Introduction
Paraquat is one of the most frequently used postemergence (POST) herbicides in Southeastern peanut (Arachis hypogaea L.) production systems. However, peanut injury can occur, reducing yield and grade characteristics (Knauft et al., 1990; Wilcut and Swann, 1990). The addition of bentazon to paraquat is a common practice to reduce peanut injury, although it can be either antagonistic or synergistic in its effect on weed control depending on the weed and herbicide rate (Wehtje et al., 1992) and often does not improve peanut yield despite the reduction in crop injury (Wehtje et al., 1986, 1992; Wilcut et al., 1989). Evaluations of cultivar response to herbicide treatments containing paraquat have been studied (Knauft et al., 1990; Wehtje et al., 1991b; Wilcut and Swann, 1990). However, most of the cultivars that have been evaluated are no longer grown in the U.S. With new cultivars being released each year having varying growth habits, seed size, and resistance to common peanut diseases, such as spotted wilt of peanut, caused by tomato spotted wilt Tospovirus (TSWV), it is important to determine whether these cultivars have tolerance to POST applications of paraquat.
The influence of peanut herbicides on the incidence of TSWV has only recently been studied. Chlorimuron has been shown to slightly increase TSWV without affecting yield (Prostko et al., 2009). Other peanut herbicides, such as imazapic, 2,4-DB, and lactofen, have not had an effect on TSWV (Dotray et al., 2006; Faircloth and Prostko, 2006). However, there is little information available regarding the effects of paraquat on TSWV. This is largely due to the fact that most studies related to paraquat influence on peanut occurred prior to the mid-1990s when TSWV became a significant disease problem in peanut. One study on cv. Georgia Green did show TSWV incidence to be higher with paraquat plus bentazon plus acifluorfen compared to a non-treated control in one out of two locations, but there was no effect of paraquat plus bentazon on TSWV in either location (Shaikh et al., 2003). There were no yield differences for any treatment in that trial. Since there is a general lack of information regarding the effects of paraquat on TSWV incidence, especially on newer TSWV resistant cultivars (Culbreath et al., 2009), our objectives were to determine the influence of paraquat on yield and TSWV incidence of two currently relevant peanut cultivars, cv. Georgia-02C (Branch, 2003) and cv. Georgia-03L (Branch, 2004).
Materials and Methods
Irrigated field trials were conducted at the Ponder Research Station located near Tifton, GA from 2006 through 2008. The soil type at this location was a Tifton sand (fine-loamy, kaolinic, thermic, Plinthic Kandiudults) with 96% sand, 2% silt, 2% clay, 1.2% organic matter, and pH 6.0. Peanut seed were planted in twin rows spaced 23 cm apart (91 cm between centers of twin rows) into plots that were 1.8 m wide by 8 m long. The Georgia-02C experiment was planted 10 May 2006 and 8 May 2007, dug on 9 October 2006 and 16 October 2007, and harvested on 15 October 2006 and 29 October 2007. The Georgia-03L experiment was planted 8 May 2007 and 12 May 2008, dug on 25 September 2007 and 24 September 2008, and harvested on 8 October 2007 and 29 September 2008. The plot areas were maintained weed-free using a combination of preemergence (pendimethalin, flumioxazin, and diclosulam) and POST (imazapic, 2,4-DB) herbicides and hand-weeding. Other common production practices and Univ. of Georgia Cooperative Extension recommendations (Beasley et al., 1997) were followed.
A randomized complete block design was used with a 3 by 4 factorial arrangement consisting of three herbicide treatments [non-treated control (NTC), paraquat at 0.14 kg ai/ha, and paraquat at 0.21 kg ai/ha plus bentazon at 0.28 kg ai/ha] and four application timings [7, 14, 21, and 28 d after ground cracking (DAGC)]. Peanut growth stage (Boote, 1982) at the time of application was V4 for the 7 DAGC application; V6 for the 14 DAGC application (except for Georgia-02C in 2006–V5); V7 for the 21 DAGC applications in 2007 (both experiments) and R1 in 2006 (Georgia-02C) and 2008 (Georgia-03L); and R1 for the 28 DAGC application. A nonionic surfactant (80/20, United Agri Products, Greeley, CO or LI-700, Loveland Products Inc., Greeley, CO) was included with all paraquat treatments at 0.25% v/v. The treatments were applied with a CO2 pressurized, backpack sprayer calibrated to deliver 140 L/ha at 220 to 275 kPa (11002 DG [drift reducing] fan nozzle tips with 51 cm nozzle spacing). Treatments were replicated four times.
