Introduction
High throughput processing of peanuts requires seed uniformity to ensure end-product consistency for release into the market place. Careful selection of peanut types and seed sizes are routinely used to standardize the production process. Unfortunately, these parameters are not always sufficient to ensure end-product uniformity, especially during the roasting process. Soluble sugar content, especially sucrose level, influences flavor development during the browning process (Mason et al., 1969; Reyes et al., 1982). Unfortunately, seed size does not always relate to soluble sugar concentration (Rodriguez et al., 1989). Roasting times are set to optimize the browning process of the “average” peanut; however, loads of peanuts with elevated soluble carbohydrates that may enter the processing pipeline may be over roasted and require removal from the production system. In most years, peanuts from the south-central US have excellent soluble sugar levels for the food industry; however, in some growing seasons high sugar contents are a significant problem in peanuts from the south-central US.
The reason(s) that some years and not others result in elevated soluble sugar contents within the peanuts remains to be elucidated. The work of Rodriguez et al. (1989) may provide an insight into one possible reason for elevated soluble carbohydrates. In their study, peanuts were harvested, separated into five maturity groups based on the hull-scrape method (Williams and Drexler, 1981), separated into different commercial categories based on size, and the sugar contents of the medium sized seed category was determined. Based on the hull-scrape method, peanut mesocarp colors with maturity and progresses from relatively immature stages shown by the yellow 1, yellow 2, and orange categories, to more mature stages shown by brown and black categories. In the Rodriguez et al. (1989) study, soluble carbohydrates decreased gradually in immature kernels from 9 g/100 g of defatted meal at the yellow 2 (Y2) stage to 7 g/100 g at the more mature brown stage. However, at full maturity, soluble carbohydrates increased in the black stage reaching 8.5 g/100 g of defatted meal. These results show that within a single seed size, elevated soluble carbohydrate levels change with developmental stage. Peanuts from the Y2 and BL stages had higher sugar levels than the orange B and brown stages.
Temperatures experienced throughout the growth of the peanut plant contribute greatly to the overall pod production and maturity levels (Bell et al., 1992; Golombek et al., 1995; Golombek et al., 2001; Ketring, 1984; Sanders et al., 1985; Vara Prasad et al., 1999; Vara Prasad et al., 2001). Bell et al. (1992) evaluated three peanut (Arachis hypogaea L.) cultivars under optimal field conditions in two environments in subtropical Australia contrasting primarily in night temperature. Mean minimum temperatures during the growing season were 16.7 C (Kingaroy, cool) and 20.1 C (Bundaberg, warm). Dry matter production was similar in both environments, but pod yields were slightly higher with warm nights (P < 0.05). This occurred despite large differences in rates of development between environments, with plants of all cultivars in the warm night environment maturing 23 to 24 d earlier. McMeans et al. (1990) found that a 7 C decrease in the upper layer soil temperature increased the soluble sugar concentration by 40% in Florunner (Norden et al., 1969) seeds. In another study, Golombeck et al. (1995) evaluated the influence of the soil temperature regimes 20/14 (T1), 26/20 (T2), 32/26 (T3), and 38/32 C (T4) (day/night) on seed composition of three Spanish genotypes of groundnut (Arachis hypogaea L.). The total soluble sugar concentration was higher in both extreme temperature regimes than in the intermediate treatments. Ketring (1984) also evaluated high temperature stress on vegetative and reproductive growth. His findings showed that a temperature of 35 C had an inhibitory effect on peanut development even when plants were grown under well-watered conditions.
Clearly, temperatures above and below the optimum for peanuts affect overall pod numbers and maturity. The present study investigated temperature effects directly on pod development under well-watered conditions to test the hypothesis that high sugar concentration was related to low temperature extremes. Peanuts were grown with separate pod and shoot-root zone temperatures. The impact of air versus soil temperature on carbohydrate accumulation in peanuts following low night temperatures was determined. The study investigated elevated, optimum, and sub-optimum pod temperatures on soluble carbohydrate levels and pod development. The strengths and limitations of the growth technique used in this study were identified.
