Introduction
Peanut grown in the Virginia-Carolina production region (V-C) generally require 140 to 160 days to reach optimum maturity and is generally seeded in early or mid-May to allow time for pods to reach maximum maturity (Jordan, 2007a; Sholar et al., 1995). Unlike peanut produced in the lower southeastern United States, delaying peanut planting in the V-C region until late May or June can result in substantial yield loss and lower market grades in years experiencing cooler temperatures in late September and October that minimize further maturation of pods (Carley et al., 2007; Mozingo et al., 1991). Additionally, stresses from biotic and abiotic sources can delay development of peanut, and in some cases these stresses can lower yield (Gascho and Davis, 1995; Shew et al., 1995; Wilcut et al., 1995). However, the indeterminate growth habit of peanut often allows compensation from early season stress, and if given sufficient recovery time and proper growing conditions, peanut yield and market grade factors may not be compromised (Mozingo et al., 1991).
Paraquat is registered for use within 28 days after peanut emergence to control small annual broadleaf weeds and grasses (Senseman, 2007; Wilcut et al., 1995). However, application later than 28 days after emergence is discouraged due to the potential for crop injury with less time for plant recovery (Johnson et al., 1993). The product label for paraquat does not address possible interactions of late planting and paraquat injury on pod yield and market grade characteristics. Growers in the V-C region have recently begun delaying planting from late April and early May until mid-May to minimize tomato spotted wilt of peanut (Brandenburg, 2007). Tomato spotted wilt virus is caused by a tospovirus that is transmitted to peanut by thrips, Frankliniella spp. However, because the planting window for peanut is relatively narrow in the V-C region compared with plantings in the lower southeastern United States, inclimate weather in mid May-that delays planting until late May or early June puts peanut at risk for yield loss and reduced market grade characteristics. Determining whether paraquat exacerbates risk associated with delayed planting has not been studied in the V-C region.
Damage from feeding by tobacco thrips can stunt peanut and reduce yield in North Carolina (Brandenburg et al., 1998). Growers often apply aldicarb in the seed furrow to control tobacco thrips (Brandenburg, 2007; Johnson et al., 1999). Concern about effects of tobacco thrips damage combined with injury from paraquat has been expressed, and determining interactions among these variables is important when formulating appropriate management strategies for weeds and tobacco thrips in peanut. Blenk et al. (1991) reported that thrips-induced injury reduced pod yield of the Virginia market type cultivar NC 7 while paraquat did not affect yield, and there was no cumulative effect of injury from thrips feeding and paraquat application. Herbert et al. (1991) suggested that the combined impact of thrips damage and herbicide injury could delay development of NC 7 sufficiently to reduce yield in Virginia and northeastern North Carolina. Brecke et al. (1996) and Funderburk et al. (1998) reported that cumulative increases in stress from thrips damage, herbicide injury, or soil moisture conditions that limit recovery from early season stress resulted in delayed pod development and reduced yield of the runner market types Florunner and Southern Runner. Research addressing interactions of aldicarb and paraquat has not been conducted with cultivars that have been released more recently and are currently being planted by most farmers in the V-C region (Jordan, 2007a). Research by Herbert et al. (1991) focused on the Virginia market type cultivar NC 7 (Wynne et al., 1979), and this cultivar is currently grown on less than 1% of hectares in the V-C region. The cultivar NC 7 has been replaced by some cultivars that require more days to reach optimum maturity.
Digging peanut when pods are at optimum reproductive development increases pod yield and improves market characteristics (Sholar et al., 1995). Most growers and their advisors in North Carolina and the lower southeastern United States use the hull scrape method to determine pod maturity (Williams and Drexler 1981). Pod mesocarp color is an indicator of pod maturation and can be used to determine if pest damage or injury from herbicide affect pod maturity.
Pesticide formulation can influence efficacy and crop response to postemergence herbicides (Klingman and Ashton, 1982; Koskinen and Harper, 1987). Manufacturers have marketed a variety of formulations of paraquat, and these currently include the paraquat formulations Gramoxone INTEON (Syngenta Crop Protection, Greensboro, NC), Firestorm (Chemtura Crop Protection, Williamstown, NJ), and Parazone (Makhteshim Agan Industries, Ltd., Houston, TX). Determining any differential peanut responses to various formulations of paraquat is important in developing appropriate strategies for use in peanut. Additionally, paraquat is often applied with other herbicides to improve control of emerged weeds, reduce peanut injury, and in some cases to provide contact and residual weed control (Wilcut et al., 1996). Determining compatibility of recently-marketed formulations is also important in developing weed management strategies associated with paraquat application to peanut.
Interactions of delayed planting and injury associated with paraquat has not been evaluated thoroughly for more recently released Virginia market type cultivars in North Carolina where delays in maturation can negatively affect pod yield and market grade characteristics. Cumulative injury to peanut from tobacco thrips feeding and paraquat application has not been clearly established. Therefore, research was conducted to determine peanut response to 1) the interaction of planting date and paraquat; 2) the interaction of aldicarb and paraquat on pod maturation, pod yield, and market grade characteristics; and 3) to paraquat formulations alone or with co-applied herbicides.
Materials and Methods
Methods Common to All Experiments
Experiments were conducted in conventionally-prepared raised seedbeds. Each peanut cultivar was seeded at a rate needed to establish in-row plant density of 13 plant/m. Plot size was two rows spaced 91 cm apart by 12 m. Seeds were placed 5 to 8 cm deep depending on soil moisture. Granular aldicarb (Temik 15G insecticide, Bayer CropScience, Research Triangle Park, NC) at 1.1 kg ai/ha was applied in the seed furrow prior to seed drop in all experiments except when aldicarb was a part of the treatment structure. Peanut was not irrigated.