Incidence of spotted wilt was measured just prior to peanut digging by counting the number of disease loci per linear row in 31 cm sections and transforming the data to percentage infection based upon total row length, a method adapted from Rodriguez-Kabana et al. (1975) for assessing southern stem rot in peanut. Peanut yield data were obtained using commercial digging and harvesting equipment. Peanut yields were adjusted to 7% moisture. Pod maturity percentage was determined by randomly collecting 100 pods from each plot and subjecting the pods to the hull scrape method (Williams and Drexler, 1981). Pods that fell in the brown to black mesocarp color categories were considered mature. All data were subjected to analysis of variance and pooled where appropriate. Means were separated according to Fisher's protected least significant difference (LSD) test at P = 0.05 (Steel and Torrie, 1980).
Results and Discussion
Cultivar Georgia-02C
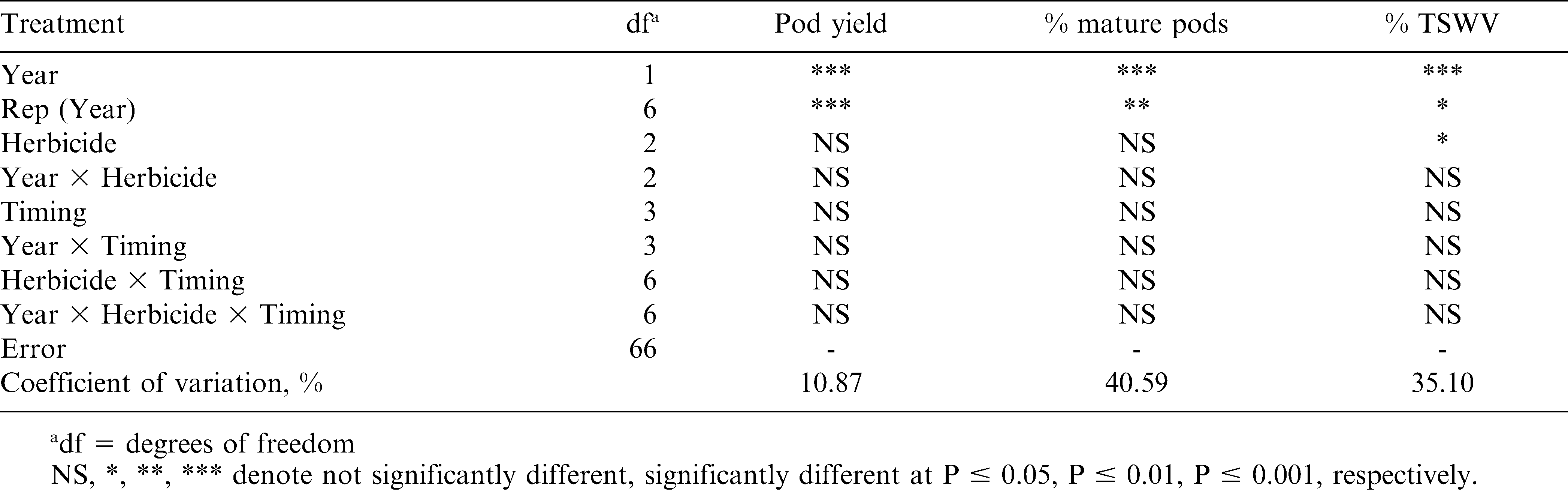
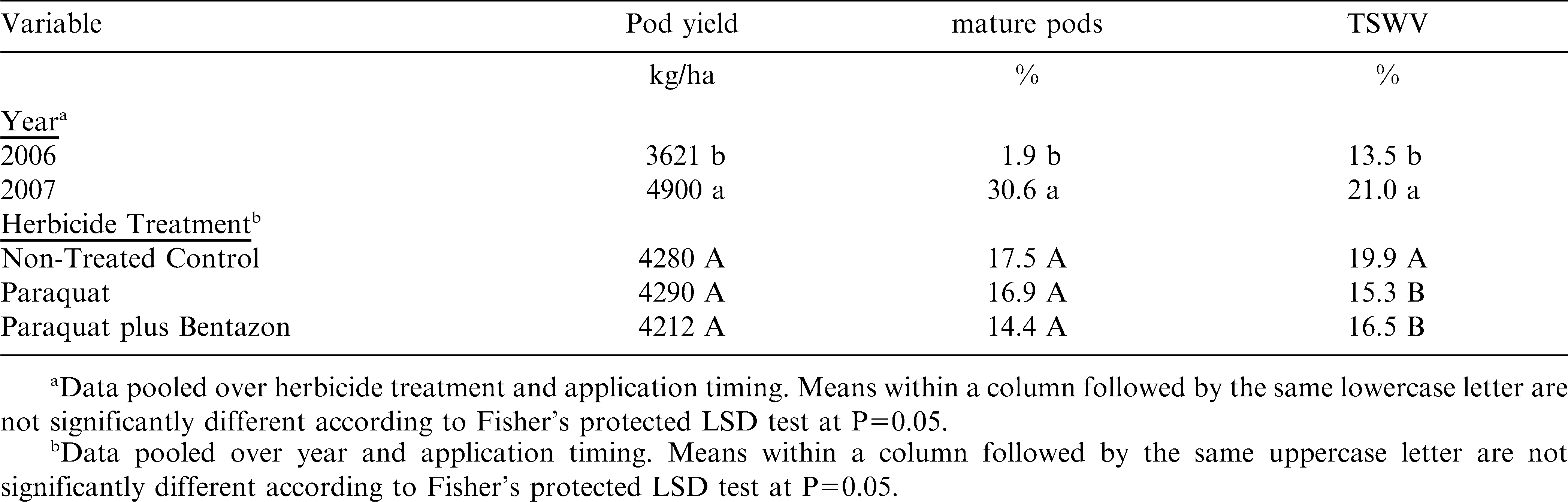
Variables analyzed include pod yield, % mature pods, and % TSWV (Table 1). There were no interactions among treatment effects, and all three variables showed differences between years. Yields were higher in 2007 than 2006, which is consistent with state averages (NASS, 2009), but is likely strongly correlated to pod maturity. Minimum temperatures dropped below 13 C for four consecutive nights (28 September to 1 October 2006), including below 9 C on 30 September, causing pod development to halt before reaching optimum digging maturity (normally 150 d after planting). Since there were no significant differences in yield among the three herbicide treatments (Table 2), this data is similar to other reports that paraquat alone at the 0.14 kg/ha rate does not reduce peanut yield compared to a NTC (Wehtje et al., 1991a, 1991b, 1994). Also, this data is similar to results in which paraquat plus bentazon yielded equally or better than a NTC (Teuton et al., 2004; Wehtje et al., 1992; Wilcut and Swann, 1990) and equal to paraquat alone (Wehtje et al., 1992). Unlike results by Knauft et al. (1990) and Wilcut and Swann (1990) in which paraquat reduced peanut yields if applied more than 7 DAGC, there was no reduction in yield of Georgia-02C regardless of application timing. More research is needed to determine if this is a result of increased tolerance or some other mechanism.
Only herbicide treatment showed significant differences on % TSWV. Incidence of TSWV was greater in the NTC than where paraquat or paraquat plus bentazon were applied (Table 2). No differences among herbicide treatments for pod maturity indicate that plant injury from paraquat was not severe enough to cause a delay in pod development.