Materials and Methods
Cultural Practices: Peanut (Arachis hypogaea L.) seeds of SunOleic 97R (Gorbet and Knauft, 2000) and Tamrun 96 (Smith et al., 1998) cultivars were planted into hydroponic rock wool pads (15 cm × 90 cm × 8 cm, w × l × d) [Fibrgro Horticultural Rock Wool, Fibrex Insulations Inc., Sarina, Ontario Canada] that had been saturated with Peters Professional water soluble fertilizer (0.95 g L−1 5-11-26 HYDRO-SOL [Scotts-Sierra Horticultural Products Company, Maryville, OH], supplemented with 0.475 g L−1 calcium nitrate [Ca Hydro Agri North America, Inc., Tampa, FL], and 0.238 g L−1 magnesium sulfate [Scotts-Sierra Horticultural Products Company, Maryville, OH]). The hydroponic system consisted of a series of three rock wool pads (Figure 1) with peanuts planted in the center pad (pad B). In initial studies of elevated temperature responses, plants were grown under greenhouse conditions with a 30/23 C day/night cycle. Pads used to study pod development (Figure 1, pads A and C) were placed on each side of the pad containing the plants.
Diagram and photographs of the hydroponic system consisting of a series of three rock wool pads with peanuts planted in the center pad (pad B). Pads used to study pod development (pads A and C) were placed on each side of the pad containing the plants. A heating pad (HP) was placed under pad A and pad temperature monitored continuously with a thermocouple (T) placed in the center of the growth pad.
Experiment 1. Three Tamrun 96 seeds were planted per pad and a total of three hydroponic rock wool pads (Figure 1, pad B) were placed on benches and nutrients maintained using a non-recycling hydroponic watering system. Fiber pads were flushed daily with reverse osmosis water to prevent salt accumulation in the pads. Plants were grown under greenhouse conditions with a 30/23 C day/night cycle. Heating pads (Tempco USA SHS01086, 20.5 × 91 cm) were placed beneath both of the fiber pads used for pod development (Figure 1, pad A and C), and pad temperatures were monitored using type-K thermocouples. Pad (pod-zone) temperatures were regulated using a CR23X datalogger (Campbell Sci, Logan, UT), and maintained at 25, 30 or 35 C ± 1 C. Pads containing the seeds (Figure 1, pad B) were exposed to air temperature only.
Experiment 2. Three Tamrun 96 or SunOleic 97R seeds were planted per pad (Figure 1, pad B) and a total of two hydroponic rock wool pads were placed on metal carts and nutrients maintained using a non-recycling hydroponic watering system. Plants were grown in EGC Model M-36 growth chambers with a 28/15 C day/night cycle using the same experimental setup, heating strips, thermocouples, and control system described in Experiment 1. Only one pad (Figure 1, pad A) was maintained at 28 C using heating strips, while the pad (Figure 1, pad B) which contained the seeds (and roots) and the third pad used for pod development (Figure 1, pad C) were allowed to track air temperature within the chamber. Pods were harvested 120 d after planting and peanut kernels were analyzed for sugar content.
Experiment 3. Three Tamrun 96 seeds were planted in each of two hydroponic rock wool pads using the same experimental setup, heating strips, thermocouples, and control system described in Experiment 1. Pad temperatures were regulated 24, 22, or 20 C. The fourth pad containing the seeds was allowed to track air temperature to a low of 15 C. Pods were harvested 120 and 141 d after planting and peanut kernels were analyzed for sugar content.
Soluble Sugar Analysis on HPLC: Initial studies used an HPLC method reported by Pattee et al. (2000) to determine sugar contents. In subsequent studies, sugars were determined from three sets of peanut seeds randomly sampled and ground into fine paste. Soluble sugars from 0.5 gram ground sample were extracted in 5 ml 80% ethanol at 60 C for 1 h followed by 10 min incubation at room temperature. The extract was centrifuged at 4000 rpm for 5 min and the supernatant of 40 mg equivalent was transferred into a clean Eppendorf tube. Oil was removed by partitioning the extract with ¼ volume of chloroform and centrifuged at 10000 rpm for 5 min. The aqueous phase was transferred to a new tube and dried on “Speed Vac”. Soluble sugars were re-dissolved in 0.4 ml deionized water overnight at 4 C and further diluted 10 times before analysis.
