffic rows of a standard variety NC-V 11 such that experimental rows were not subjected to any traffic for plot maintenance after planting, and each experimental unit was bordered on one side by the NC-V 11 standard.
In 2006, 22 experimental virginia-type lines, plus virginia-type cultivars NC-V 11, VA 98R (Mozingo et al. 2000), and Wilson (Mozingo et al. 2004) and runner-type cultivar Georgia-03L (Branch 2004) were evaluated. In 2007, 30 experimental lines, plus virginia-type cultivars NC-V 11, Gregory, and CHAMPS, and runner-type cultivar Georgia-03L were evaluated. In 2008, 28 experimental lines; virginia-type cultivars NC-V 11, Gregory, CHAMPS, Phillips (Isleib et al. 2006), and Univ. of Florida release Florida Fancy (B.L. Tillman, pers. commun.); and runner-type cultivar Georgia-03L were evaluated. Two of the tested experimental lines have recently been released [cultivar Bailey (T.G. Isleib, pers. commun), tested as N03081T; and cultivar Georgia-08V (Branch 2009), tested as GA 012535]. All lines beginning with “N” are from the N.C. State Univ. breeding program provided by T.G. Isleib, those beginning with “VT” are from Virginia Polytechnic Inst. and State Univ. provided by F.M. Shokes and D.L. Coker, and those beginning with “FL” are from the Univ. of Florida provided by B.L. Tillman (Figs. 1– 4). Experimental lines that showed promising levels of resistance to multiple diseases were retained in the test, while lines which were highly susceptible were usually dropped the following year in favor of untested alternatives. The Georgia-03L runner-type was included as a standard in all tests because it is considered to have a significant level of resistance to spotted wilt (Branch, 2005), as well as late leaf spot and stem rot (Branch and Culbreath, 2008). NC-V 11 was included in all tests because it is the current virginia-type standard for South Carolina conditions. Like all commercially viable virginia-type alternatives, NC-V 11 is highly susceptible to stem rot, but has reduced susceptibility to the combination of late leaf spot (Chapin et al., 2008) and tomato spotted wilt (Mozingo et al., 2006).
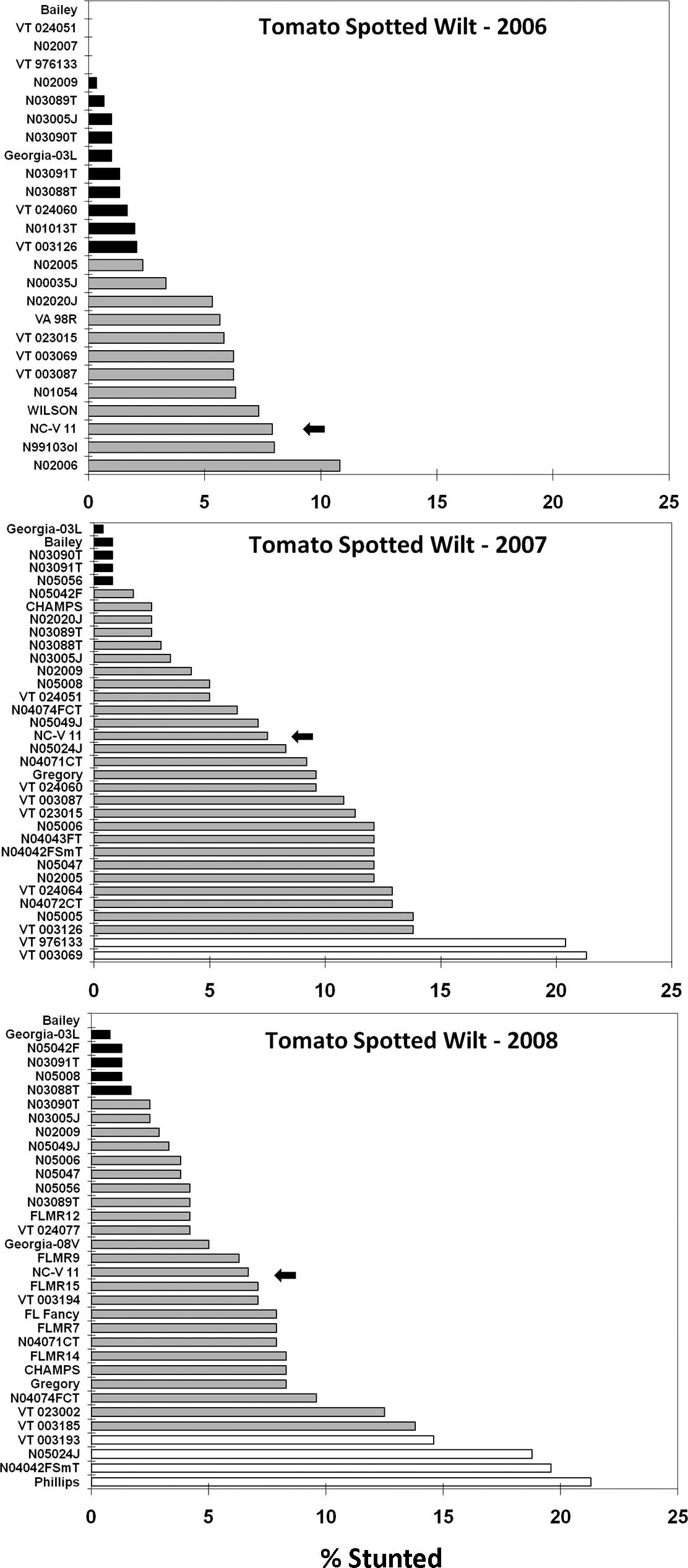
Peanut cultivar effect on tomato spotted wilt disease at Blackville, SC as measured by percent stunted row length within 20 d prior to digging. A standard virginia-type cultivar (NC-V 11) is marked with the arrow. Cultivars with shading different from the standard are significantly different (protected LSD test, P< 0.1).
