Introduction
Arachis pintoi Krapov. & W.C. Gregory is a herbaceous, perennial legume, exclusively native to Brazil. It is considered a multiple use legume, being grown for forage; ground cover in fruits orchards, forest, and low tillage systems; erosion control; and ornamental purposes. Although several cultivars have been released in different countries, little is known about the genetic diversity of the germplasm stored at genebanks.
Germplasm characterization consists of studies of eco-geographic and demographic adaptation (Martins, 1984), and according to Solbrig (1980) involves mostly the parameters of the vital cycle of the organism, genetic and physiological studies, plant pathology, and yield evaluation, among other studies. Characterization often also involves taxonomic confirmation and should produce an easy and quick way to differentiate the germplasm, using highly heritable and visible traits (Hawkes et al., 2000). Breeding programs should begin only after appropriate germplasm characterization (Cameron, 1983).
Susceptibility to root-knot nematodes, especially M. arenaria, is one of the major problems that groundnut cultivars face in the southeastern USA. A. pintoi is considered by some authors as representative of the tertiary or quaternary gene pool of A. hypogea L., and so the germplasm could be a source of resistance to M. arenaria if genetic barriers could be overcome.
The goal of this research was to evaluate the nematode resistance of several A. pintoi germplasm accessions stored at the USDA-NPGS germplasm bank.
Material and Methods
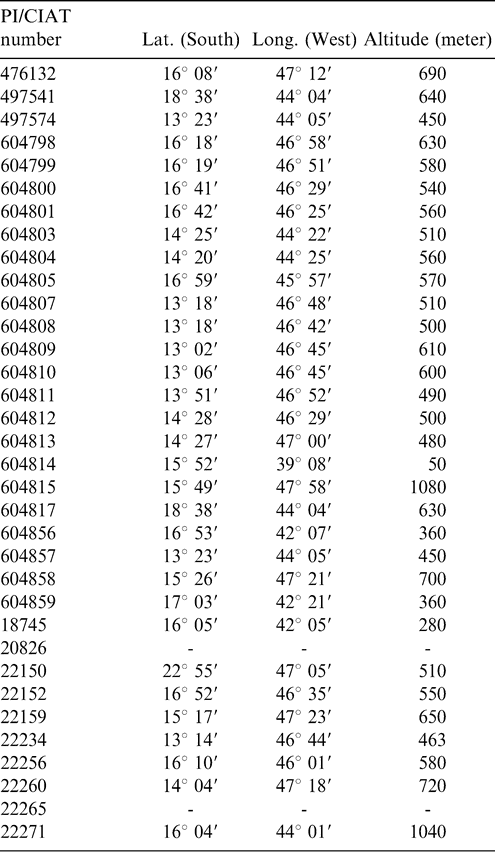
Accessions of A. pintoi stored in the Southern Regional Plant Introduction Station of the National Plant Germplasm System (NPGS) located at Griffin, Georgia were transferred to the University of Florida in 2001 and 2002. A list of these accessions with information related to the respective PI numbers and sites of collection is presented in Table 1.
Stems of the germplasm accessions were cut and placed in vermiculate trays under an automated mist system (10 sec every 30 min) for rooting. After 45 d under the mist systems, the rooted cuttings were transferred to 150 cm3 Conetainers® filled with methyl-bromide-fumigated fine sand topsoil. After transferring, plants were allowed to establish for 2 wk and then used in this experiment. On November 12th of 2003, plants were inoculated with either M. arenaria race 1, M. javanica, or M. incognita race 1. During the experimental period, plants were watered daily and fertilized with 20-20-20 fertilizer every 2 wk. The green house temperature ranged from 15 to 25°C during the 12 wk that the trial lasted.