The amount of soluble sugars was analyzed using a VP Series HPLC system fitted with a SIL-10AD auto-injector and an evaporative light scattering detector-LT (Shimadzu, Columbia, MD). Samples equivalent to 50 to 200 µg were separated on a 4.6 × 250 mm YMC Polyamine II column (Waters, Milford, MA) with an mobile phase of 75% acetonitrile in water (volume:volume) at a flow rate of 1.5 ml/min. Individual sugars were identified by their retention time in comparison with the corresponding sugar standard. Quantification of individual sugars was calculated using peak area.
Results
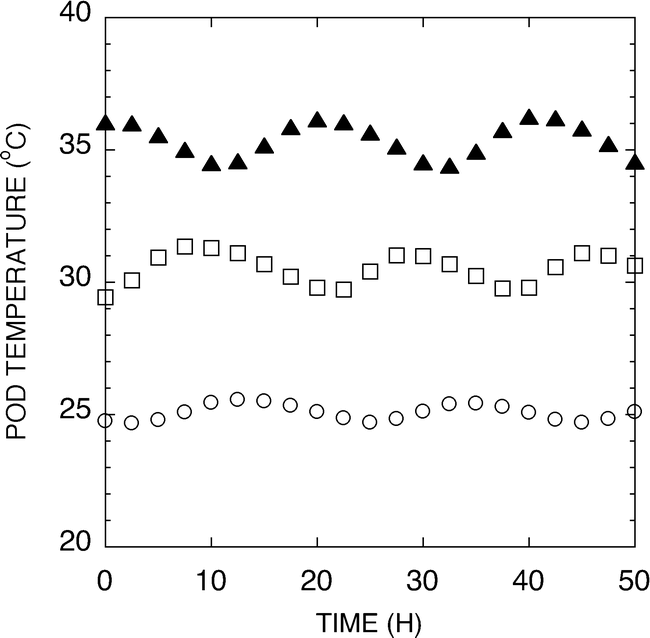
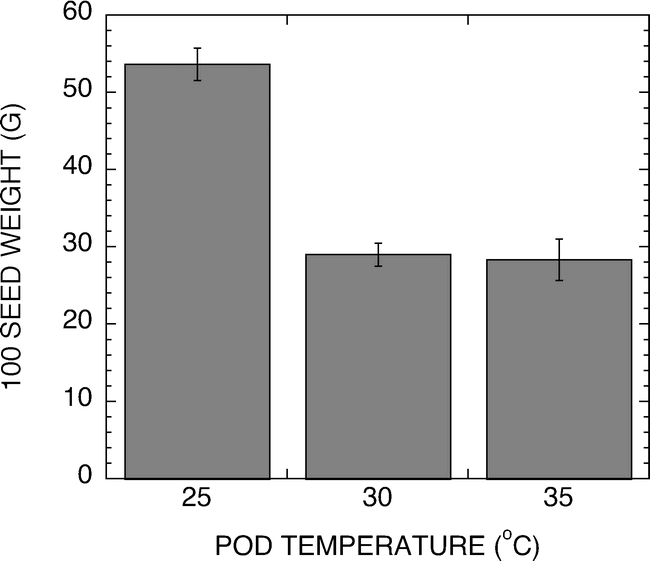
A hydroponic system was developed for use in evaluation of temperature effects on peanut pod development under greenhouse or growth chamber conditions. Figure 2 shows representative temperatures of the test pads during a 50 h period. The temperature was measured in the center of the pad and found to track the setpoint temperature ±1.5 C. Peanuts harvested from the 25, 30 or 35 C pads were analyzed for weight per 100 seed and the sensitivity of the peanut development to elevated temperatures is shown in Figure 3. The 25 C samples exhibited twice the weights per 100 seeds of either the 30 or 35 C samples. It should be noted that peanuts harvested from the 35 C pads were only found in the upper one-inch of the pad. Pegs failed to penetrate as deeply into these pads, presumably because of the severe temperature stress associated with the 35 C treatment. Because the pads were heated from the bottom, the upper most section of the pad would have the coolest temperature.