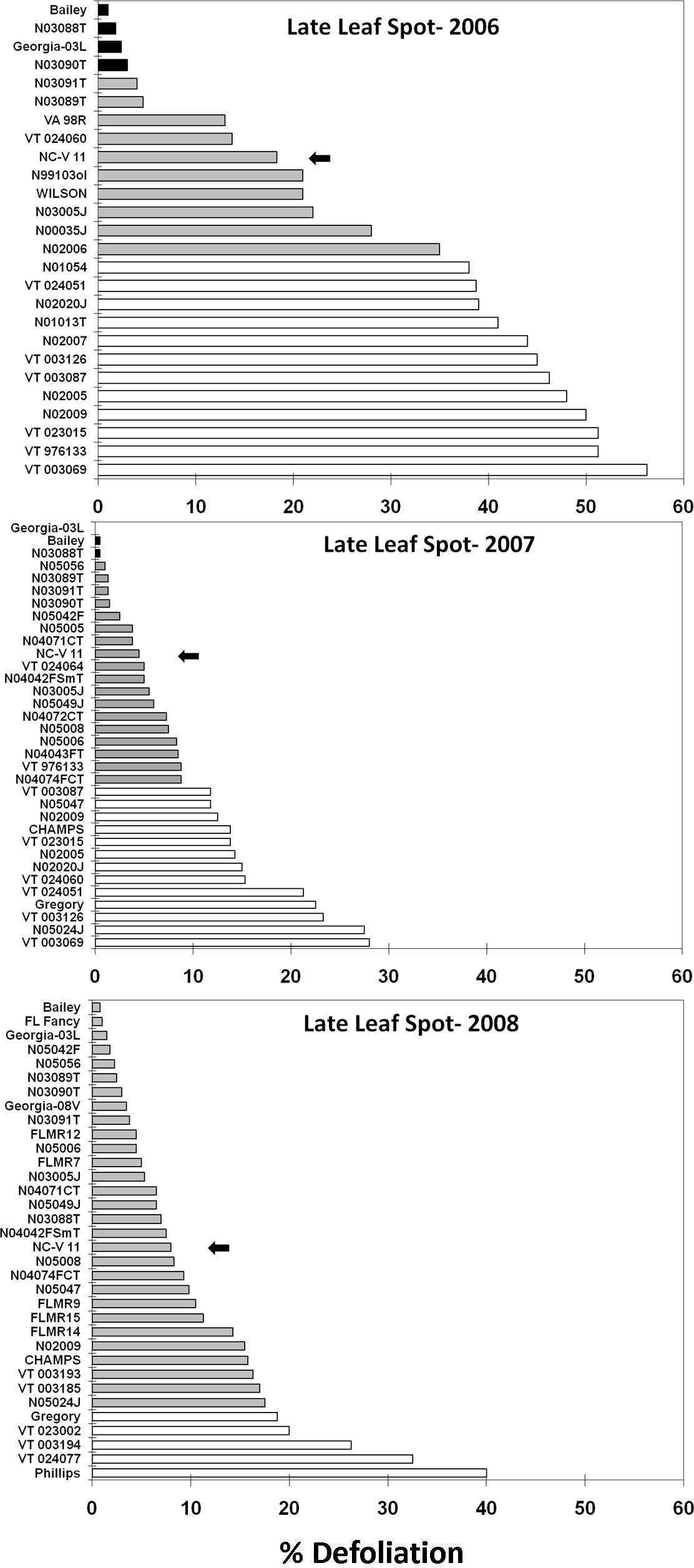
Peanut cultivar effect on late leaf spot, Cercosporidium personatum (Berk. and Curt.) Deighton, defoliation at Blackville, SC measured within 7 d prior to digging. A standard virginia-type cultivar (NC-V 11) is marked with the arrow. Cultivars with shading different from the standard are significantly different (protected LSD test, P< 0.1).