Cultivar Georgia-03L
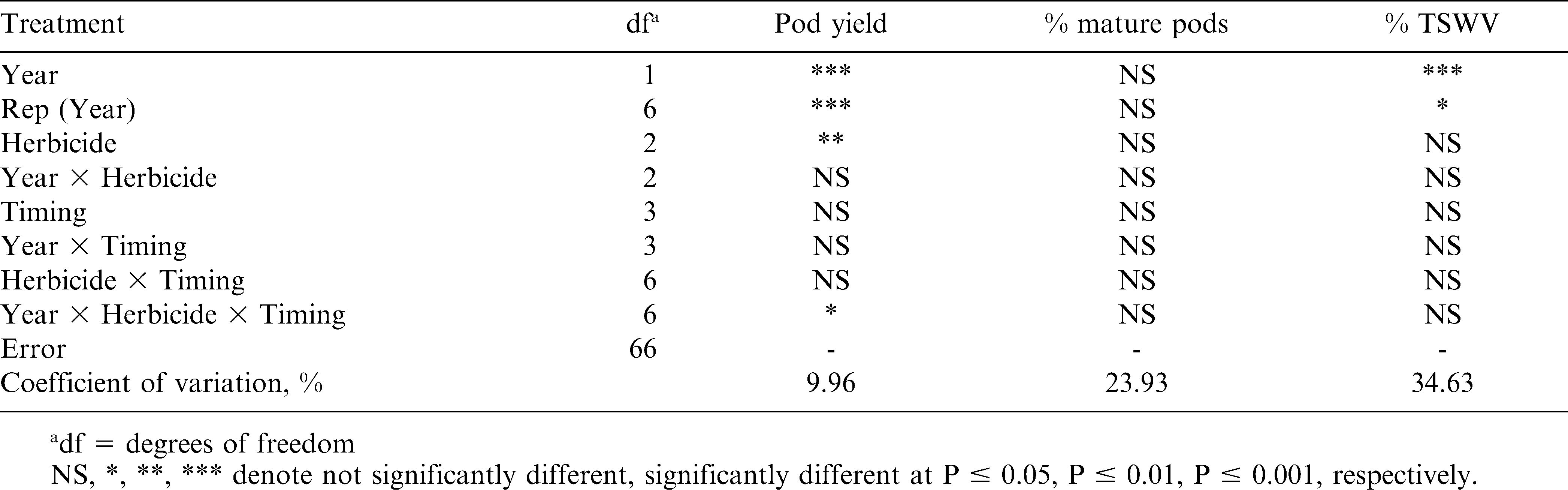
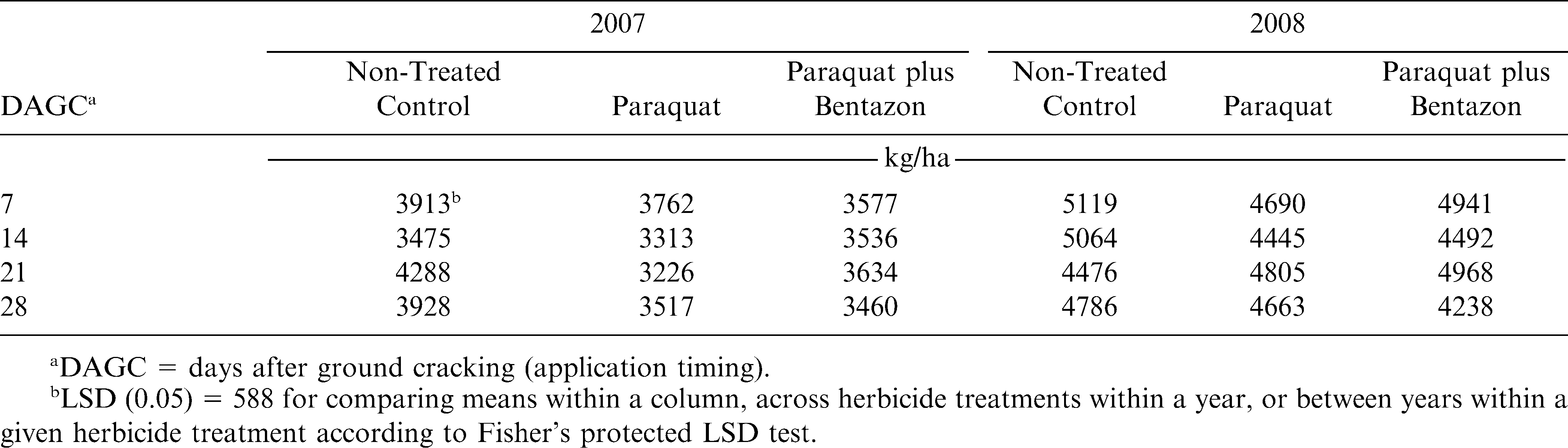
There was an interaction of year by herbicide treatment by application timing for pod yield with Georgia-03L (Table 3). In 11 of the 12 comparisons across years, peanut yields in 2008 were higher than in 2007, which also correspond with state averages between the two years (NASS, 2009). When comparing the three-way interaction by herbicide treatment, there were two instances with significant differences. In both cases (21 DAGC in 2007 and 14 DAGC in 2008), the NTC had higher yields than paraquat alone. With the 21 DAGC application in 2007, the NTC also had higher yields than paraquat plus bentazon. There were no instances where there was a yield difference between paraquat alone and paraquat plus bentazon. These two occurrences support the claims that paraquat will reduce yields if applied more than 7 DAGC (Knauft et al., 1990; Wilcut and Swann, 1990); however, there were twice as many instances in which there was no reduction in yield when herbicide treatments were applied later than 7 DAGC (Table 4). Further comparisons among application timings demonstrate no differences regardless of when paraquat alone was applied in either year while paraquat plus bentazon produced reduced yields at the 28 DAGC application compared to 7 or 21 DAGC in 2008 (Table 4).
For TSWV incidence, only a difference between years was observed at the P ≤ 0.05 level (Table 3), in which 2007 had higher disease pressure than 2008 (Table 5), which likely also contributed to higher yields in 2008. Yet, there was a year by herbicide treatment interaction at the P ≤ 0.10 level, in which the NTC (37.1%) resulted in higher TSWV incidence when compared to paraquat plus bentazon (28.4%) in 2007. This is contradictory to results by Shaikh et al. (2003) on Georgia Green (a more TSWV susceptible cultivar), where TSWV was reported to be less in NTC plots compared to herbicide treatments with paraquat and/or bentazon. However, this data is similar to what was observed on Georgia-02C peanut (Table 2). Also similar to the Georgia-02C results, there were no differences among any herbicide treatment factors for pod maturity (Tables 3 and 5), which demonstrates that plant injury from paraquat treatments did not cause a delay in pod development.