Herbicides were applied using a CO2-pressurized backpack sprayer calibrated to deliver 140 L/ha using regular flat fan nozzles (Spraying Systems Co., Wheaton, IL). Fields at Lewiston-Woodville and Whiteville were fumigated with metam sodium at 18 L/ha 2 weeks prior to planting to control Cylindrocladium black rot (caused by Cylindrocladium crotalaria Bell and Sobers) (CBR) and suppress nematodes. Metam sodium was injected during the subsoiling and bedding process 30 cm below seed placement. All other production and pest management practices were held constant across the experiment and were based on Cooperative Extension Service recommendations (Brandenburg, 2007; Jordan, 2007a 2007b; Shew, 2007).
Entire test areas were maintained weed free by applying pendimethalin (Prowl herbicide, BASF Corp., Research Triangle Park, NC) at 1.1 kg ai/ha preplant incorporated (PPI), S-metolachlor (Dual Magnum herbicide, Syngenta Crop Protection, Research Triangle Park, NC) at 1.1 kg ai/ha preemergence (PRE), and clethodim at 0.14 kg ai/ha (Select 2EC herbicide, Valent USA Corp., Walnut Creek, CA) plus crop oil concentrate (Agri-Dex crop oil concentrate, Helena Chemical Co., Memphis, TN) at 1.0% (v/v) were applied postemergence (POST) in late July of both years. Escaped broadleaf weeds were removed by hand when needed to maintain weed-free plots. Additional POST herbicides were applied to control broadleaf weeds and will be listed for each experiment. Nonionic surfactant (Induce nonionic surfactant, Helena Chemical Co., Memphis, TN) at 0.125% (v/v) was applied with paraquat in all experiments. Adjuvant was not applied with 2,4-DB.
In experiments where pod yield and market grade characteristics were determined, peanut pods were dug and vines inverted for each planting date based on pod mesocarp color for the no-herbicide control (Jordan et al., 2005; Williams and Drexler, 1981). Peanut was threshed 4 to 7 days after digging and dried to final moisture of 8%. Percentages of extra large kernels (%ELK), total sound mature kernels (%TSMK), and farmer fancy pods (%FP) were determined using Federal and State Cooperative grading criteria (USDA, 2005).
Interactions of Planting Date and Herbicide Treatment
The experiment was conducted in North Carolina during 2003 and 2004 at the Peanut Belt Research Station located near Lewiston-Woodville on a Norfolk loamy sand soil (fine-loamy, siliceous, thermic, Typic Paleudults). Treatments consisted of a factorial arrangement of planting date (4) by herbicide (2). Planting dates were May 5, May 15, and May 25 during both years and June 4, 2003 or June 6, 2004. Herbicides were no-herbicide or paraquat (Gramoxone MAX, Syngenta Crop Protection, Greensboro, NC) at 0.14 kg ai/ha was applied 24 to 28 days after peanut emergence. The Virginia market-type cultivar VA 98R (Mozingo et al., 2000) was used. The experimental design was a randomized complete block with a split plot treatment arrangement with planting date serving as the whole plot unit and herbicide treatment serving as subplot units in the planting date experiment. Data for all variables were subjected to analyses of variance appropriate for treatment structure for each experiment using appropriate error terms for fixed and random effects (Carmer et al., 1989; SAS, 2006). Means of significant main effects and interactions were separated using Fisher's Protected LSD test at p ≤ 0.05.
Interactions of Aldicarb and Paraquat
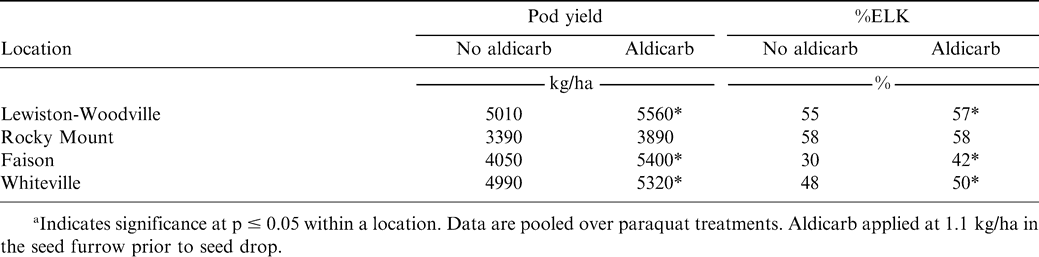
The experiment was conducted in North Carolina during 2003 at Lewiston-Woodville and during 2004 in two separate fields at both Lewiston-Woodville and at the Upper Coastal Plain Research Station located near Rocky Mount. The experiment was also conducted during 2005 at Lewiston-Woodville, Rocky Mount, and in grower's fields located near Faison and Whiteville. A total of 9 sites were included in the experiment. The peanut cultivar NC-V 11 (Wynne et al., 1991) was established as described previously. Soil at Rocky Mount was a Goldsboro sandy loam (fine-loamy, siliceous, thermic Aquic Paleudalts). Soil at Faison was Autryville fine sandy loam (fine-loamy, siliceous, thermic, Typic Paleudults) and the soil at Lewiston-Woodville and Whiteville was Norfolk loamy sand. Fields at Lewiston-Woodville and Whiteville during all years were fumigated with metam sodium as described previously to control CBR and suppress plant parasitic nematodes. Fields at Faison and Rocky Mount did not have a history of CBR or nematodes and were not fumigated.