Tomato plants were used to propagate the nematodes and then the Hussey and Barker (1973) method was used to extract eggs and juveniles. In this method, roots are cleaned, split in small pieces and washed in a 0.525% sodium hypochlorite (NaClO) solution for 2 min. The roots are then stirred strongly and passed through a 200-mesh sieve (openings 0.149–0.074 mm). The eggs and juveniles are collected on a 500-mesh (openings 0.028 mm) sieve placed under the 200-mesh one. Eggs are subsequently rinsed with H2O, pored to a beaker and water is added to bring the volume to 1000 ml. A sample is taken, placed on a slide, and the number of eggs per ml is estimated by counting under the microscope. Prior to injecting the egg suspension into the soils, the solution was diluted to 300 eggs per ml. This procedure was followed for each one of the three nematodes used in this experiment.
To each container 5 ml of egg suspension was applied, which brings the total eggs per container to 1500 or 10 eggs cm3 of soil. Application was delivered with a veterinarian surgical syringe, and during the whole process the eggs were kept in continual suspension by a magnetic stirrer.
The experimental design was a randomized complete block, with four replications for M. arenaria, and three replications for M. javanica and M. incognita. A single plant constituted each replication. Arachis hypogaea cv. ‘Florunner’ was used as a susceptible control to verify inoculum viability.
Twelve weeks after inoculation plants were removed from the containers and soil was carefully washed from the roots with tap water. Plants were then placed in a bucket with roots immersed in a 0.25% Phloxine B solution to stain the egg masses. Roots were rated for gall index (GI), gall size (GS), and percent galled area (GA) in a 1–9 scale and after that a damage index (DI) was calculated based on the same parameters (Sharma et al., 1999). DI was calculated by the following equation: DI = (GI+GS+GA)/3. GI, GS, GA and DI scales are presented in table 2.
Number of egg masses (EI) was rated with a 1–9 scale similar to gall index, where 1 represented no egg masses and 9 more than 100 egg masses. Accessions with EI = 1 were considered highly resistant to nematode reproduction and with EI = 9 were highly susceptible. Intermediate values followed the DI scale.
Results and Discussion
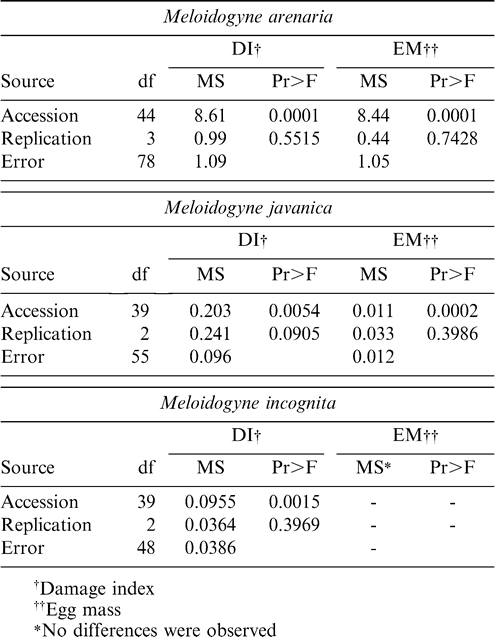
Reaction to Meloidogyne arenaria, M. javanica, and M. incognita was established in accordance with the methodology proposed by Sharma et al. (1999). The analysis of variance of A. pintoi reaction to M. Arenaria, M. javanica, and M. incognita showed significant (P < 0.01) differences among the accessions (Table 3).