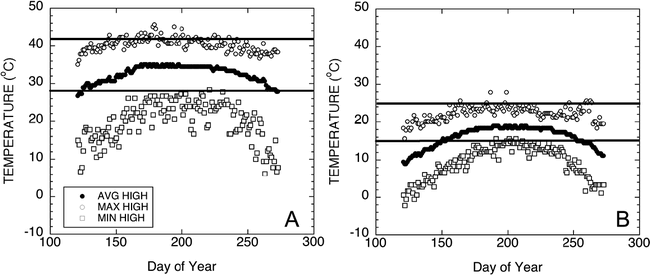
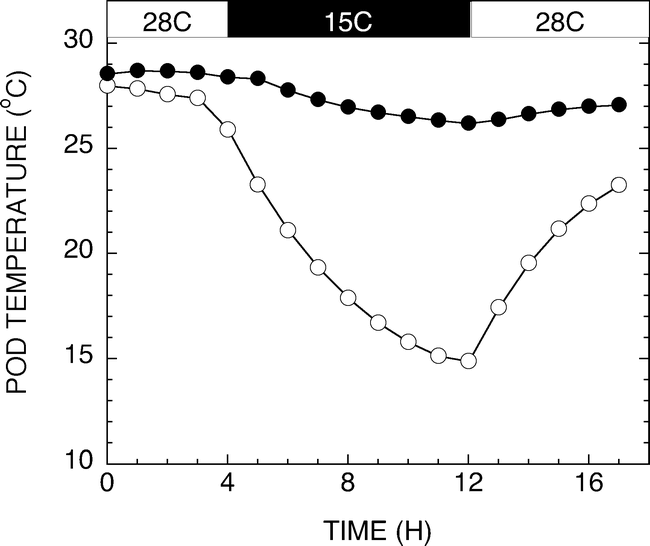
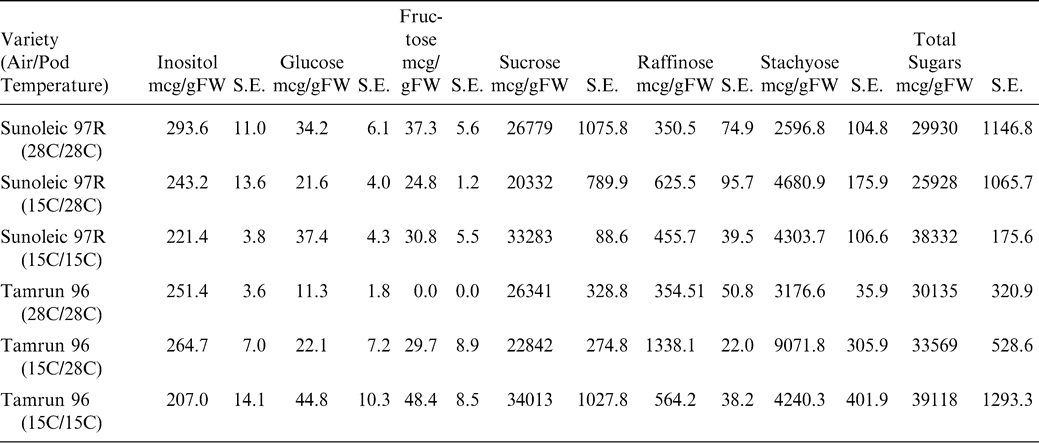
Historical high and low temperatures of the production regions of West Texas from 1922 to 2007 are shown in Figure 4. Historical temperature data for Seminole, Texas was obtained from the Western Regional Climate Center database located at http://www.wrcc.dri.edu/cgi-bin/cliRECt.pl?tx8201. Minimum high and minimum low temperatures were evaluated across the months associated with normal growing seasons. A daytime high temperature of 28 C and a nighttime low of 15 C were chosen as relevant test temperatures. Figure 5 shows a representative diurnal cycle of temperatures within the pads of growth chambers maintained at 28 C during the day and 15 C during the night. Pad temperatures begin to decrease as chamber air temperatures switched from 28 to 15 C. The pad temperatures gradually decline throughout the night reaching the target temperature of 15 C just before the switch to daytime chamber temperatures. Likewise, the daytime pad temperatures begin at 15 C and gradually increase to 28 C during the day. The fluctuations in pad temperatures are not unlike soil temperature fluctuations routinely observed in the field (Burke and Upchurch, 1995).
