Introduction
Peanut (Arachis hypogaea L.) is the most widely cultivated legume crop in northern Ghana because of its adaptation to the climatic conditions and its ability to grow relatively well on poor soils (Gascho and Davis, 1995; Kaleem, 1990). Peanut is a major source of protein in human diets and haulms are important components of livestock feed in Ghana (Marfo, 1997). Edible oil is also extracted from peanut seed and is used for local consumption. Peanut is effective in rotation with corn and other cereals due to biological nitrogen fixation by peanut (Gascho and Davis, 1995; Kaleem, 1990).
Peanut production in the savanna ecology is of great potential in the West African region (Schilling and Misari, 1992). However, weed interference is a major constraint to optimum production requiring considerable investment of human labor to minimize negative impact on pod yield (Akobundu, 1987; FAO, 1994; Frimprong, 2002). Yield loss due to weed interference in West Africa is estimated to be 50 to 80% (Akobundu, 1987; Carson, 1979). The relatively slow initial growth of peanut and morphological characteristics that do not allow it to grow above weeds influences susceptibility of peanut to early season weed interference (Akobundu, 1987; Subrahmaniyan et al., 2002). Although peanut is susceptible to weed interference, cultivars with running growth habit can suppress weeds by providing ground cover more quickly than cultivars with a more upright growth habit (Akobundu, 1980). Three timings of weed removal during the first 6 weeks after plant (WAP) are recommended to substantially reduce weed interference in peanut (Akobundu, 1987).
Despite importance of peanut to the economy of Ghana, current information on weeds and other management constraints is limited. In order to formulate and transfer effective and sustainable weed management strategies in peanut, documentation of distribution of weeds and characterization of agronomic and weed management practices are needed. The objectives of this study were to (1) document the distribution and density of weeds in peanut cropping systems, (2) document current weed management practices of farmers, and (3) assess yield response of peanut to current farmer weed management strategies.
Materials and Methods
Prior to sampling each field, a short survey (Table 1) was administered to farmers to document information on method of land preparation; peanut cultivar; date of planting; and method, timing, and frequency of weed control practices. Cropping systems implemented also were documented. The survey was conducted in 48 farmer fields during 2003, 2004, and 2005 between August and October by randomly evaluating four 1-m2 quadrants per field in the Northern, Upper East, and Upper West savannah regions of Ghana. Weed species, density of each species, weed biomass at harvest, peanut haulm production, and pod yield were recorded based on the 1-m2 quadrants. Latin binomial and authority for each weed over the duration of the survey are presented in Table 2. Weed and peanut haulm biomass were individually oven dried at 80 C for 48 h. Average weed occurrence in farms was calculated using the Summed Dominance Ratio (SDR) approach (Dangol, 1991).
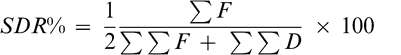 1
1
Results and Discussion
Characteristics of peanut cropping systems
The surface soil texture of the survey area generally consisted of silt loam (48%), sandy loam (48%) and silty clay loam (2%) (data not presented). Forty-four percent of farmers prepared land with a tractor while 37% used livestock to plow fields and 20% used traditional hoeing to prepare land for planting (data not presented). Nine and 18% of farmers planted peanut in April and May, respectively, while 41% planted in June and 32% planted in early July (data not presented). Planting coincided with the stability of rainfall and temperatures suitable for optimum for germination and seedling establishment (Abudulai et al., 2007).
Similar to a survey by Bolfrey-Arku et al. (2006) in southern Ghana, hoeing was the dominant weed control method practiced by 91% of farmers (data not presented). Fifty-two percent of farmers weeded only once, which is less than recommended to optimize peanut yield (Akobundu, 1987). Thirty-six percent of the farmers weeded their fields twice at 3 and 6 WAP while only 2% weeded three times. Bolfrey-Arku et al. (2006), in southern Ghana, reported that 33 and 59% of farmers weeded peanut fields once or twice, respectively. Herbicides were applied by 9% of farmers in northern Ghana (data not presented) compared with only 4% of farmers in southern Ghana (Bolfrey-Arku et al., 2006).