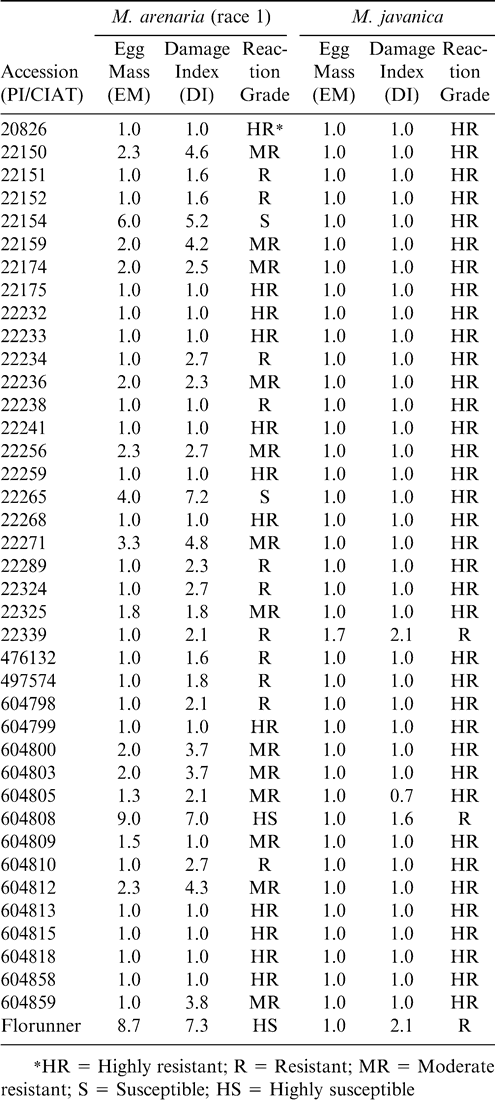
M. arenaria reaction of A. pintoi germplasm is presented in Table 4. Large genetic variability was observed among the accessions with respect to this characteristic. Among the 44 accessions evaluated, 12 were classified as highly resistant, 14 were classified as resistant, 15 were considered moderately resistant, 2 were considered susceptible, and one was considered highly susceptible. Overall 93% of the accessions presented some level of resistance and only 7% were classified as susceptible.
The A. pintoi accessions also demonstrated significant variation (P < 0.01) in response to infestation with M. javanica (Table 3). Although, significant variation was presented in M. javanica reaction, all 39 accessions evaluated were classified as highly resistant or resistant (Table 4).
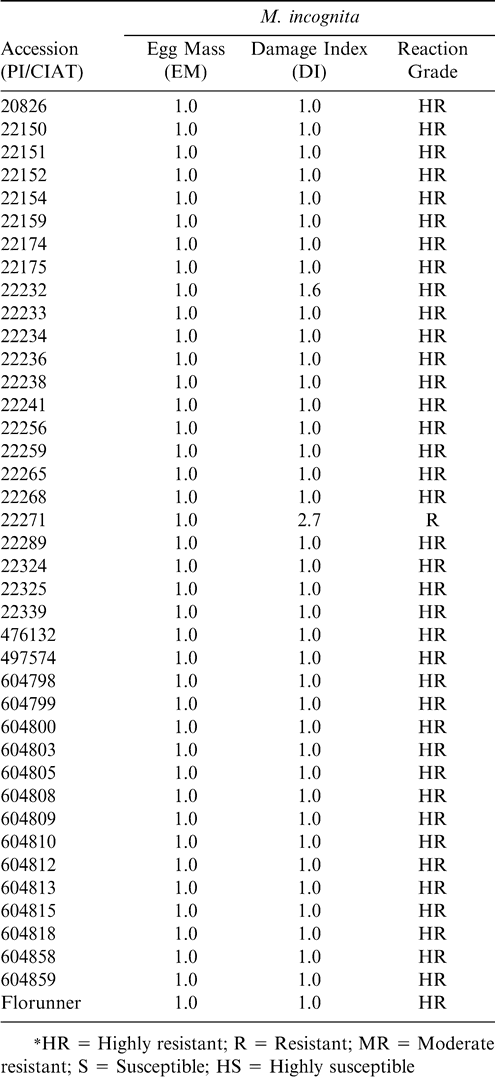
In the case of M. incognita reaction significant differences were observed among accessions only for DI (Table 3). All except two accessions showed no galling or egg mass production (Table 5). Other reports have shown that in general Arachis pintoi have near immunity to M. incognita. In fact, A. hypogaea is used as a non-host differential for M. incognita in the standard test to characterize populations of root-knot nematodes into major species and races. A. hypogaea is however generally susceptible to M. arenaria.
Nematode resistance is a valuable attribute for any species that will be incorporated into agriculture systems. It is more important with perennial plants that will have long-term exposure to soil borne problems. For a forage crop, nematode susceptibility can affect the ability to persist over a long period in the pasture. In the case of A. pintoi, which is known as multiple use legume, this characteristic could improve its utilization as ground cover and in crop rotations with cultures that are susceptible to root-knot nematodes. This is the case of the common peanut planted in the southeastern USA, which requires a crop rotation with bahiagrass (Paspalum notatum). The introduction of A. pintoi in bahiagrass pasture could improve nematode control and additionally improve the nutritive value of the pasture.