Summary and Conclusions
These results show that there is minimal evidence of damage (beyond normal) or yield reduction from paraquat on newer TSWV-resistant peanut cultivars. Crop injury data were not collected, but it was noted that injury symptoms typical of paraquat (including some stunting and leaf necrosis) were observed from all applications. The data would suggest that application of paraquat based herbicide programs at 7 DAGC might reduce the likelihood of potential yield loss compared to later applications. However, those results were not uniform and later applications up to 28 DAGC were also found to have no yield reduction, especially in Georgia-02C. Neither paraquat nor paraquat plus bentazon caused a reduction in pod maturity compared to the NTC, confirming that yield reduction by paraquat should be considered negligible in most cases, regardless of whether visible peanut injury occurs. The data also shows that paraquat based herbicide applications resulted in either equal or better control of TSWV compared to the NTC in the two cultivars evaluated. It is not understood whether this might be a result of reduced thrips feeding and subsequent infection or a synergistic effect on the genetic resistance to TSWV, or perhaps both. More research is needed to expand the knowledge of such phenomena.
Acknowledgements
Appreciation is extended to Jesse Parker, Charlie Hilton, and Kevin Baldree for their outstanding technical support.
Literature Cited
Beasley J. P. , Baldwin J. A. , Brown S. L. , Brown S. M. , Padgett B. , Bader M. J. , and Shurley D. 1997 Georgia peanut production guide Univ. of Georgia Coop. Ext. Serv. Guid Agron. 95–001 Athens, GA .
Boote K. J. 1982 Growth stages of peanut (Arachis hypogaea L.). Peanut Sci 9 : 35 – 40 .
Branch W. D. 2003 Registration of ‘Georgia-02C’ peanut. Crop Sci 43 : 1883 – 1884 .
Branch W. D. 2004 Registration of ‘Georgia-03L’ peanut. Crop Sci 44 : 1485 – 1486 .