Generally, cereals such as corn preceded peanut, but peanut following peanut was also practiced. Farmers planted improved peanut cultivars with bunch or erect morphological characteristics, primarily the cultivar China, although some farmers planted locally-derived cultivars such as Bugla. Intercropping peanut with cereals was common but monocropping of peanut for several years prior to rotation with cereals was also practiced (data not presented). Although pod yield was the primary criteria for cultivar selection, cultivars were also selected by farmers based on their suppressive ability against weeds; the runner morphology of the improved cultivar Manipintar was preferred on these farms (Abudulai et al., 2007).
Prevalent weed species and their level of dominance
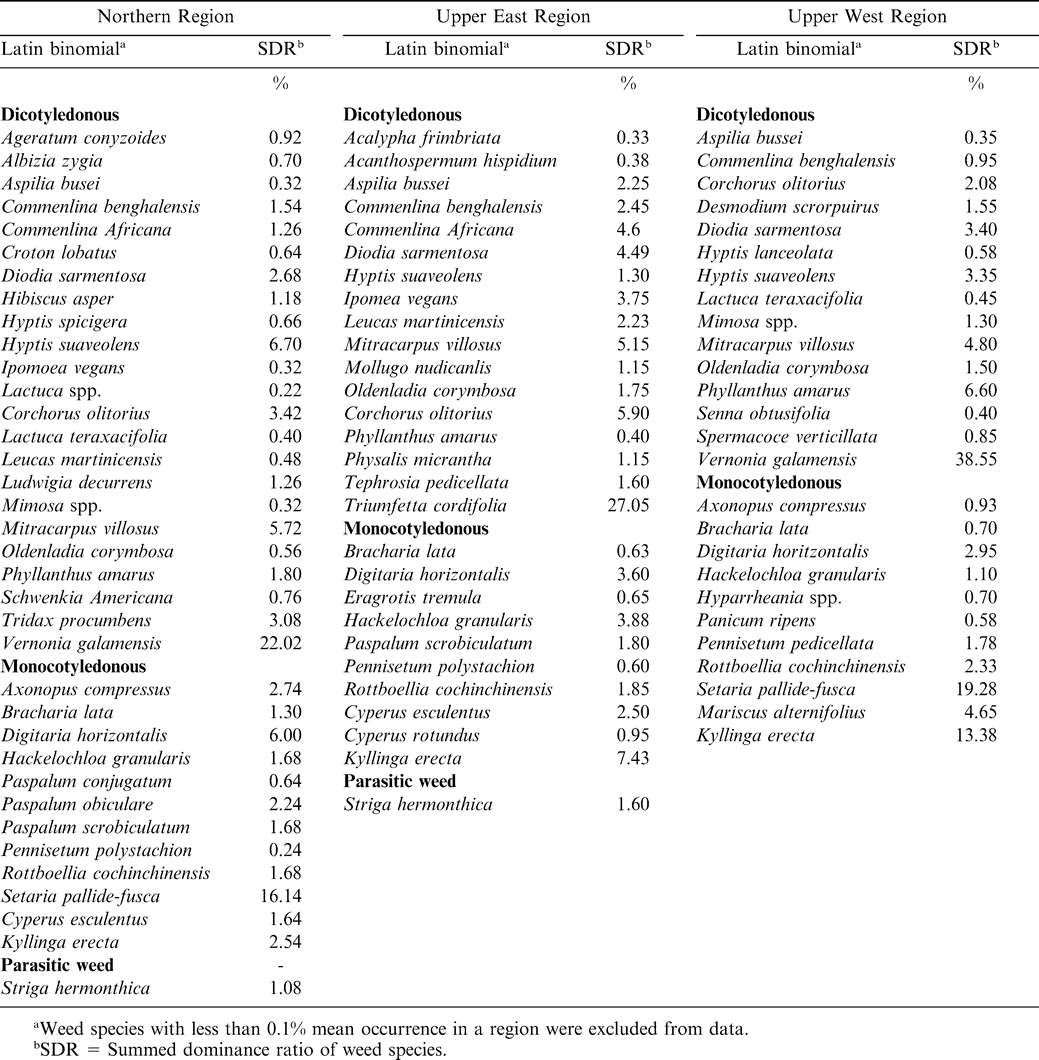
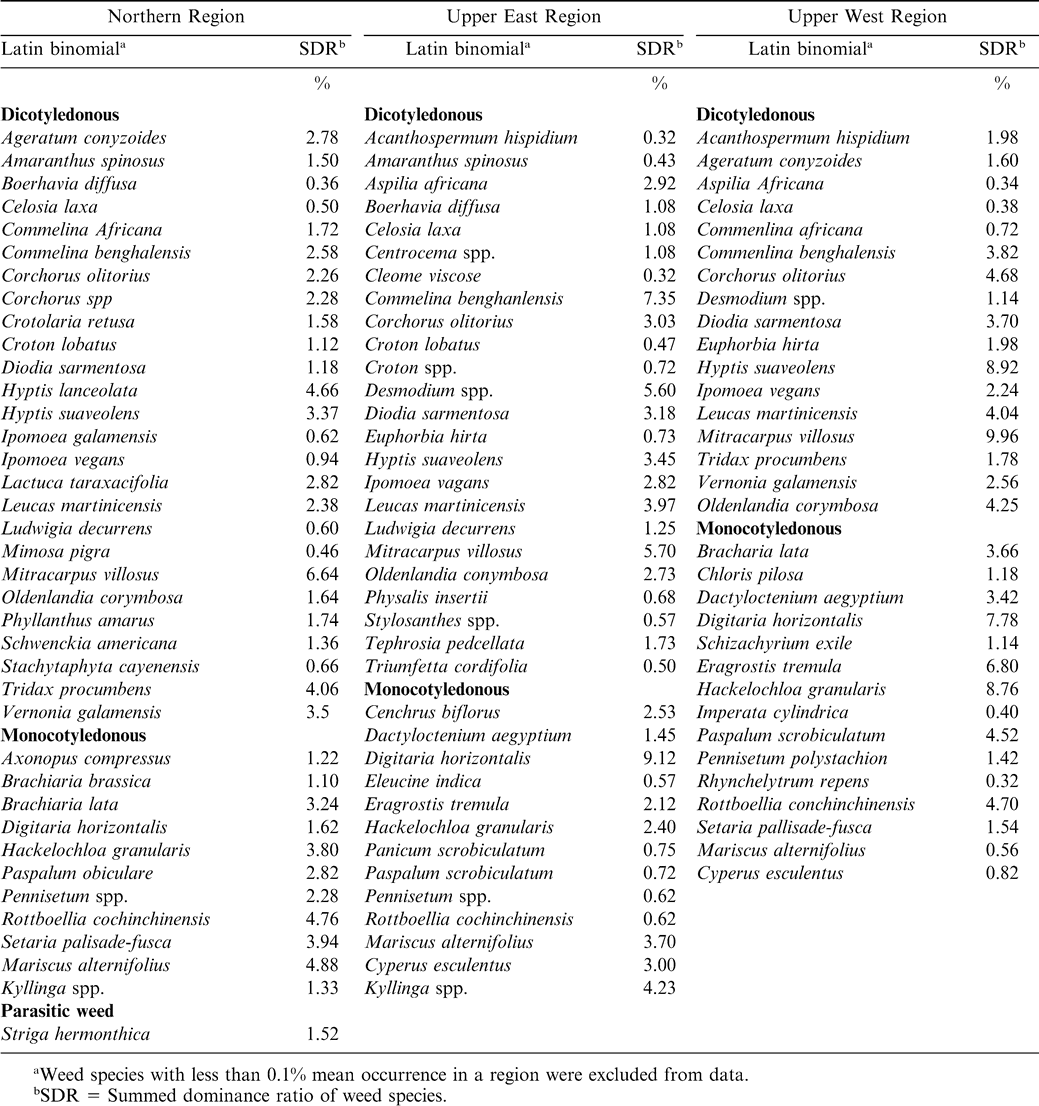
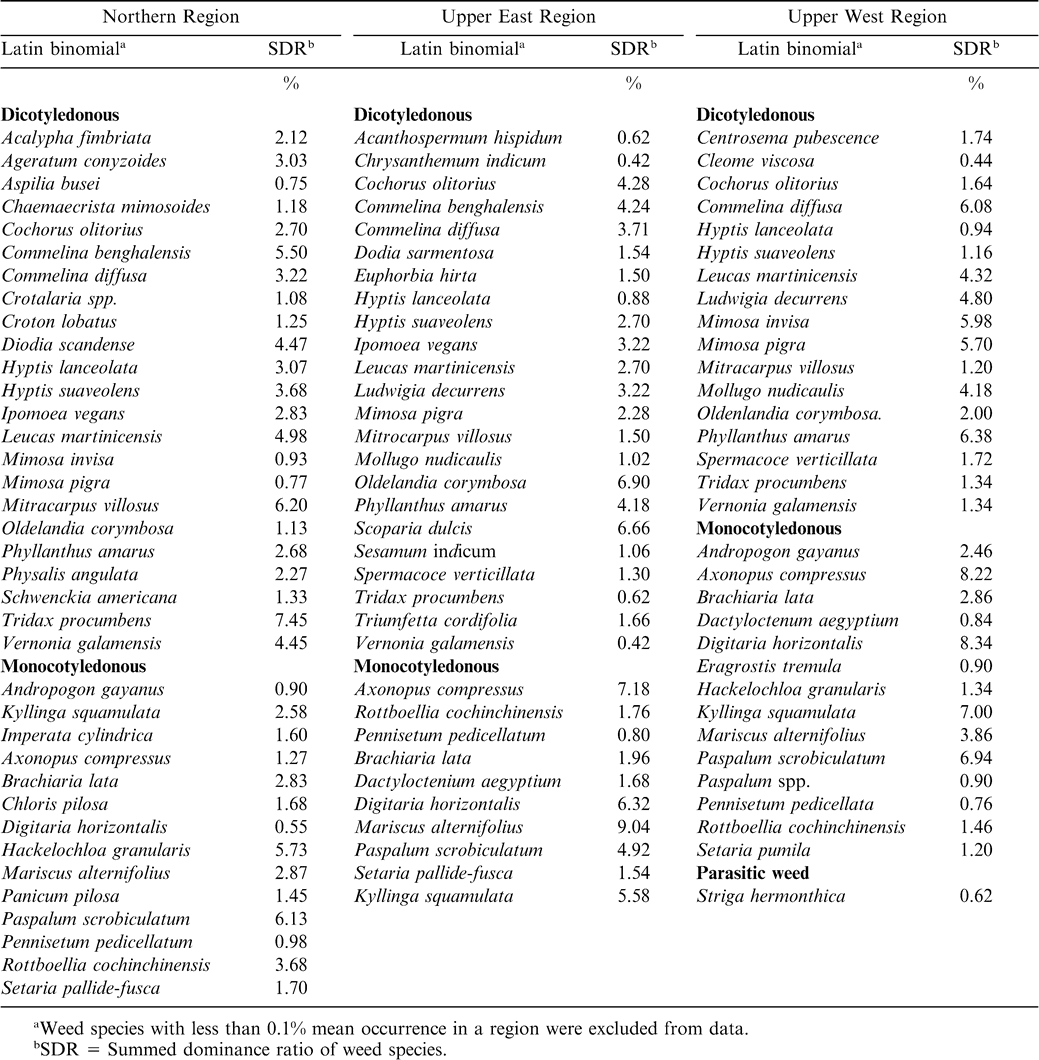
Weed species associated with peanut fields were highly diversified in species composition and density in the three regions (Tables 3– 5). Generally, dicotyledonous weeds dominated the flora followed by monocotyledonous weeds (grasses and sedges) and the parasitic weed Striga hermonthica. Average weed occurrence (at least 5% SDR) for the three years included the dicotyledonous weeds: Corchorus olitorius, Commelina benghalensis, Commelina diffusa, Desmodium spp., Hyptis suoveolens, Mimosa invisa, Mimosa pigra, Mitracarpus villosus, Oldenlandia corymbosa, Phyllanthus amarus, Scoparia dulcis, Tridax procumbens, Triumfeta cordiflora, and Vernonia galamensis; and the monocotyledonous weeds Axonopus compresus, Cyperus spp., Digitaria horizontalis, Eragrostis tremula, Hackelochloa granularis, Kyllinga spp., Paspalum scrobiculatum, Rottboellia cochinchinensis, and Setaria pallide-fusca.