Treatments during all years consisted of a factorial arrangement of 2 levels of aldicarb and 2 levels of herbicide. Aldicarb rates were 0 and 1.1 kg/ha applied in the seed furrow. Paraquat applied 28 days after peanut emergence following both aldicarb treatments during all years. During 2003 and 2004, 2,4-DB (Butyrac 200 product label, Agri Star, Ankeny, IA) was also applied in mid-August following aldicarb treatments. A no-herbicide control was not included in this experiment. However, in 2005 a no-herbicide control was included rather than 2,4-DB. Baughman et al. (2002) and Jordan et al. (2003) reported that 2,4-DB did not affect yield of runner and Virginia market type peanut when applied at rates that exceeded those recommended by the manufacturer or when applied 1, 3, or 5 wk prior to digging at optimum maturity when peanut was planted in early to mid-May. At Lewiston-Woodville and Rocky Mount during all years, acifluorfen (0.28 kg ai/ha) plus bentazon (0.84 kg ai/ha) (Storm herbicide, BASF Corp., Research Triangle, NC) plus 2,4-DB (0.14 kg/ha) were applied over the entire test areas 2 weeks after paraquat application to control escaped weeds. Nonionic surfactant was included at 0.25% (v/v). At Faison and Whiteville, imazapic (Cadre herbicide, BASF Corp., Research Triangle Park, NC) at 70 g ai/ha plus nonionic surfactant at 0.25% (v/v) were applied 2 wks after paraquat application to control escaped weeds.
Visual estimates of tobacco thrips injury from feeding were determined based on a scale of 0 to 5 where 0 = no visible feeding, 1 = noticeable feeding but no stunting, 2 = noticeable feeding and moderate stunting, 3 = severe feeding and severe stunting, 4 = severe feeding and stunting and death of terminals, and 5 = plant death. Visual estimates were not recorded for each plot but represent an average assessment of thrips damage from plots not receiving paraquat but receiving aldicarb or no in-furrow insecticide. Failure to record data for each plot was an oversight in the experiment and limits ability to make early season conclusions relative to interactions among damage from thrips feeding and injury from paraquat. Visual estimates of injury were recorded the day paraquat was applied. Within 1 week prior to peanut inversion during 2003 and 2004 but not 2005, approximately 100 pods were removed from 3 plants in each plot and were subjected to mesocarp color determination (Williams and Drexler, 1981). Pods were placed into 5 categories including white, yellow, orange, brown, and black mesocarp color designations. Because of the subjective nature of defining colors using this procedure, pods in the brown and black categories were combined, and this fraction was considered to be pods in the development stage ready for digging and is referred to as the percentage of mature pods (%MP).
The experimental design for all other experiments was a randomized complete block. Each subplot was replicated four times. Data for all variables were subjected to analyses of variance appropriate for treatment structure for each experiment using appropriate error terms for fixed and random effects (Carmer et al., 1989; SAS, 2006). Means of significant main effects and interactions were separated using Fisher's Protected LSD test at p ≤ 0.05.
Comparison of Paraquat Formulations
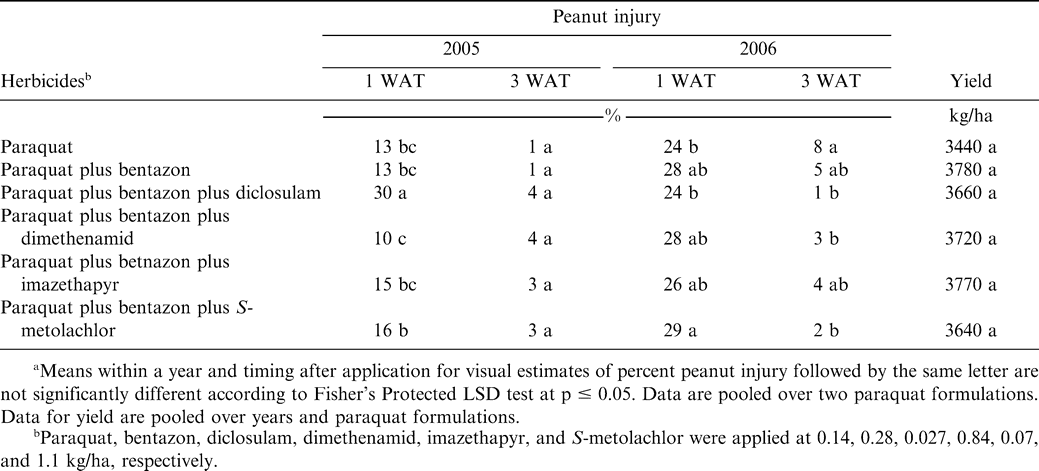
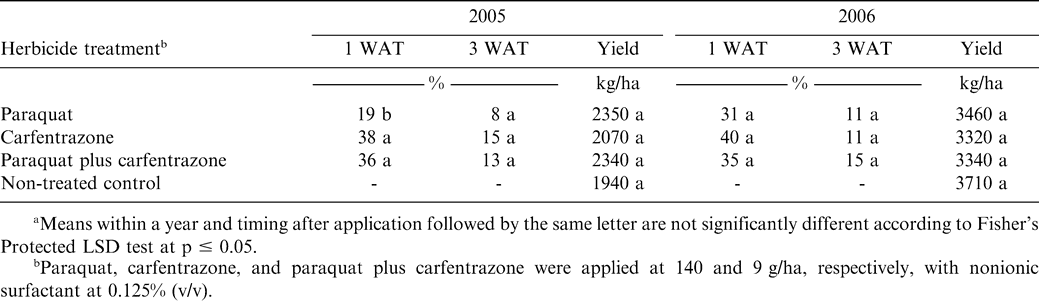
The experiment was conducted at the Peanut Belt Research Station during 2005 and 2006 on the Norfolk loam sand soil described previously. The cultivar Gregory (Isleib et al., 1999) was planted as described previously. Metam sodium was applied as described previously to control CBR and suppress nematodes. Paraquat at 0.14 kg/ha as Gramoxone MAX (220 g/L, Syngenta Crop Protection Co., Greensboro, NC) or Gramoxone INTEON (175 g/L, Syngenta Crop Protection Co., Greensboro, NC) was applied alone or with bentazon (Basagran herbicide, BASF Corp., Research Triangle Park, NC), bentazon plus diclosulam (Strongarm herbicide, Dow AgroScience, Indianapolis, IN), bentazon plus dimethenamid (Outlook herbicide, BASF Corp., Research Triangle Park, NC), bentazon plus imazethapyr (Pursuit herbicide, BASF Corp., Research Triangle Park, NC), and S-metolachlor (Dual Magnum herbicide, Syngenta Crop Protection, Greensboro, NC) 1 week after peanut emergence. Bentazon, diclosulam, dimethenamid, imazethapyr, and S-metolachlor were applied at 0.28, 0.027, 0.84, 0.07, and 1.1 kg/ha, respectively. In a separate set of experiments conducted during 2006 at Lewiston-Woodville and Rocky Mount and during 2007 at Lewiston-Woodville, peanut injury following application of the paraquat formulations Gramoxone INTEON and Firestorm were compared when each formulation was applied at 0.14 kg/ha. In a final set of experiments conducted during 2005 and 2006, paraquat at 0.14 kg/ha (Gramoxone INTEON) was applied alone or with carfentrazone at 9 g ai/ha (Aim herbicide, FMC Corp., Parsippany, PA). Carfentrazone was also applied alone. A non-treated control was included in all experiments. Lactofen (Cobra 2EC herbicide, Valent USA Corp., Walnut Creek, CA) at (0.22 kg ai/ha) plus 2,4-DB at 0.28 kg/ha plus crop oil concentrate at 1.0% (v/v) were applied over the entire test area 5 weeks after planting to control escaped broadleaf weeds in both experiments at Lewiston-Woodville. Acifluorfen (0.28 kg/ha) plus bentazon (0.84 kg/ha) plus 2,4-DB (0.28 kg/ha) was applied at Rocky Mount over the entire test area to control escaped broadleaf weeds. Nonionic surfactant at 0.25% (v/v) was included with acifluorfen plus bentazon plus 2,4-DB.
Visual estimates of percent peanut injury were recorded for all plots in all experiments 1 and 3 weeks after treatment (WAT) using a scale of 0 to 100% where 0 = no injury and 100 = plant death. Foliar chlorosis, necrosis, and plant stunting were considered when making the visual ratings. Peanut pods were harvested as described previously in the experiment that included paraquat alone or co-applied herbicides. Pod yield was not determined in the experiment comparing the paraquat formulations Gramoxone INTEON and Firestorm. The experimental design for all other experiments was a randomized complete block. Each subplot was replicated four times. Data for all variables were subjected to analyses of variance appropriate for treatment structure for each experiment using appropriate error terms for fixed and random effects (Carmer et al., 1989; SAS, 2006). Means of significant main effects and interactions were separated using Fisher's Protected LSD test at p ≤ 0.05.
Results and Discussion
Interactions of Planting Date and Herbicide Treatment
Although not quantified for each plot, peanut expressed symptoms within the first few weeks after application of paraquat that are characteristic for this herbicide (data not presented). Foliar injury and symptoms following application of both paraquat and 2,4-DB were transient in nature (Senseman, 2007; Wilcut et al., 1995).
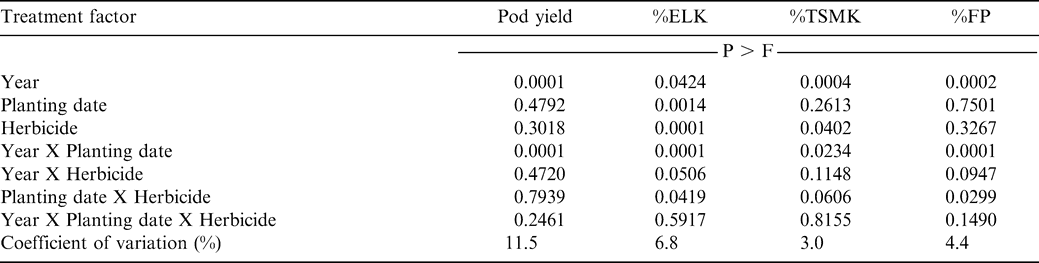
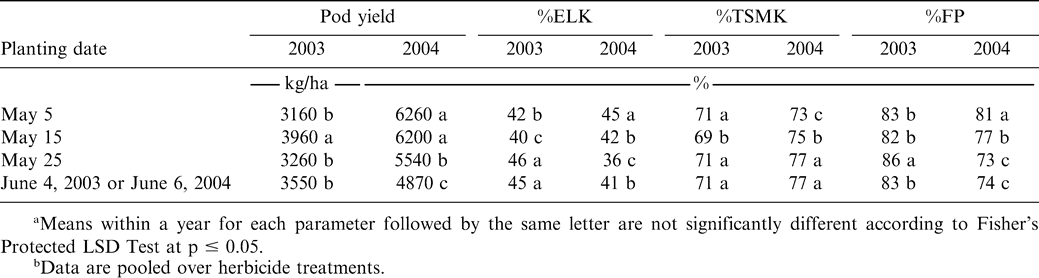
Interactions of year X planting date X herbicide and planting date X herbicide were not significant for pod yield (Table 1). Additionally, main effects of planting date and herbicide treatments were not significant for pod yield (Table 1). While the interaction of year X herbicide was not significant, the interaction of year X planting date was significant (Table 1). Pod yield was lower when peanut was planted May 5 or 25 or in early June compared with yield of peanut planted May 15 during 2003 (Table 2). In contrast, yield during 2004 was higher when peanut was planted May 5 or 15 compared with the later plantings. The optimum planting period for peanut in North Carolina is during early to mid-May (Carley et al., 2007; Jordan, 2007a). Concern over TSWV caused by a tospovirus, has resulted in recommendations to plant peanut between May 10 and 20 (Brandenburg, 2007; Johnson et al., 1993). Visual symptoms of TSWV were not apparent in these experiments during either year, and these data indicate that planting on May 15 provided high yields during both years even in the absence of TSWV.