Graphs of the historical high and low temperatures of the production region of Seminole, Texas, taken from 85 years of weather data. Data presented in this graph were obtained from the Western Regional Climate Center database located at http://www.wrcc.dri.edu/cgi-bin/cliRECt.pl?tx8201. Maximum and minimum high (Figure 4A) and low (Figure 4B) temperatures were evaluated across the months associated with normal growing seasons. Average maximum and minimum temperatures are illustrated using the solid circle.
Graph of a representative diurnal cycle of temperatures within the pads of growth chambers maintained at 28 C during the day and 15 C during the night. Pad temperatures without external heating (open circles) begin to decrease as chamber air temperatures switch from 28 to 15 C. The pad temperatures gradually decline throughout the night reaching the target temperature of 15 C just before the switch to daytime chamber temperatures. Pads provided heating (closed circles) maintained a 28 C ± 1 C temperature.
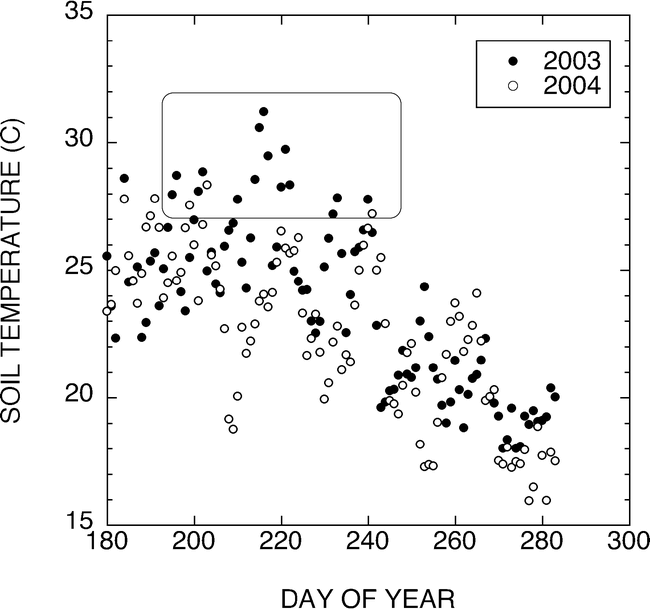
Initial studies under these temperature regimes evaluated the effect of either low (15 C) or control (28 C) air temperature and either low (15 C) or control (28 C) pad temperatures on pod development and sugar levels. Table 1 shows the sugar characteristics of Sunoleic 97R and Tamrun 96 peanuts grown under these thermal regimes. When air and pad temperatures were the same, the sucrose concentration accounted for approximately 87% of the total sugars in the peanut. When pad temperatures were warmer than the air temperature, the sucrose only accounted for 78 and 68% of the total sugar in the SunOleic 97R and Tamrun 96. Increases in raffinose and stachyose contents increased as the sucrose concentration decreased. The highest sugar contents were observed in the peanuts that developed under the 15 C/15 C air/pad temperatures. A 64 and 49% increase in sucrose concentration was observed in the SunOleic 97R and Tamrun 96, when the peanuts developed under the 15 C/15 C air/pad temperatures were compared to the peanuts from the 15 C/28 C air/pad temperatures. These results show a clear temperature response in sucrose accumulation related solely to the temperature experienced by the developing pod and independent from the air temperature experienced by the root and shoot.