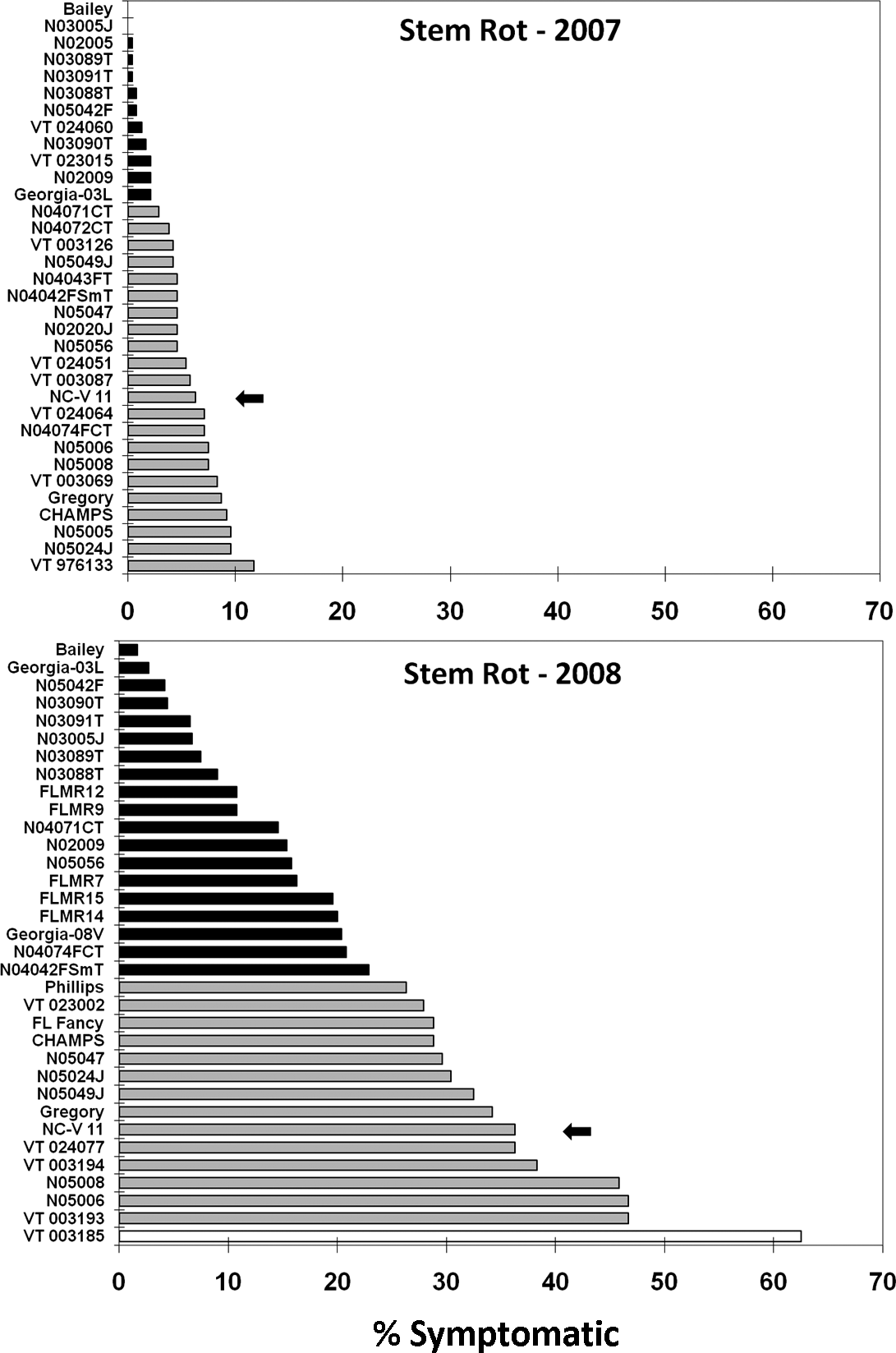
Peanut cultivar effect on stem rot, Sclerotium rolfsii Sacc., at Blackville, SC measured as percent of symptomatic row length within 2 h after digging. A standard virginia-type cultivar (NC-V 11) is marked with the arrow. Cultivars with shading different from the standard are significantly different (protected LSD test, P< 0.1).