The interaction of year X planting date X herbicide was not significant for %ELK (Table 1). However, the year X planting date interaction was significant (Table 1). Percent ELK was lower when peanut was planted May 5 or 15 compared with planting later in the 2003 season (Table 2). In contrast, the highest %ELK during 2004 was observed for the early planting date. While the interaction of planting date X herbicide was not significant for %ELK, the interaction of year X paraquat was significant for this market grade factor (Table 1). Paraquat reduced %ELK from 45% (non-treated peanut) to 41% in 2003 but did not affect %ELK during 2004 (data not presented in tables). Previous research suggests that paraquat can reduce %ELK compared with non-treated peanut by increasing the proportion of other kernels (Knauft at al., 1990), which may indicate that the herbicide affected grade by delaying maturity.
While the interaction of year X planting date X herbicide was not significant for %TSMK, the year X planting date interaction was significant (Table 1). Lower %TSMK was noted for peanut planted May 15 compared with planting earlier than this date or when planting May 25 or June 4 during 2003 (Table 2). In 2004, higher %TSMK was noted when peanut was planted May 25 or June 6 compared to planting May 5 or 15.
The interaction of the year X planting date X herbicide was not significant for %FP (Table 1). Year X herbicide was also not significant for %FP, however, %FP was affected by the interaction of year X planting date and planting date X herbicide (Table 1). During 2003, %FP was highest for the May 25 planting date (Table 2). However, the highest %FP was noted for the May 5 planting date during 2004. When pooled over years and planting dates, pod yield following paraquat did not differ from non-treated peanut (4630 vs. 4460 kg/ha, data not shown in tables). Likewise, %TSMK and %FP was not affected by paraquat and ranged from 73 to 74% and 78 to 80%, respectively (data not shown in tables).
A major objective of this research was to determine if peanut planted on different dates in the V-C region varied in response to paraquat applied within the manufacturer's recommendation. Lack of interactions involving planting date and herbicide in our study during two years with considerable differences in response to planting date suggests that growers can expect peanut to respond similarly to paraquat regardless of planting date.
Interactions of Aldicarb and Paraquat
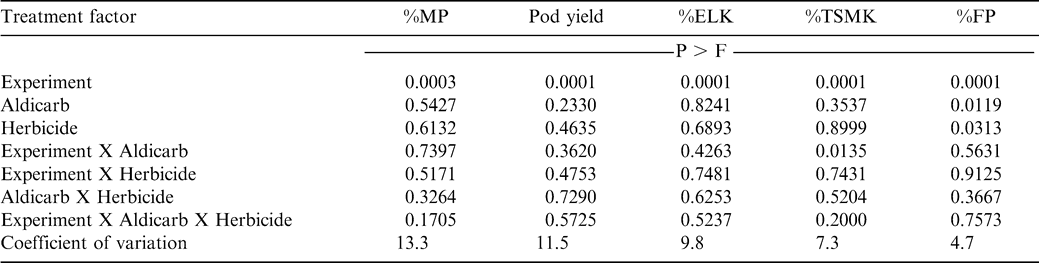
The interaction of site X aldicarb X herbicide was not significant for percentage of mature pods, pod yield, %ELK, %TSMK, and %FP at the sites conducted during 2003 and 2004 (Table 3). Aldicarb X herbicide and site X herbicide were also not significant (Table 3). However, the interaction of site X aldicarb was significant for %TSMK (Table 3). At 4 of the 5 sites there was no difference in %TSMK when comparing aldicarb-treated peanut with peanut not treated with aldicarb (data not shown in tables). However, %TSMK was lower at Lewiston-Woodville at one site during 2004 when aldicarb was applied (77 vs. 80%, data not shown). It is suspected that feeding of tobacco thrips may have impacted early season growth sufficiently to delay pod development. A lower %TSMK is indicative of delayed kernel development.
When pooled over 5 experiments, %MP (65 to 70%), pod yield (4180 to 4540 kg/ha), %ELK (50 to 51%), and %TSMK (74 to 75%) when comparing among combinations of aldicarb and paraquat (data not shown in tables). While none of the interactions were significant that included experiment as a factor, herbicide and aldicarb main effects were significant for %FP (Table 3). However, the interaction of these treatment factors was not significant. The %FP increased from 81 to 83% when aldicarb was applied (data not shown in tables). Additionally, the %FP following application of paraquat and 2,4-DB was 81 and 84%, respectively (data not shown in tables). Damage from tobacco thrips, in absence of aldicarb, and injury from paraquat compared with 2,4-DB most likely resulted in a lower %FP. A lower %FP reflects smaller pods and is indicative of less development and delayed maturation of pods.