In the case of A. pintoi, nematode resistance is remarkably important to permit a wide use of the species as forage crop or even as a cover crop. Also this is important due to the fact that the species could be considered a useful source of genes for A. hypogaea, which is worldwide cultivated. Since direct crossing among the two species is not possible, some authors include A. pintoi in the tertiary gene pool of A. hypogaea. However, with the recent progress of molecular biology tools, direct transfer could be achieved even for non-related species of the genus, which makes this source of resistance potentially important.
Even though, knowledge about sources of nematode resistance is extremely important to the general use of the species and for its use in breeding programs of A. hypogaea, little was know about A. pintoi germplasm accessions response to root-knot nematodes. Information available is usually restricted to one or a few accessions. Sharma et al. (1999) studied M. javanica race 3 reaction of 161 accessions of wild Arachis species.
They reported that of the nine accessions of A. pintoi evaluated, eight were considered susceptible or highly susceptible, but a single accession was classified as moderately resistant. By contrast, all A. pintoi accessions were highly resistant to the M. javanica population used in this research (Not classified as a race).
Queneherve et al. (2002) examined A. pintoi reaction to Radopholus similis, Pratylenchus coffeae, Hoplolaimus seinhorsti, Meloidogyne incognita and M. mayaguensis. Forty-five days after inoculation R. similis, H. seinhorsti and P. coffeae multiplied in the roots. A. pintoi did not allow the multiplication of M. incognita and M. mayaguensis, indicating the inability of A. pintoi to act as a host to these two root-knot nematodes.
Santiago et al. (2002) investigated the A. pintoi reaction to M. paranaensis and M. incognita races 1, 2, 3, and 4. They reported that no root penetration by M. incognita and M. paranaensis juveniles had occurred, and hence there was no gall or egg mass formation. They concluded that in general A. pintoi accessions had an antagonistic effect on the nematodes, suggesting that they could be used as an interccrop or cover crop to reduce M. paranaensis and M. incognita populations. This research supports this conclusion and includes populations of M. arenaria since many accessions presented resistance to this species.
Nelson et al. (1989) evaluated the resistance to M. arenaria of 116 wild Arachis spp. genotypes, including a single A. pintoi accession. Resistance was identified in accessions from 11 of 15 wild species tested and in 10 of 20 accessions belonging to undescribed species. Results of field and greenhouse experiments were similar; 26 of 31 accessions common to both tests gave similar responses in both tests. Among these species, the authors identified A. batizocoi Krapov. & W.C. Gregory and A. cardenasii Krapov. & W.C. Gregory as species that are both resistant to M. arenaria and compatible with A. hypogaea. These sources of M. arenaria resistance were used to develop the germplasm line TxAG-6 (Simpson et al., 1993).
TxAG-6 is a amphiploid formed by first crossing A. cardenasii/A. diogoi Hoehne, and then crossing the 50% pollen fertile F1 hybrid with A. batizocoi. The resulting tri-species hybrid (2n = 20) was <1% pollen stained and produced no fruit. The chromosome number was doubled with colchicine to form TxAG-6 (Simpson et al., 1993).
TxAG-6 is about 89% pollen stained and is highly fertile, both selfed or when crossed with A. hypogaea. The fertile amphiploid was crossed with Florunner to incorporate M. arenaria resitance, and five back-crosses later produced the designated breeding line, TP262-3-5, which was later denominated cultivar ‘Coan’ (Simpson and Starr, 2001). In each backcross cycle, selection was made for agronomic characters similar to Florunner and resistance to root-knot nematodes (Nelson et al., 1990; Starr et al., 1990).
TxAG-6 was also used to produce the breeding line TP301-1-8, which resulted from seven back-crosses with Florunner. In each generation, selection was made for agronomic characters matching those of Florunner, the recurrent parent, and for resistance to root-knot nematodes. TP301-1-8 was named and register as cultivar ‘NemaTAM’ (Simpson et al., 2003).