Peanuts were produced using standard practices for virginia-type cultivars in conventional tillage (Chapin et al., 2008). Planting dates were 2 June 2006, 7 May 2007, and 1 May 2008. The intent was to plant in early May to maximize the incidence of spotted wilt disease (Brown et al., 2005) and stem rot (Hagen et al. 2001), however in 2006, the original planting on 6 May was destroyed by feral hogs and had to be replanted on the indicated date. The 2006 replanting and all subsequent tests were protected by an electric fence. A reduced fungicide program consisting of three chlorothalonil treatments (1.26 kg ai/ha) was applied at 47, 80, and 105 DAP in 2006; 43, 57, and 92 DAP in 2007; and 48, 71, and 95 DAP in 2008. The objective was to prevent complete defoliation from late leaf spot prior to harvest maturity, yet have sufficient leaf spot infection to distinguish cultivar susceptibility differences. Chlorothalonil has no activity against stem rot or spotted wilt disease. In-furrow insecticides were applied for thrips suppression (2006: acephate 1.1 kg/ha ai Orthene 97, Valent U.S.A. Corporation, Walnut Creek, CA); 2007 and 2008: aldicarb 5.6 kg/ha ai Temik 15G, Bayer CropScience, Research Triangle Park, NC). Acephate was used in the first year of the study in an attempt to provide some in-furrow suppression of direct thrips injury with minimal effect on TSW stunting. However, after potato leafhopper, Empoasca fabae (Harris), injury confounded yield evaluations for 2006, aldicarb was used for suppression of thrips and leafhopper injury in subsequent tests. Both acephate and aldicarb suppress spotted wilt (Herbert et al. 2007), however planting during the first week of May generally increases spotted wilt infection, thus allowing sufficient viral infection to make distinctions among cultivars and breeding lines. Pre-emergence applied herbicides were S-metolachor (1.39 kg/ha ai Dual Magnum, Syngenta Crop Protection, Greensboro, NC) and flumioxazin (0.1 kg/ha ai Valor, Valent U.S.A. Corp.). Imazapic (0.07 kg/ha ai Cadre DG, BASF Corp., Research Triangle Park, NC) and clethodim (0.21 kg/ha ai Select 2 EC, Valent U.S.A. Corp) were applied post-emergence.
Disease and Leafhopper Injury
Tomato spotted wilt incidence was evaluated within 20 d prior to digging by visually estimating the total row length of stunted plants with spotted wilt symptoms in each plot and then converting to a percentage of row length. Late leaf spot was evaluated by visually estimating the percent leaflet defoliation in each plot within 7 d prior to digging. In the first two years of the study, late leaf spot comprised more than 95% of leaf spot lesions. In 2008, early leaf spot comprised up to 25% of observed lesions, but the defoliation measured was still attributed primarily to the more aggressive late leaf spot disease. Stem rot was evaluated within 2 hr after digging by visually estimating the total row length symptomatic for stem rot based on the presence of characteristic sclerotia and mycelial growth. In 2006, leafhopper injury was evaluated prior to harvest by visually estimating the percent of leaflets with terminal chlorosis characteristic of leafhopper feeding.
Yield
Plots were inverted with a KMC peanut digger (Kelly Manufacturing Company, Tifton, GA) at 119, 135, and 139 DAP in 2006, 2007, and 2008, respectively, and subsequently harvested with a two-row Hobbs 525 combine (Hobbs Manufacturing Company, Albany, GA) modified with a bagging attachment. Harvested plots were weighed with a digital scale and weights adjusted to 10% moisture. Yield data for 2006 are not presented because the previously mentioned late replanting (plots destroyed by feral hogs) in combination with severe leaf spot defoliation necessitated an unrealistically early digging date to attempt stem rot evaluations. No grade data were taken as this was not an objective of these experiments. Grade characteristics and yield performance of the tested cultivars are adequately measured in other regional tests under standard fungicide programs which usually inhibit assessment of disease resistance.
Data Analysis
The data were subjected to analysis of variance using the general linear models procedure (PROC GLM) of SAS statistical software (SAS Inst. Inc., 1985). Where a significant treatment effect was measured by ANOVA, means were separated with a protected LSD test. The analysis of variance significance level for mean separations was set at P = 0.1 as deemed appropriate for screening purposes. Percentage data for disease incidence variables were transformed using arcsin(x) or (x+0.5)0.5 as appropriate prior to analysis. Pearson correlation coefficients (PROC CORR, SAS Inst. Inc., 1985) were calculated for yield and ranked disease level of tomato spotted wilt, leaf spot, stem rot, and a composite average rank of the three diseases.