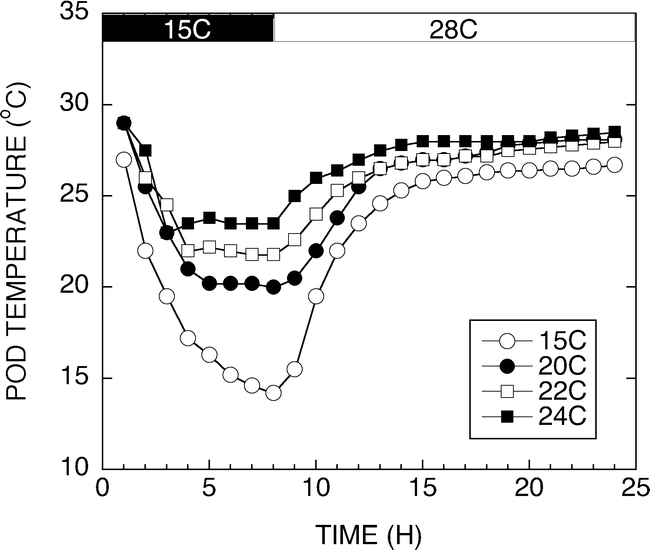
The results from this experiment raised the question of how low must the temperature around the developing pod go before the increases in sucrose concentration are observed. Figure 6 shows a typical 24 h temperature trace of pad temperatures set for 15, 20, 22, and 24 C. The peanuts were exposed to unique pad temperatures for 6 to 8 h every day throughout development. Peanuts were harvested 120 d after planting and at 141 d after planting. Sucrose accumulation was measured and the temperature effect on sucrose concentration is shown in Figure 7. Peanuts harvested at 120 DAP had significantly higher sugar contents than those harvested 141 DAP. Peanuts from plants grown for 120 d had significant differences in sucrose concentration across the four developmental temperatures. Peanuts developed under 15 or 20 C night pad temperatures exhibited increased sucrose contents compared with the 22 C and 24 C grown peanuts. The 22 C grown peanuts had lower sucrose contents than the 15 and 20 C peanuts, however, they had elevated sucrose contents compared with the 24 C grown materials. Peanuts harvested at 141 d after planting had significantly lower sucrose contents at all temperatures compared with the 120 DAP peanuts. Similar sucrose contents were observed with the 20, 22 and 24 C peanuts, while the 15 C peanuts had a slightly lower sucrose content.
Graph of sucrose concentrations from peanuts grown at varying temperatures and harvested 120 and 141 d after planting. Peanuts harvested at 120 DAP had significantly higher sugar contents than those harvested 141 DAP. Peanuts from plants grown for 120 d had significant differences in sucrose concentration across the four developmental temperatures.
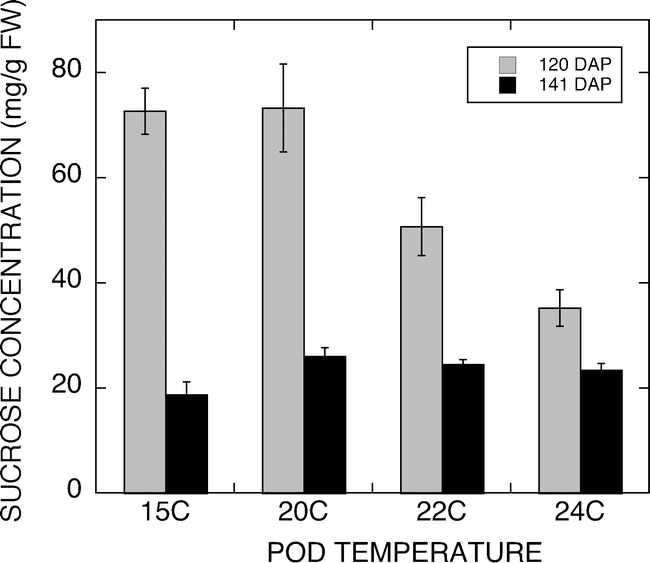
The results of the present study support earlier findings that sucrose contents in peanuts vary with peanut maturity (Rodriguez et al., 1989). The results also support earlier reports of delays in pod development associated with cooler soil temperatures (Bell et al., 1992). In an effort to relate our observations of temperature effects on sucrose concentration in peanuts with reported high sucrose concentration in the 2003 West Texas peanut crop, we compared soil temperatures at the 2-inch depth during the 2003 and 2004 growing seasons. Figure 8 shows that average daily soil temperatures were above the 27 C optimum described by Mahan et al. (2005) for 15 d during the peak pod development period in 2003 compared to 3 d during this same period during the 2004 growing season.