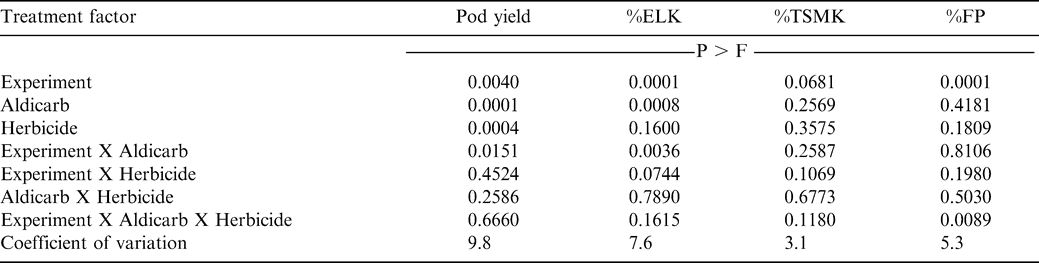
In the 2005 experiments, the interaction of experiment X aldicarb X paraquat was not significant for pod yield, %ELK, or %TSMK (Table 4). However, this interaction was significant for %FP. In contrast to results from experiments conducted during 2003 and 2004, the interaction of experiment X aldicarb was significant for pod yield and %ELK during 2005 (Table 4). Applying aldicarb increased pod yield at Lewiston-Woodville, Faison, and Whiteville compared with the no-aldicarb treatment (Table 5). Paraquat did not affect %ELK, %TSMK, or %FP nor did paraquat interact with aldicarb in experiments during 2005. The %ELK was higher when aldicarb was applied in these experiments at 3 of 4 locations (Table 5). There was no difference in pod yield or %ELK when comparing aldicarb treatments at Rocky Mount.
The main effect of herbicide treatment was significant for pod yield (Table 4). When pooled over experiments and aldicarb treatments, pod yield of peanut treated with paraquat was 4480 kg/ha compared with yield of 4950 kg/ha when paraquat was not applied (data not shown in tables). These data indicate that under conditions that do not favor recovery from early season injury, yield loss from paraquat can occur. Johnson et al. (1993) reported that paraquat reduced peanut yield. In contrast, other research (Wilcut et al., 1995) indicated that peanut recovers from early season injury from paraquat, and yield is not affected. Blenk et al. (1991) reported that thrips-induced injury reduced pod yield of the Virginia market type cultivar NC 7 while paraquat did not affect yield; there was no cumulative effect of injury from thrips feeding and paraquat application. In contrast, Brecke at al. (1996) and Funderburk et al. (1998) reported that two or more early season stresses from thrips feeding, herbicide damage, or dry weather could delay pod maturation and reduce yield of runner market type peanut. However, in their research interactions of aldicarb and paraquat were not compared. Herbert et al. (1991) reported that the combined effects of thrips damage in absence of aldicarb or carbaryl and injury from acifluorfen, paraquat, or pyridate may be sufficient to slow peanut development and prevent plants from recovering rapidly enough to prevent yield loss of Virginia market type peanut planted in Virginia or northeastern North Carolina.
When comparing within experiments, the interaction of aldicarb X paraquat was significant for %FP at Faison. At this location, the highest %FP was in the treatment with aldicarb, but no paraquat, while the lowest %FP occurred when both aldicarb and paraquat were not applied (data not shown in tables). Aldicarb and paraquat did not affect %FP at Lewiston-Woodville, Rocky Mount, and Whiteville (data not shown in tables).
Although damage from tobacco thrips feeding and paraquat applications was not more severe during 2005 than the previous two years, environmental conditions most likely contributed to the relatively poor ability of peanut to recover from tobacco thrips damage during 2005. Heat unit accumulation at Lewiston-Woodville during 2003, 2004, and 2005 from peanut emergence to digging was approximately 2648, 2788, and 2739, respectively (13 C base and 35 C ceiling, data not shown in tables). This quantity of heat unit accumulation is sufficient for adequate pod maturation to optimize pod yield and quality if soil moisture is not limiting for the cultivar NC-V 11 (Jordan, 2007a). However, less rainfall occurred during this time period at Lewiston-Woodville during 2005 (25 cm) than 2003 (67 cm) or 2004 (70 cm) (data not shown). Rainfall was also more limited from June through September at Rocky Mount during 2005 (39 cm) compared with 2004 (57 cm). Rainfall amount near the experiment at Faison and Whiteville during this time period was 36 and 44 cm, respectively (data not shown in tables). Ability of peanuts to recover from early season thrips damage is often associated with weather conditions later in the season (Marios and Wright, 2003). Growing conditions during 2003 and 2004 were considered ideal for peanut growth and development and pod maturation, and even though early season damage was present, peanut recovered and transient effects on foliage did not translate negatively on pod yield, pod maturity, or market grade characteristics. High yields during 2005 may have been a reflection of locations and field history of peanut production rather than conditions affecting peanut recovery from early season thrips damage. At Faison and Whiteville during 2005, peanut had not been planted in these fields in previous years, and therefore yields overall may have been higher due to less disease or some other factor associated with peanut growth other than dry weather (Shew, 2007). Although lack of aldicarb could result in damage from nematodes, fields in these experiments did not have a history of nematode populations, and in many cases fields were fumigated with metam sodium, a soil fumigant that reduces nematode populations (Shew, 2007).
Comparison of Paraquat Formulations
The main effect of co-applied herbicide and the interaction of year by co-applied herbicide were significant 1 WAT. However, main effect of paraquat formulation and paraquat formulation by complement herbicide were not significant. Although main effects of paraquat formulation, complement herbicide, and the interaction of paraquat formulation by complement herbicide were not significant, the interaction of year by complement herbicide was significant 3 WAT. The interaction of year by paraquat formulation by complement herbicide was not significant at either rating.
Peanut injury 1 WAT following application of paraquat was 21% (Gramoxone INTEON) and 22% (Gramoxone MAX) when pooled over years and co-applied herbicides (data not shown). These respective formulations of paraquat injured peanut 4 and 3% by 3 WAT and pod yield was 3300 and 3250 kg/ha (data not shown in tables).