Results and Discussion
Tomato Spotted Wilt
In 2006, 16 lines or cultivars had significantly less spotted wilt stunting than the NC-V 11 standard (Fig. 1): Bailey, VT024051, N02007, VT976133, N02009, N03089T, N03005J, N03090T, GA-03L, N03091T, N03088T, VT024060, N01013T, VT003126, N02005, and N00035J. In 2007, five lines or cultivars had significantly less spotted wilt stunting than the NC-V 11 standard: GA-03L, Bailey, N03090T, N03091T, and N05056. Two breeding lines had significantly more spotted wilt stunting than the standard: VT976133 and VT003069. In 2008, six lines or cultivars had significantly less spotted wilt stunting than the NC-V 11 standard: Bailey, GA-03L, N05042F, N03091T, N05008, and N03088T. Four lines or cultivars had significantly more spotted wilt stunting than the standard: VT003193, N05024J, N04002FSmT, and Phillips.
Although the NC-V 11 standard is considered susceptible to spotted wilt, this cultivar along with CHAMPS and Gregory have the best resistance currently available in commercially acceptable virginia-types. Our results demonstrate not only that there is a consistently higher level of resistance in some of the experimental virginia-type cultivars, but that some of these cultivars have resistance at least equivalent to that found in the resistant runner standard Georgia-03L. The consistency of resistance in Bailey is particularly interesting, and there also appears to be a consistent level of TSW resistance in N03091T and four Bailey sister lines (N03005J, N03088T, N03089T, and N03090T), even though only N03005J, Bailey, and N03091T were statistically significant in all three test years. The other line that had at least numerically lower TSW stunting relative to the NC-V 11 standard in all three test years was N02009. Trends in cultivar performance relative to the standard were generally consistent from year to year, but VT976133 was the exception, with unexplained contradictory results in 2007 versus 2006.
Late Leaf Spot
In 2006, four lines and cultivars had significantly less late leaf spot defoliation than the NC-V 11 standard (Fig. 2): Bailey, N03088T, GA-03L, and N03090T. Twelve lines had significantly more late leaf spot defoliation than the standard: N01054, VT024051, N02020J, N00013T, N02007, VT003126, VT003087, N02005, N02009, VT023015, VT976133, and VT003069. In 2007, three lines or cultivars had significantly less late leaf spot defoliation than the NC-V 11 standard (GA-03L, Bailey, and N03088T) while 13 (VT003087, N05047, N02009, Champs, VT023015, N02005, N02020J, VT024060, VT024051, Gregory, VT003126, N05024J, and VT003069) had significantly more late leaf spot defoliation than the standard. In 2008, none of the tested lines had significantly less late leaf spot defoliation than the NC-V 11 standard. However, five lines or cultivars had significantly more late leaf spot defoliation than the standard: Gregory, VT023002, VT003194, VT024077, and Phillips.
NC-V 11 has the best resistance to late leaf spot among commercially viable virginia-type cultivars, but our results show that significantly better resistance is present in Bailey. This level of resistance is comparable to that found in the resistant runner-type cultivar Georgia-03L. The Bailey sister lines (N03088T, N03089T, and N03090T) and N03091T also showed a consistent trend of less leaf spot defoliation than the NC-V 11 standard. Equally important is the documentation of numerous experimental lines with greater late leaf spot susceptibility than the current standard. Gregory and Phillips are the two most susceptible varieties to late leaf spot currently in commercial use. The experimental lines we found to have comparable leaf spot susceptibility would thus be poor candidates for release under South Carolina conditions.
Stem Rot
In the 2006 test there were no ratable levels of stem rot due to the previously mentioned necessity of late replanting. In 2007, 12 lines or cultivars (Bailey, N03005J, N02005, N03089T, N03091T, N03088T, N05042F, VT024060, N03090T, VT023015, N02009, and Georgia-03L) had significantly less stem rot than the NC-V 11 standard (Fig. 3). In 2008, 19 lines or cultivars (Bailey, Georgia-03L, N05042F, N03090T, N03091T, N03005J, N03089T, N03088T, FLMR12, FLMR9, N04071CT, N02009, N05056, FLMR7, FLMR15, FLMR14, GEORGIA-08V, N04074CT, and N04042FSmT) had significantly less stem rot than the NC-V 11 standard. One breeding line (VT003185) had significantly more stem rot than the standard.