Graph of soil temperatures at the 2-inch depth during the 2003 and 2004 growing seasons. Average daily soil temperatures were above the 27 C optimum described by Mahan et al. (Mahan et al., 2005) for 15 d during the peak pod development period in 2003 compared to 3 d during this same period during the 2004 growing season.
Discussion
The present study investigated temperature effects directly on pod development under well-watered conditions. Peanuts were grown with separate pod-zone and shoot-root zone temperatures. The rock wool pads provided an excellent substrate for evaluating temperature effects on pod development. Pad temperatures of 30 C or below provided an even distribution of pods throughout the pads. The pads maintained at 35 C, however, showed poor pod distribution with all of the pods located on the upper surface of the pad. The heat source for the pads is below the rock wool pad and a thermal gradient radiates from the bottom to the top of the pad. Temperatures from thermocouples placed in the middle of the pad were used to regulate overall pad temperature. Clearly, a 35 C temperature was not maintained on the upper surface of the pad, thereby permitting the pods to develop at a lower temperature when they were concentrated in the upper surface of the pad.
The impact of air versus soil temperature on carbohydrate accumulation in peanuts following low night temperatures was determined using the hydroponic rock wool system. The results of this study showed that sucrose contents in both the standard (Tamrun 96) and high oleic (SunOleic 97R) peanuts were related to cool pad temperatures and not to cool air temperatures. The low temperature induced sucrose increase is consistent with the findings of McMeans et al. (1990) showing a 7 C decrease in the upper soil temperature increased the soluble sugar concentration by 40% in Florunner seeds.
The findings of the present study showed that a 2 C decline in pad temperature resulted in increased sucrose contents in peanuts harvested at 120 d after planting. An additional 2 C decline in pad temperature further increased sucrose contents to a level equivalent to the 15 C pad treatment. It is interesting to note that the peanuts allowed to develop an additional 21 d no longer showed the temperature-induced differential in sucrose content. The differences in our observations most likely relate to increased pod maturity in the 141 DAP peanuts. Although not quantified in this study, a clear difference in maturity was observed using the hull scrape method prior to peanut harvest.
Based upon previous reports of peanut temperature responses (Bell et al., 1992), the elevated soil temperatures in 2003 shown in Figure 8 would have shortened the time to maturity of existing pods. Peg development at high temperature could be reduced (Vara Prasad et al., 1999) and those pegs that did form during this 2-week period would have experienced high soil surface temperatures that would inhibit peg penetration. The consequence of the high temperature stress would be increased sucrose contents in the 2003 peanuts because of a higher proportion of mature (early season) and immature (late season) peanuts compared to the 2004-growing season that experienced more favorable temperatures for pod development.
Summary and Conclusions
In summary, this study describes a novel procedure for evaluating temperature effects on peanut pod development. With pad temperatures below 30 C, pods were evenly distributed throughout the rock wool pads, and normal pod development was observed. The findings of this study supported those of earlier studies linking low temperature induced reduction in pod development with increased sucrose concentration in the peanuts.
Acknowledgements
The authors thank Jacob Sanchez for his excellent technical assistance. Mention of a commercial or proprietary product does not constitute an endorsement by the USDA. USDA offers its programs to all eligible persons regardless of race, color, age, sex, or national origin.