During 2003, a total of 38 weed species were observed in the Northern Region, of which 23 were dicotyledonous, 14 were monocotyledonous (11 grasses and 3 sedges), and 1 parasitic weed (Table 3). In the Upper East Region, 29 species were observed with 17 being dicotyledonous, 11 monocotyledonous (7 grasses and 4 sedges), and 1 parasitic weed (Table 3). In the Upper West Region, the weed flora composed of 15 dicotyledonous and 11 monocotyledonous species (9 grasses and 2 sedges). Weed flora distribution in 2004 in the Northern Region consisted of 38 species (26 dicotyledonous, 11 monocotyledonous, and 1 parasitic weed) (Table 4). In the Upper East Region 24 dicotyledonous and 13 monocotyledonous were observed. In the Upper West Region a total of 33 species were distributed as 16 dicotyledonous and 17 monocotyledonous species (Table 4). In 2005, weeds in peanut cropping system in the surveyed area revealed 37 species (23 dicotyledonous and 14 monocotyledonous), 33 species (23 dicotyledonous and 10 monocotyledonous), and 32 species (17 dicotyledonous, 14 monocotyledonous, and 1 parasitic species) in the Northern, Upper East, and Upper West Regions, respectively (Table 5).
There were differences between levels of dominance of the various weed types at the different locations (data not shown). Locations with less weed dominance had a relatively more effective weed management with associated higher peanut haulm and pod yield than those with higher weed infestations. Due to their rapid proliferation and regeneration ability through stem cuttings , dicotyledonous weeds such as Commelina benghalensis and Commelina africana L. have been identified by the farmers as difficult to manage, as hoeing leads to breakage and multiplication of their vegetative propagules (Akobundu, 1987). Nitrogen fixation by the previous peanut crop could also stimulate growth of weeds (Kaleem, 1990). Despite the higher abundance of dicotyledonous weeds relative to monocotyledonous weeds, the latter poses a more serious threat to both peanut and farmer management efforts. Monocotyledonous weeds such as Digitaria horizontalis, Cynodon dactylon (L.) Pers., and Paspalum scrobiculatum L. regenerate quickly while Rottboellia cochinchinensis and Ishaemunm rogosum Salisb. are highly competitive (Akobundu, 1987). Most of the grasses are anatomically C-4 plants and express higher photosynthetic rates than peanut, especially under conditions of elevated temperatures and limited soil moisture (Akobundu, 1987). Although parasitic weeds, especially Striga hermonthica, form a small proportion of weeds in the peanut cropping system, they constitute the most important biotic weed pest in cereal-peanut intercropping systems (Parker and Riches, 1993).