The stem rot incidence measured in NC-V 11, Gregory, Champs, and Phillips reinforces the fact that all virginia-type cultivars currently available to producers are highly susceptible to this soil-borne disease. Our results demonstrate that some experimental lines have a significant level of stem rot resistance and that in some cases this resistance is equivalent to that of the resistant runner type cultivar Georgia-03L. Bailey demonstrated the most impressive stem rot resistance. Other lines that showed significant resistance in both stem rot tests were the Bailey sister lines (N03088T, N03089T, N03090T, and N03005J), plus, N03091T, N05042F, and N02009. N02005, VT024060, and VT023015 demonstrated stem rot resistance in the 2007 test, but were not included in the 2008 test due to poor performance against late leaf spot and /or spotted wilt disease. The Florida lines (FLMR7, FLMR9, FLMR12, FLMR14, FLMR15) which were tested only in 2008, demonstrated less stem rot susceptibility than the standard, but greater susceptibility than the Bailey sister lines. N04071CT and N05056 also had significantly reduced stem rot in 2008 and had numerically lower levels of stem rot in 2007 test when lower infection levels probably reduced the ability to detect differences.
Yield
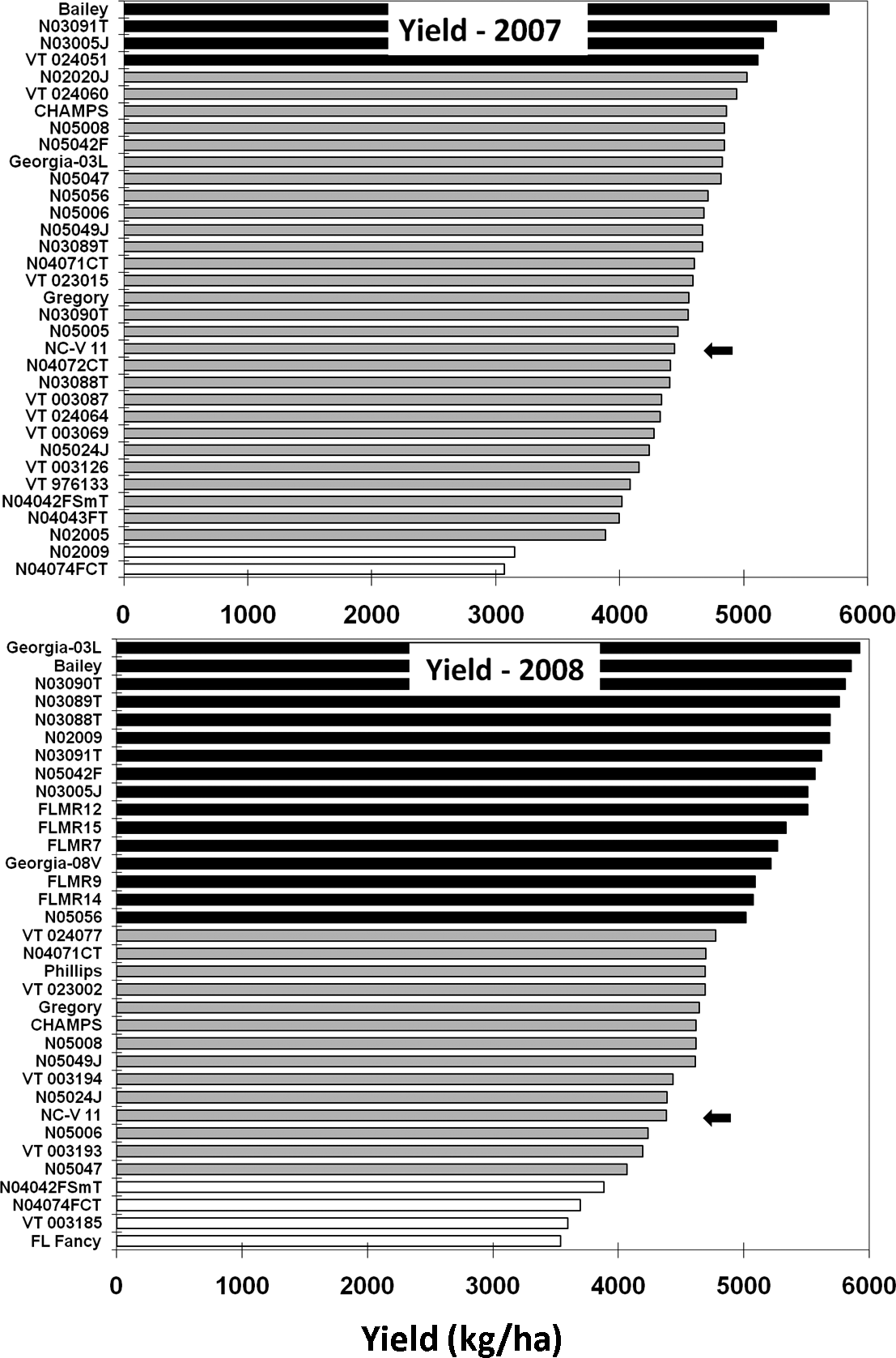
In 2007, four lines had significantly greater yield than the NC-V 11 standard (Fig. 4): Bailey, N03091T, N03005J, and VT024051. Two breeding lines had significantly lower yield than the standard: N02009 and N04074F. In 2008, 16 lines or cultivars (Georgia-03L, Bailey, N03090T, N03089T, N03088T, N02009, N03091T, N05042F, N03005J, FLMR12, FLMR15, FLMR7, GEORGIA-08V, FLMR9, FLMR14, and N05056) had significantly higher yield than the NC-V 11 standard; four (N04042FSmT, N04074CT, VT003185, and Florida Fancy) had significantly lower yield than the standard.