Culbreath A. , Beasley J. , Kemerait R. , Prostko E. , Brenneman T. , Smith N. , Tubbs S. , Paz J. , Olatinwo R. , Tillman B. , Gevens A. , Weeks R. , and Hagan A. 2009 Peanut Rx: Minimizing diseases of peanut in the southeastern United States, the 2009 version of the peanut disease risk index. 41 – 56 In Prostko E. P. (ed.) 2009 Peanut Update. Spec. Pub. CSS-09-0114, Univ. of Georgia Coop. Ext, Athens, GA .
Dotray P. A. , Grichar W. J. , Baughman T. A. , Prostko E. P. , and Gilbert L. V. 2006 Peanut response and weed control with Cobra. Amer. Peanut Res. Educ. Soc. Abstr 38 : 81 .
Faircloth W. H. and Prostko E. P. 2006 Influence of herbicides on peanut yield, grade, and seed quality. Amer. Peanut Res. Educ. Soc. Abstr 38 : 78 .
Knauft D. A. , Colvin D. L. , and Gorbet D. W. 1990 Effect of paraquat on yield and market grade of peanut (Arachis hypogaea) genotypes. Weed Technol 4 : 866 – 870 .
NASS, National Agricultural Statistics Service 2009 Quick Stats: Agricultural Statistics Data Base [Online]. Available at http://www.nass.usda.gov/QuickStats/ (verified 4 May 2009).
Prostko E. P. , Kemerait R. C. , Jost P. H. , Johnson W. C. , Brown S. N. , and Webster T. W. 2009 The influence of cultivar and chlorimuron application timing on spotted wilt disease and peanut yield. Peanut Sci 36 : 85 – 91 .
Rodriguez-Kabana R. , Backman P. A. , and Williams J. C. 1975 Determination of yield losses to Sclerotium rolfsii in peanut fields. Plant Dis. Rep 59 : 855 – 858 .
Shaikh N. P. , MacDonald G. E. , and Brecke B. J. 2003 Proc. South. Weed Sci. Soc 56 : 174 (abstr.) .
Steel R. G. D. and Torrie J. H. 1980 Principles and procedures of statistics: A biometric approach. 2nd ed McGraw-Hill New York .
Teuton T. C. , Main C. L. , MacDonald G. E. , Ducar J. T. , and Brecke B. J. 2004 Green peanut tolerance to preemergence and postemergence herbicides. Weed Technol 18 : 719 – 722 .
Wehtje G. , Brecke B. , and Bostick J. P. 1994 Peanut tolerance to paraquat as influenced by seed size. Peanut Sci 21 : 12 – 16 .
Wehtje G. , McGuire J. A. , Walker R. B. , and Patterson M. G. 1986 Texas panicum (Panicum texanum) control in peanuts (Arachis hypogaea) with paraquat. Weed Sci 34 : 308 – 311 .
Wehtje G. , Wilcut J. W. , Dylewski D. P. , McGuire J. A. , and Hicks T. V. 1991a Antagonism of paraquat phytotoxicity in peanuts (Arachis hypogaea) and selected weed species by naptalam. Weed Sci 39 : 634 – 639 .
Wehtje G. , Wilcut J. W. , McGuire J. A. , and Hicks T. V. 1991b Foliar penetration and phytotoxicity of paraquat as influenced by peanut cultivar. Peanut Sci 18 : 67 – 71 .
Wehtje G. , Wilcut J. W. , and McGuire J. A. 1992 Influence of bentazon on the phytotoxicity of paraquat to peanuts (Arachis hypogaea) and associated weeds. Weed Sci 40 : 90 – 95 .
Wilcut J. W. and Swann C. W. 1990 Timing of paraquat applications for weed control in Virginia-type peanuts (Arachis hypogaea). Weed Sci 38 : 558 – 562 .
Wilcut J. W. , Wehtje G. , Hicks T. V. , and McGuire J. A. 1989 Postemergence weed control systems without dinoseb for peanuts (Arachis hypogaea). Weed Sci 37 : 385 – 391 .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
Notes
Author Affiliations
1Dept. of Crop and Soil Sciences, University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793-0748.
2 Dept. of Plant Pathology, University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793-0748.
*Corresponding author: R.S. Tubbs (Email: tubbs@uga.edu)