When pooled over paraquat formulations, paraquat plus bentazon plus diclosulam was the most injurious herbicide treatment during 2005 when evaluated 1 WAT (Table 6). While the combination of paraquat plus bentazon plus S-metolachlor was more injurious than paraquat plus bentazon plus dimethenamid, all other herbicide combination injured peanut similarly. However, injury 3 WAT was 4% or less and no differences were noted among all herbicide treatments. In 2006, the most injurious treatment consisted of paraquat plus bentazon plus S-metolachlor 1 WAT (Table 6). This treatment was more injurious than paraquat alone or paraquat plus bentazon plus diclosulam. By 3 WAT, peanut injury was 8% or less with paraquat alone being more injurious than paraquat plus bentazon applied with diclosulam, dimethenamid, or S-metolachlor. In contrast to differences noted among herbicide treatments for visual injury, there were no differences in peanut yield when comparing co-applied herbicides or combinations of paraquat formulations and co-applied herbicides. Pod yield ranged from 3440 to 3780 kg/ha when pooled over years and paraquat formulations (Table 6).
In a second set of experiments, peanut injury did not differ when comparing the paraquat formulations Gramoxone INTEON with Firestorm (data not shown). Peanut injury 1 WAT ranged from 30 to 34% (Lewiston-Woodville, 2005), 23% (Rocky Mount, 2005), and 25–26% (Lewiston-Woodville, 2006) (data not shown in tables). By 3 WAT, injury was 10% or less at all locations (data not shown in tables). Prostko (2006) reported that peanut response to the paraquat formulations Gramoxone MAX and Gramoxone INTEON did not differ.
Additionally, pod yield was similar when comparing these herbicide treatments to non-treated peanut (Table 7). These experiments were conducted under two environments that were considerably different with respect to rainfall. During 2005, rainfall was limited and is reflected in yields ranging from 1940 to 2350 kg/ha (Table 7). In contrast, adequate and timely rainfall occurred in 2006 and is reflected in pod yield ranging from 3320 to 3710 kg/ha (Table 7).
Collectively, these experiments indicate that peanut most likely will respond the same way to paraquat when peanut are planted at dates ranging from early May through early June when paraquat is applied within label recommendations (application within 28 days after peanut emergence). This finding is beneficial in terms of developing recommendations to control weeds and to manage tomato spotted wilt in the V-C region for Virginia market types. These data suggest that practitioners should be able to alter planting date in a manner that minimizes TSWV while still using paraquat to control weeds. Although damage from tobacco thrips feeding in absence of aldicarb and injury from paraquat affected pod yield and market grade characteristics in some but not all experiments, responses to these pesticides were independent. These data also indicate that peanut response to more recently marketed formulations of paraquat most likely will be similar to responses noted with previous formulations. The recently marketed formulation Gramoxone INTEON was compatible with herbicides that are often co-applied with paraquat when considering peanut response under weed-free conditions.
Acknowledgements
The North Carolina Peanut Growers Association, Inc.; the National Peanut Board, the North Carolina Biotechnology Center (grant 2002-CFG-8012); the USAID Peanut CRSP (grant LAG-G-00-96-0013-00), and Syngenta Crop Protection provided financial support of this research. Dewayne Johnson, Carl Murphy, Brenda Penny, Curtis Fountain, and Michael Shaw and staff at the Peanut Belt Research Station and the Upper Coastal Plain Research Station provided technical assistance. Appreciation is also expressed to Vic Swinson and Charles Lennon for allowing these experiments to be conducted on their farms.
Literature Cited
Baughman T. A. , Grichar W. J. , and Jordan D. L. 2002 Tolerance of Virginia-type peanut to different application timings of 2,4-DB. Peanut Sci 29 : 126 – 128 .
Blenk E. S. , Linker H. M. , and Coble H. D. 1991 Interaction of tobacco thrips (Frankliniella fusca), paraquat, and mechanical defoliation on peanut (Arachis hypogaea) growth, quality, and yield. Proc. Am. Peanut Res. and Ed. Soc 22 : 76 .
Brandenburg R. L. 2007 Peanut insect management. 2007 Peanut Information. North Carolina Coop. Ext. Serv. Ser. AG-331 75 – 93 in 132 .
Brandenburg R. L. , Herbert D. A. , Sullivan G. A. , Naderman G. C. , and Wright S. F. 1998 The impact of tillage practices on thrips injury of peanut in North Carolina and Virginia. Peanut Sci 25 : 27 – 31 .
Brecke B. J. , Funderburk J. E. , Teare I. D. , and Gorbet D. W. 1996 Interactions of early-season herbicide injury, tobacco thrips injury, and cultivar on peanut. Agron. J 88 : 14 – 18 .
Carley D. S. , Jordan D. L. , Dharmasri C. L. , Sutton T. B. , Brandenburg R. L. , and Burton M. G. 2008 Peanut (Arachis hypogea L.) response to planting date and potential of canopy reflectance as an indicator of pod maturation. Agron. J 100 : 376 – 380 .
Carmer S. G. , Nyquist W. E. , and Walker W. M. 1989 Least significant differences for combined analyses of experiments with two- or three- factor treatment designs. Agron. J 81 : 665 – 672 .
Funderburk J. E. , Gorbet D. W. , Tearre I. D. , and Stavisky J. 1998 Thrips injury can reduce peanut yield and quality under conditions of multiple stress. Agron. J 90 : 563 – 566 .
Gascho G. J.
and
Davis J. G.
1995
Soil fertility and plant nutrition.
383
–
418
in
Pattee H. E.
and
Stalker H. T.