Yield results generally reflected which lines and cultivars exhibited superior disease resistance. There was a significant negative correlation between yield and TSW in 2007 (r = −0.42, N = 34, P = 0.01) and 2008 (r = −0.65, N = 34, P<0.01); between yield and leaf spot in 2008 (r = −0.42, N = 34, P = 0.01); between yield and stem rot in 2008 (r = −0.81, N = 34, P<0.01); and between yield and the composite disease rank in both 2007 (r = −0.41, N = 34, P = 0.02) and 2008 (r = −0.74, N = 34, P<0.01). In 2007 a negative correlation of yield with leaf spot and stem rot level also approached statistical significance (P = 0.10). The significant negative correlation of yield and composite disease score in both 2007 and 2008 suggests a cumulative effect of disease susceptibility on yield, with the best example of multiple disease resistance being the outstanding yield performance of Bailey, which exhibited the most consistent resistance to TSW, late leaf spot, and stem rot. Stem rot resistance of Bailey was particularly striking in that replicates of this cultivar had markedly brighter pods than any other cultivar under significant stem rot pressure in the absence of fungicides for control of soilborne pathogens. The low yields of N02005 and N02009 in 2007 were almost certainly due, in part, to high levels of seed-transmitted Diplodia collar rot caused by Lasiodiplodia theobromae (Pat.) Griffon & Maubl., syn. Diplodia gossypina Cooke, observed in these plots at harvest. N02005 and N02009 had 12 and 26% of row length, respectively, with necrotic stems and pycnidia characteristic of Diplodia collar rot in the 2007 test. The low yield of Florida Fancy in 2008, and probably the level of TSW observed in this variety, were caused by poor seed quality and an inadequate plant population (>1 m stand gaps at 14 DAP).
Leafhopper Injury and Canopy Size
In the 2006 test, Bailey, its three sister lines (N03088T, N03089T, and N03090T), and N03091T had significantly more hopperburn than NC-V 11 (F = 14.4; df = 1,25; P<0.001; data not shown). None of the other lines tested in 2006 were measurably different from the NC-V 11 standard with regard to hopperburn injury. The high levels of hopperburn in the 2006 test probably resulted from our use of acephate rather than aldicarb in-furrow for thrips suppression that year. Potato leafhopper injury is a less consistent economic threat than any one of the three diseases evaluated in this study, in that leafhopper injury can be readily suppressed with in-furrow insecticides used for thrips control, or by foliar insecticide if necessary.
In addition to a predisposition to leafhopper injury, Bailey, its sister lines (N03088T, N03089T, and N03090T), and N03091T produced a very large canopy with a poorly defined main stem at maturity. This characteristic would make it more difficult for growers to stay on the row during digging; however this potential problem is mitigated by increasing grower use of GPS guidance systems for planting and digging.
Summary and Conclusions
These results are encouraging in that we have identified and measured, relative to commercial standards, substantial levels of resistance in experimental virginia-type breeding lines for the three most economically important diseases under South Carolina production conditions. It is particularly encouraging that some lines have substantially reduced susceptibility to all three diseases and better yield performance than current standard cultivars under reduced fungicide programs. In separate tests, we have also confirmed the yield potential of two of the most promising lines (Bailey and N03091T) under maximum fungicide protection programs (Chapin and Thomas, unpublished data). Beyond direct yield improvement, the deployment of lines with multiple disease resistance could have three specific potential benefits: reduced fungicide inputs for both foliar and soil disease control, prolonged utility of currently available fungicides, and allowing growers to begin harvest earlier by advancing initial planting dates. At present, planting before the first week of May is constrained by the threat of increased levels of TSW. Beginning peanut harvest earlier would not only reduce the risk of weather related harvest loss to peanut, but would also advance cotton harvest, which typically is postponed until after peanut harvest by producers of both commodities. The levels of multiple disease resistance found in our tests have the potential to make a significant positive impact on virginia-type peanut production, particularly in South Carolina where the combination of TSW, and intensive late leaf spot and stem rot disease is so prevalent. From a grower perspective, it is imperative that new virginia-type cultivar releases have improved disease resistance in order to reduce both production costs and the risk of severe yield loss from disease control failures.