Information published and available seems to support the results obtained in this research with respect to nematode reaction of A. pintoi. The source of resistance of these accessions could be used in breeding programs of A. hypogaea and more important qualify A. pintoi as potential forage, at least by this criteria, in environments where nematode infestation is a factor. Another positive outcome of this result is the ability of the species to suppress the multiplication of nematodes, and then be an important cover crop to species with nematode susceptibility problems.
Acknowledgements
This work was funded partially by the National Council for Scientific and Technological Development (CNPq), a foundation linked to the Ministry of Science and Technology (MCT), to support Brazilian research, and the Brazilian Agriculture Research Corporation (Embrapa).
Literature Cited
Cameron D. G. 1983 To breed or not to breed. 237 – 250 In McIvor J. G. and Bray R. A. Genetic resources of forage plants CSIRO Melbourne .
Hawkes J. G. , Maxted N. , and Ford-Lloyd B. V. 2000 The Ex Situ Conservation of Plant Genetic Resources Kluwer Academic Publishers Dordrecht, The Netherlands .
Hussey R. S. and Baker K. R. 1973 A comparison of methods of collecting inocula for Meloidogyne spp. including a new technique. Plant Dis. Rep 57 : 1025 – 1028 .
Martins P. S.
1984
Biological aspects of Brazilian herbaceous legumes.
173
–
184
In
Aguiar-Perecin M. L. R.
,
Martins P. S.
, and
Bandel G.
Topicos de Citogenetica e evolução de plantas
FEALQ
São Paulo, Brazil
.
Nelson S. C. , Simpson C. E. , and Starr J. L. 1989 Resistance to Meloidogyne arenaria in Arachis spp. germplasm. Supplement to the Journal of Applied Nematology. (Annals of Applied Nematology) 21 : 654 – 660 .
Nelson S. C. , Starr J. L. , and Simpson C. E. 1990 Expression of Resistance to Meloidogyne arenaria in Arachis batizocoi and A. cardenasii. Journal of Nematology 22 : 423 – 425 .
Queneherve P. , Bertin Y. , and Chabrier C. 2002 Arachis pintoi: a cover crop for bananas: Advantages and disadvantages as regards nematology. Infomusa 11 : 28 – 30 .
Santiago D. C. , Homechin M. , Krzyzanowski A. A. , Carvalho S. , and Fonseca I. C. 2002 Antagonist effect of Arachis pintoi on Meloidogyne paranaensis and M. incognita. Nematologia-Mediterranea 30 : 147 – 152 .
Simpson C. E. , Starr J. L. , Nelson S. C. , Woodard K. E. , and Smith O. D. 1993 Registration of TxAG-6 and TxAG-7 peanut germplasm. Crop Science 33 : 1418 .
Simpson C. E. and Starr J. L. 2001 Registration of COAN Peanut. Crop Science 41 : 918 .
Simpson C. E. , Starr J. L. , Church G. T. , Burow M. D. , and Paterson A. H. 2003 Registration of NemaTAM Runner Peanut. Crop Science 43 : 1561 .
Sharma S. B. , Ansari M. A. , Varaprasad K. S. , Singh A. K. , and Reddy L. J. 1999 Resistance to Meloidogyne javanica in wild Arachis species. Genetic Resources and Crop evolution 46 : 557 – 568 .
Solbrig O. T. 1980 Demography and natural selection, in Demography and evolution in plant population Blackwell Oxford .
Starr J. L. , Schuster G. L. , and Simpson C. E. 1990 Characterization of the resistance to Meloidogyne arenaria in an interspecific Arachis hybrid. Peanut Science 17 : 106 – 108 .
Notes
Author Affiliations
1Brazilian Agriculture Research Corporation (Embrapa), Embrapa Cerrados, BR 020 Km 18, PO Box 08223, Planaltina, DF-Brazil 73310-970.
2University of Florida, Agronomy Department, 304 Newell Hall, PO Box 110500, Gainesville, Florida 32611-0500.
*Corresponding author: marcelo@cpac.embrapa.br