Advances in Peanut Science
Amer. Peanut Res. Educ. Scoc., Inc
Stillwater, OK
614
.
Herbert D. A. , Wilcut J. W. , and Swann C. W. 1991 Effects of various postemergence herbicide treatments and tobacco thrips (Frankliniella fusca) injury on peanut yields in Virginia. Peanut Sci 18 : 91 – 94 .
Isleib T. G. , Rice P. W. , Mozingo R. W. , Mozingo R. W. , and Pattee H. E. 1999 Registration of “Gregory” peanut. Crop Sci 39 : 1526 .
Johnson A. W. , Minton N. A. , Brenneman T. B. , Burton G. W. , Culbreath A. K. , Gascho G. J. , Baker S. H. , and Johnson W. C. 1999 Managing nematodes, fungal diseases, and thrips on peanut with pesticides and crop rotations of bahia grass, corn, and cotton. Peanut Sci 26 : 32 – 39 .
Johnson W. C. , Chamberlin J. R. , Brenneman T. B. , Todd J. W. , Mullinix B. G. , and Cardina J. 1993 Effects of paraquat and alachlor on peanut (Arachis hypogaea) growth, maturity, and yield. Weed Technol 7 : 855 – 959 .
Jordan D. L. 2007a Peanut production practices. 23 – 46 in 2007 Peanut Information, North Carolina Coop. Ext. Ser. AG-331 132 .
Jordan D. L.
2007b
Peanut weed management:.
47
–
74
in
2007 Peanut Information
North Carolina Coop. Ext. Ser. AG-331
132
.
Jordan D. L. , Spears J. F. , and Wilcut J. W. 2003 Tolerance of peanut (Arachis hypogaea) to herbicides applied postemergence. Peanut Sci 30 : 8 – 13 .
Jordan D. L. , Johnson D. P. , Spears J. F. , Penny B. , Brandenburg R. , Faircloth J. , Phipps P. , Herbert A. , Coker D. , and Chapin J. 2005 Determining peanut pod maturity and estimating the optimal digging date: using pod mesocarp color for digging Virginia market type peanut. North Carolina Coop. Ext. Ser. Pub. AG-633
Klingman G. C.
and
Ashton F. M.
1982
Formulations, drift, and calculations.
95
–
114
in
Noordhoff L. J.
Weed Science: Principles and Practices
John Wiley and Sons
New Yorl
449
.
Knauft D. A. , Colvin D. L. , and Gorbet D. W. 1990 Effect of paraquat on yield and market grade of peanut (Arachis hypogea) genotypes. Weed Technol 4 : 866 – 870 .
Koskinen W. C. and Harper S. S. 1987 Herbicide properties and processes affecting application. 9 – 18 in McWhorter C. G. and Gebhardt M. R. Methods of Applying Herbicides Weed Sci. Soc. of Am Champaign, IL 358 .
Marois J. J. and Wright D. L. 2003 Effect of tillage system, phorate, and cultivar on tomato spotted wilt of peanut. Agron. J 95 : 386 – 389 .
Mozingo R. W. , Coffelt T. A. , and Isleib T. G. 2000 Registration of ‘VA 98R’ peanut. Crop Sci 40 : 1202 – 1203 .
Mozingo R. W. , Coffelt T. A. , and Wright F. S. 1991 The influence of planting and digging dates on yield, and grade of four Virginia-type peanut cultivars. Peanut Sci 18 : 55 – 62 .
Prostko E. P. 2006 Peanut weed management issues – 2006. The University of Georgia Weed Science Homepage Available at: http://www.cropsoil.uga.edu/weedsci/index.htm.
SAS Institute 2006 GLM Procedure. 100 SAS Campus Drive Cary, NC 27513–2414.
Senseman S. A. 2007 Herbicide handbook. 9th edition Weed Science Society of America Lawrence, KS .
Shew B. B. 2007 Peanut disease management:. 94 – 118 in 2007 Peanut Information, North Carolina Coop. Ext. Ser. AG-331, 132 .
Shew B. B. , Beute M. K. , and Stalker H. T. 1995 Toward sustainable peanut production: progress in breeding for resistance to foliar and soilborne pathogens of peanut. Plant Dis 12 : 1259 – 1261 .
Sholar J. R.
,
Mozingo R. W.
, and
Beasley J. P.
1995
Peanut cultural practices.
354
–
382
in
Pattee H. E.
and
Stalker H. T.
Advances in Peanut Science
Amer. Peanut Res. Educ. Scoc., Inc
Stillwater, OK
614
.
USDA, Agricultural Marketing Service 2005 Peanut Inspection Program U.S. Gov. Print Office Washington, D.C .
Williams E. J. and Drexler J. S. 1981 A non-destructive method for determining peanut pod maturity, pericarp, mesocarp, color, morphology, and classification. Peanut Sci 8 : 134 – 141 .
Williams E. J. , Baldwin J. A. , and Beasley J. B. 2004 A turbo-blaster for pod maturity. Proc. Am. Peanut Res. and Educ. Soc 36 : 73 – 74 .
Wilcut J. W. , York A. C. , Grichar W. J. , and Wehtjie G. R. 1995 The biology and management of weeds in peanut (Arachis hypogaea). 207 – 224 in Pattee H. E. and Stalker H. T. Advances in Peanut Science. Stillwater: Am. Peanut Res. and Ed. Soc.
Wynne J. C. , Coffelt T. A. , Mozingo R. W. , and Anderson W. F. 1991 Registration of “NC-V 11” peanut. Crop Sci 31 : 484 – 485 .
Wynne J. C. , Mozingo R. W. , and Emery D. A. 1979 Registration of NC 7 peanut (Reg. No. 22). Crop Sci 19 : 563-b.