Weed dry matter production in farmer fields at crop maturity
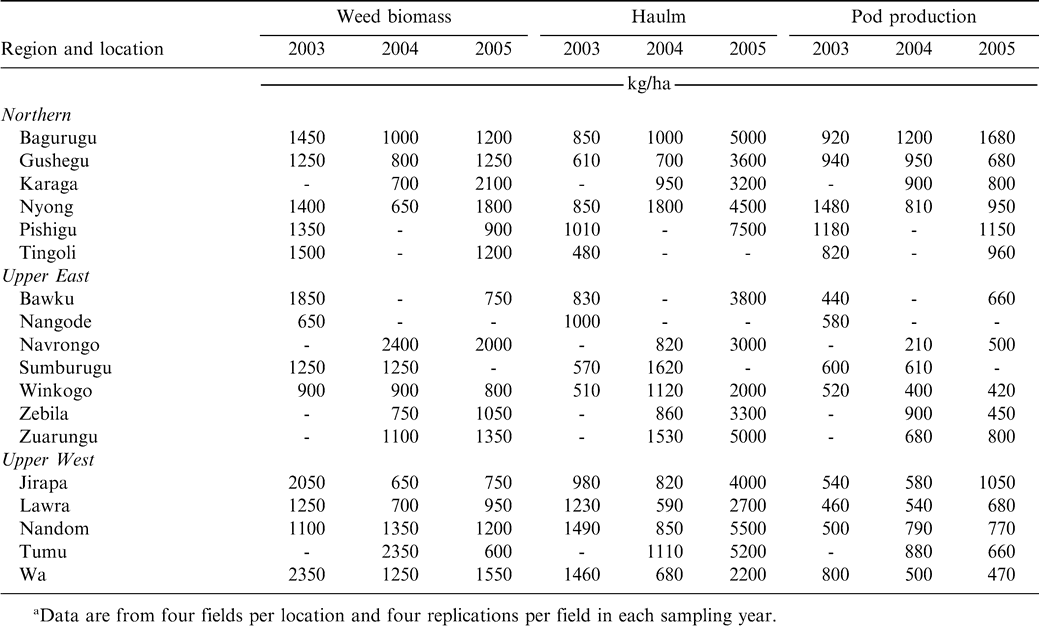
Despite weed management interventions using hoeing, weed dry matter production at crop harvest in farmers fields during the three years and ranged from 650 to 2350 kg/ha, 650 to 2400 kg/ha, 600 to 2100 kg/ha during 2003, 2004 and 2005, respectively (Table 6). In 2003, the highest weed biomass was produced at Wa in the Upper West Region but this was similar to results at all the locations in Northern Region and two other locations each in the Upper West and Upper East Regions. In 2004, weed biomass production was highest at Navrongo in the Upper East Region but this value was not different from weed production at three locations in the Upper West and two locations each in the Upper East and Northern Regions. In 2005, Karaga, in the Northern Region, recorded the highest weed biomass production but this was similar to most locations in the three regions.
Generally, the dominantly one hoe-weeding regime practiced by farmers in the survey region does not reduce weed interference sufficiently to optimize peanut yield (Akobundu, 1987). Akobundu (1987) observed that the growth of peanut is slow and therefore could require more than two hand weeding regimes to offer adequate protection of the crop against weed interference.
Peanut haulm production in farmer fields at crop maturity
Peanut haulm production in farmer fields in each year of the survey was low (Table 6). In 2003, haulm production was lowest and in the range of 510 to 1490 kg/ha (Table 6). Haulm production was higher and ranged from 590 to 1800 kg/ha during 2004 (Table 6). Production increased considerably and was high at all locations during 2005, ranging from 860 to 5500 kg/ha (Table 6). Higher rainfall during 2005 compared with 2003 or 2004 most likely contributed to higher yields (data not presented). The wide variation in peanut haulm in farmer fields in the three years is indicative of non-attainment of the potential haulm production for the zone. This shortfall in haulm production has the implication of limited appropriate agronomic crop management practices by farmers and could potentially reduce haulm used as feed for livestock production.