Acknowledgements
We appreciate the support of South Carolina peanut growers through the South Carolina Peanut Board and the National Peanut Board. Dr. Dennis L. Coker (formerly with the Tidewater REC, Virginia Polytechnic Inst. and State Univ.) supplied seed for testing in 2006. This is Technical Contribution No. 5572 of the Clemson Univ. Exp. Stn., and is based upon work supported by CSREES/USDA, under project number SC-1700293.
Literature Cited
Branch W. D. 2004 Registration of ‘Georgia-03L’ peanut. Crop Sci 44 : 1485 – 1486 .
Branch W. D. and Culbreath A. K. 2008 Disease and insect assessment of candidate cultivars for potential use in organic peanut production. Peanut Sci 35 : 61 – 66 .
Branch W. D. 2009 Registration of ‘Georgia-08V’ peanut. J. Plant. Reg 3 : 143 – 145 .
Brown S. L. , Culbreath A. K. , Todd J. W. , Gorbet D. W. , Baldwin J. A. , and Beasley J. P. 2005 Development of a method of risk assessment to facilitate integrated management of spotted wilt of peanut. Plant Dis 89 : 348 – 356 .
Chapin J. W. , Prostko E. P. , and Thomas J. S. 2008 Peanut money-maker production guide – 2008. Clemson University Extension Circular 588. 60 .
Chapin J. W. and Thomas J. S. 2005 Effect of fungicide treatments, pod maturity, and pod health on peanut peg strength. Peanut Sci 32 : 119 – 125 .
Culbreath A. K. , Todd J. W. , Gorbet D. W. , Brown S. L. , Baldwin J. , Pappu H. R. , and Shokes F. M. 2000 Reaction of peanut cultivars to spotted wilt. Peanut Sci 27 : 35 – 39 .
Hagan A. K. , Weeks J. R. , Bowen K. L. , and Wells L. 2001 Influence of production practices on peanut disease and yield. Alabama Agric. Exp. Sta. Bull. 643. 11 .
Herbert D. A. , Malone S. , Aref S. , Brandenburg R. L. , Jordan D. L. , Royals B. M. , and Johnson P. D. 2007 Role of insecticides in reducing thrips injury to plants and incidence of tomato spotted wilt virus in virginia market-type peanut. J. Econ. Entomol 100 : 1241 – 1247.
Isleib T. G. , Rice P. W. , Mozingo R. W. , Copeland S. C. , Graeber J. B. , Pattee H. E. , Sanders T. H. , Mozingo R. W. , and Coker D. L. 2006 Registration of ‘Phillips’ peanut. Crop Sci 46 : 2308 – 2309 .
Isleib T. G. , Rice P. W. , Mozingo R. W. , Mozingo R. W. , and Pattee H. E. 1999 Registration of ‘Gregory’ peanut. Crop Sci 39 : 1526 .
Mozingo R. W. , Coffelt T. A. , and Isleib T. G. 2000 Registration of ‘VA 98R’ peanut. Crop Sci 40 : 1202 – 1203 .
Mozingo R. W. , Coffelt T. A. , Phipps P. M. , and Coker D. L. 2006 Registration of ‘CHAMPS’ peanut. Crop Sci 46 : 2711 – 2712 .
Mozingo R. W. , Coffelt T. A. , Swann C. W. , and Phipps P. M. 2004 Registration of ‘Wilson’ peanut. Crop Sci 44 : 1017 – 1018 .
SAS Institute 1985 SAS/STAT guide for personal computers, version 6 edition SAS Institute Cary, NC .
Shew B. B. , Beute M. K. , and Stalker H. T. 1995 Toward sustainable peanut production: progress in breeding for resistance to foliar and soilborne pathogens of peanut. Plant Dis 79 : 1259 – 1261 .
Wynne J. C. , Coffelt T. A. , Mozingo R. W. , and Anderson W. F. 1991 Registration of ‘NC-V 11’ peanut. Crop Sci 31 : 484 – 485 .
Notes
Author Affiliations
1 Department of Entomology, Soils, and Plant Sciences, Clemson University, Edisto REC, 64 Research Road, Blackville, SC 29817 USA.
2 Crop Science Department, North Carolina State University, Box 7629, Raleigh, NC 27695.
3 Tidewater AREC (retired), Virginia Polytechnic Institute and State University, 6321 Holland Rd., Suffolk, VA 23437.
4 Department of Crop and Soil Sciences, University of Georgia, P. O. Box 748, Tifton, GA 31793.
5 Agronomy Department, University of Florida, North Florida REC, 3925 Highway 71, Marianna FL 32446.
*Corresponding author email: jchapin@clemson.edu