Literature Cited
Bell M. J. , Wright G. C. , and Hammer G. L. 1992 Night temperature affects radiation-use efficiency in peanut. Crop Sci 32 : 1329 – 1335 .
Golombek S. D. , Sridhar R. , and Singh U. 1995 Effect of soil temperature on the seed composition of three Spanish cultivars of groundnut (Arachis hypogaea L.). J. Agr. Food Chem 43 : 2067 – 2070 .
Golombek S. D. , Sultana A. , and Johansen C. 2001 Effect of separate pod and root zone temperatures on yield and seed composition of three Spanish cultivars of groundnut (Arachis hypogaea L). J. Sci. Food Agric 81 : 1326 – 1333 .
Gorbert D. W. and Knauft D. A. 2000 Registration of SunOleic 97R′ peanut. Crop Sci 40 : 1190 – 1191 .
Ketring D. L. 1984 Temperature effects on vegetative and reproductive development of peanut. Crop Sci 24 : 877 – 882 .
Mahan J. R. , Burke J. J. , Wanjura D. F. , and Upchurch D. R. 2005 Determination of temperature and time thresholds for BIOTIC irrigation of peanut on the Southern High Plains of Texas. Irrig. Sci 23 4 : 145 – 152 .
Mason M. E. , Newell J. A. , Johnson B. R. , Koehler P. E. , and Walker G. R. 1969 Nonvolatile flavor components of peanuts. J Agr. Food Chem 17 : 728 – 732 .
Norden A. J. , Lipscomb R. W. , and Carver W. A. 1969 Registration of ‘Florunner’ peanuts. Crop Sci 9 : 850 .
Pattee H. E. , Isleib T. G. , Giesbrecht F. G. , and McFeeters R. F. 2000 Investigations into genotypic variations of peanut carbohydrates. J. Agr. Food Chem 48 : 750 – 756 .
Reyes F. G. R. , Poocharoen B. , and Wrolstad R. E. 1982 Maillard browning reaction of sugar-glycine model systems: changes in sugar concentration, color and appearance. J. Food Sci 47 : 1376 – 1377 .
Rodriguez M. M. , Basha S. M. , and Sanders T. H. 1989 Maturity and Roasting of Peanuts as Related to Precursors of Roasted Flavor. J. Agr. Food Chem 37 : 760 – 765 .
Sanders T. H. , Blankenship P. D. , Cole R. J. , and Hill R. A. 1985 Temperature relationships of peanut leaf canopy, stem, and fruit in soil of varying temperature and moisture. Peanut Sci 12 : 86 – 89 .
Smith O. D. , Simpson C. E. , Black M. C. , and Besler B. A. 1998 Registration of ‘Tamrun 96’ peanut. Crop Sci 38 : 1403 .
Vara Prasad P. V. , Craufurd P. Q. , and Summerfield R. J. 1999 Sensitivity of peanut to timing of heat stress during reproductive development. Crop Sci 39 : 1352 – 1357 .
Vara Prasad P. V. , Craufurd P. Q. , Kakani V. G. , Wheeler T. R. , and Boote K. J. 2001 Influence of high temperature during pre- and post-anthesis stages of floral development on fruit-set and pollen germination in peanut. Aust. J. of Plant Physiol 28 3 : 233 – 240 .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity. Peanut Sci 8 : 134 – 141 .
Notes
1USDA, ARS Plant Stress and Germplasm Development Unit, 3810 4th Street, Lubbock, Texas 79415.
2USDA, ARS National Peanut Research Laboratory, 1011 Forrester Dr, S.E., Dawson, GA 39842 (Current address: Texas AgriLife Research and Extension, 1619 Garner Field Road, Uvalde, TX 78801).
3USDA, ARS Market Quality & Handling Research, 236A Schaub Hall, North Carolina State University, Raleigh, NC 27695-7631.
*Corresponding author email: john.burke@ars.usda.gov