Peanut pod production in farmer fields
Peanut pod production in farmer fields exhibited significant variation among locations in each of the three years (Table 6). Pod production was low ranging from 520 to 1480 kg/ha during 2003, 210 to 1200 kg/ha during 2004, and 420 to 1680 kg/ha during 2005 (Table 6). Generally, pod production was highest in the Northern Region and Bagurugu gave the highest yield in 2005 due likely to a better production environment of rainfall (Kasei, 1988), soil fertility, and cropping history.
Relationship between weed biomass, peanut haulm, and pod production
Weed biomass and peanut haulm were negatively correlated with pod production in each year of the survey (Table 7). The correlation coefficients were moderate in 2003 for both weed biomass and haulm production and above average for haulm production in 2004. However, weed biomass gave consistently high correlation coefficients with pod yield in 2004 and 2005, while the coefficient of haulm with pod yield was high only in 2005.
The potential haulm yield of some peanut cultivars including a local cultivar at Nyankpala in the Northern Region ranged from 2180 to 3000 kg/ha (Marfo, 1997). The prospective kernel yield of peanut cultivars including some local entries in the zone could be high, in the range of 760 to 6200 kg/ha (Marfo, 1997). It is apparent that effective weed management regimes which remove weed interference increase peanut haulm and pod yield. FAO (1994) reported that in-row weed density of approximately 1 plant/m resulted in a yield loss of 32 to 45%. The dominance of annual weeds in the cropping system could confer additional interference due to similarity in root habit of the crop and the associated weeds of annual growth cycle (Akobundu, 1987). Consequently, weed biomass at crop harvest was high enough to indicate likely high degree of weed interference during crop development with subsequent reduction in peanut yield. Prolonged weed-peanut interference most likely was responsible for the low haulm and pod yield in these fields.
Peanut cultivars with high haulm production could offer some measure of weed suppression to reduce weed-peanut interference and promote higher haulm and pod yield (Akobundu, 1987). Reducing peanut haulm and pod yield due to weeds will require combination of management practices including improved seedbed preparation, timely planting, appropriate fertilizer application, timely weeding at the appropriate frequency, crop rotation, and fallow management. Furthermore, additional research should include greater capacity building of farmers and Agricultural Extension agents in integration of proven and alternative weed management tactics including selective herbicides to increase peanut production in northern Ghana (Akobundu, 1990; Labrada, 2002).
Acknowledgements
We are grateful to the United States Agency for International Development (USAID) Peanut CRSP (LAG-G-00-96-90013-00) for financial support. Appreciation is expressed to Fred Anaman, D. Opare-Atakora, Issifu Ramat, and Osman Iddrisu for technical assistance.
Literature Cited
Abudulai M. , Dzomeku I. K. , Salifu A. B. , Nutsugah S. K. , Brandenburg R. L. , and Jordan D. L. 2007 Influence of cultural practices on soil arthropods, leaf spot, pod damage, and yield of peanut in Northern Ghana. Peanut Sci 34 : 72 – 78 .
Akobundu I. O. 1980 Weed science research at the International Institute of Tropical Agriculture and research needs in Africa. Weed Sci 28 : 439 – 445 .
Akobundu I. O. 1987 Weed control in other food crops. In Weed Science in the Tropics: Principles and Practices A John Wiley and Sons publication Chichester 522 .
Akobundu I. O. 1990 The role of weed control in integrated pest management for tropical root and tuber crops. 23 – 29 In Hahn S. K. and Cravenness F. E. Integrated Pest Management for the Tropical Root and Tuber Crops IITA Ibadan, Nigeria .
Bolfrey-Arku G. , Owusu-Akyaw M. , Afu J. V. K. , Adu-Mensah J. , Anno-Nyako F. O. , Moses E. , Osei K. , Osei-Yeboah S. , Mochiah M. B. , Adama I. , Dankyi A. A. , Brandenburg R. L. , Bailey J. E. , and Jordan D. L. 2006 Survey of weed management in peanut (Arachis hypogaea L.) fields in southern Ghana, West Africa. Peanut Sci 33 : 90 – 96 .
Carson A. G. 1979 Weed competition and control in peanut (Arachis hypogaea L). Ghana J. Agric. Sci 9 : 169 – 173 .
Dangol D. R. 1991 Rice field weeds in Chitwan Valley, Nepal. 1627 – 1628 In Maclean J. Rice Science for a Better World – International Rice Research Institute Newsletter Los Baños, Philippines .
FAO
1994
Weed Management for Developing Countries.
30
–
34
335
–
339
In
Labrada R.
,
Casely J. C.
, and
Parker C.
FAO Plant Production and Protection Paper 120. Food and Agriculture Organization of the United Nations. Rome.
Frimpong A. 2002 Production guide on peanut and bambara peanut. 1 – 11 In Clottey V. A. , Alhassan A. Y. , Asante S. K. , and Owusu R. K. Savanna Agricultural Research Institute Tamale Ghana .
Gascho G. J.
and
Davis J. G.
1995
Soil fertility and plant nutrition.
383
–
418
In
Pattee H. E.
and
Stalker H. T.
Advances in Peanut Science
Am. Peanut Research and Education Society
Stillwater, OK
.
Labrada R. 2002 The need for improved weed management in rice. 306 In Dat Van and Duffy R., Proc. 20th Session of the International Rice Commission. July 23–26, 2002. Bangkok, Thailand.
Kaleem F. 1990 Assessment of nitrogen fixation by legumes and their relative contribution to a succeeding maize crop in the savannah zone of Ghana. 123 – 127 In Rudat H. , Mercer-Quarshie H. , von Bargen J. , Marfo K. O. , Salifu A. B. , Nyamekye A. L. , Afuakwa J. J. , Dekuku R. C. , and Sallah P. Y. K., Proc. of the Second Workshop on Improving Farming Systems in the Interior Savanna Zone of Ghana. 24–26th April, 1990. Nyankpala Agricultural Experiment Station, Nyankpala. Ghana.
Kasei C. N. 1988 The physical environment of Semi-arid Ghana. 350 – 354 In Challenges in Dryland Agriculture. A Global Perspective, Proceeding of the International Conference on Dry land Farming, August 1988, Amarillo/Bushland, Texas. Texas A and M University Press. College Station, Texas.
Marfo K. O. 1997 The performance and association among some important peanut yield traits in northern Ghana. 133 – 140 In Mercer-Quarshie H. , Marfo K. O. , Langyintuo A. S. , and Owusu R. K., Proc. of the 3rd Workshop on Improving Farming Systems in the Interior Savanna Zzone of Ghana. 27–29th April, 1993. Nyankpala Agricultural Experiment Station, Tamale, Ghana.
Parker C. and Riches C. R. 1993 Parasitic weeds of the world: Biology and control CAB International Wallingford UK 332 .
Schilling R. and Misari S. M. 1992 Assessment of research achievements in the savannah region in West Africa. 97 – 112 In Nigam S. N. Groundnut – a global perspective: Proceedings of International Workshop, 25–29 Nov. 1991. ICRISAT Centre, Patancheru, A.P. 502 324, India.
Subrahmaniyan K. , Kalaiselvan P. , and Arulmozih N. 2002 Weed control in groundnut (Arachis hypogaea L.) with polyethylene film mulching. Int. J. Pest Manage 48 : 261 – 264 .
Notes
Author Affiliations
1 Department of Agronomy, University for Development Studies, P. O. Box TL 1882, Tamale, Ghana.
2 CSIR-Savanna Agricultural Research Institute, P. O. Box 52, Tamale, Ghana.
3 Department of Entomology, North Carolina State University, Box 7613, Raleigh, NC 27695-7613.
4 Department of Crop Science, North Carolina State University, Box 7620, Raleigh, NC 27695-7620.
*Corresponding author: david_jordan@ncsu.edu