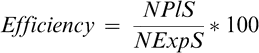Introduction
Among 80 species in the genus Arachis L., 31 belong to the Arachis section. The most economically important species of Arachis is the A. hypogaea L., cultivated peanut, which is produced for human consumption (Gálgaro et al., 1997).
Argentina is one of the major exporters of peanuts supplying an average of 245,000 tons/year (Georgalos, 1999). Ninety-eight percent of the seeded area is located in the semi-arid south-central zone of the Province of Cordoba. The cultivar Florman INTA, which was released to the national market in 1985, represents 85% of the seeded area (Gorgas et al., 1997).
Development of suitable protocols for plant regeneration is one of the main prerequisites for the genetic improvement of crop plants using biotechnological methods (Rey et al., 2000). Recently, an array of regeneration protocols has been published in peanut that describe plant development through somatic embryogenesis or shoot morphogenesis (Ozias-Akins and Gill, 2001). However, different genotypes of the same crop species often do not respond identically in tissue culture (Ozias-Akins et al., 1992). Since genotype can affect tissue culture responses, the main objective of this research was to determine the optimum concentration of auxins and cytokinins in the basal medium needed during the organogenesis process of the peanut cv. Florman INTA.
Materials and methods
Seeds of peanut Arachis hypogaea L. cv. Florman INTA were supplied by the National Institute of Agriculture in Manfredi, Cordoba, Argentina. Seeds were surface sterilized in 70% ethanol for 3 min followed by immersion in 40% sodium hypochlorite (55 g/l active chlorine) on a shaker for 20 min. They were rinsed three times with sterile deionized water. The seeds were germinated in vessels on humidified filter paper (5 seeds per vessel) for 3–5 d. The first two green or white leaves (2–5 mm in length) were aseptically dissected under a stereomicroscope. Each leaflet was transversely bisected and both halves, distal and proximal, were cultured in contact with the medium in either the abaxial or adaxial position. Each half-leaflet constituted one explant (Mroginski et al., 1981).
The nutrient medium consisted of Murashige and Skoog (1962) salts and vitamins, 3% sucrose and 0.8% agar (MS). The pH of the medium was adjusted to 5.8 with NaOH and HCl prior to adding the agar. The medium was autoclaved at 121 C for 20 min. Seed germination and plant regeneration were in a growth room at 25±2 C under a 16/8 h day/night photoperiod with an illumination of 50 µmol.m−2.s−1 of photosynthetically active radiation (PAR) provided by cool white fluorescent lamps.
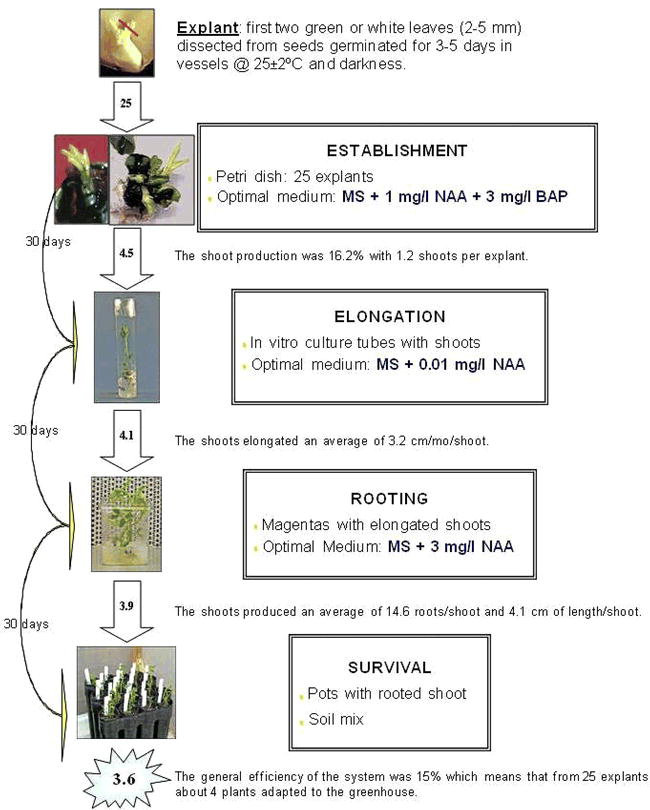
The regeneration process of peanut Florman INTA was divided into four steps as classified by Mroginski et al., 1981:
-
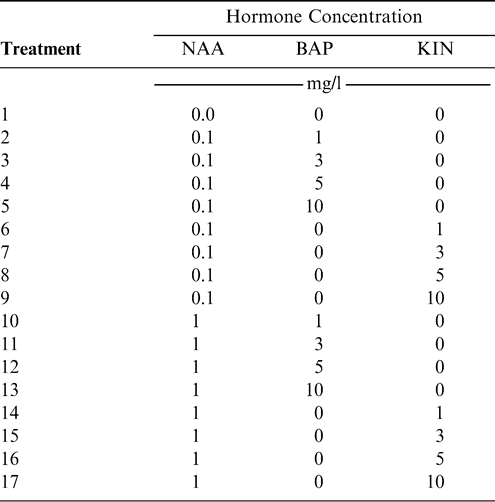
(i) Establishment: Each treatment consisted of 20 to 25 explants in a single Petri dish and three replicate plates. The medium used was MS supplemented with NAA in combination with different concentrations of BAP or KIN for a total of 17 treatments (Table 1). Plates were arranged randomly on the shelves in the growth room. Each experiment was repeated at least three times. After 4 weeks, growth response was determined in terms of type of induction obtained (bud, callus and/or root), number of buds per explant, and type of growth (multiple buds and shoots). Since each subsequent response can be affected by the previous medium, explants for the following step were randomly taken from the prior optimum medium.
-
(ii) Elongation: The shoots obtained in the establishment period were transferred to tubes containing MS supplemented with NAA (0, 0.01 or 1 mg/l) in combination with different concentrations of BAP (0, 1, 3 or 5 mg/l), for a total of 12 treatments. After 4 weeks, shoot development (elongated, not elongated) and height in centimeters were recorded.
-
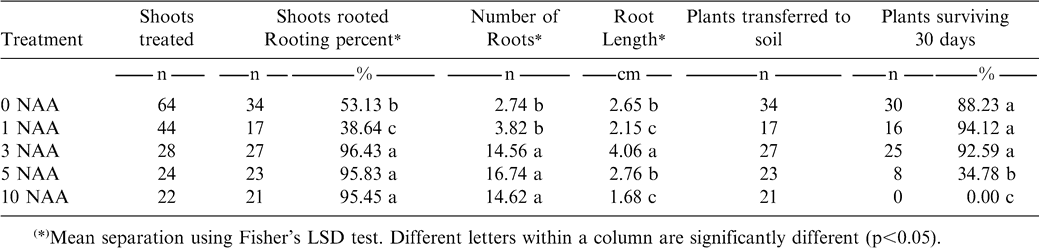
(iii) Rooting: Elongated shoots were transferred to medium composed of MS with different concentrations of NAA (0, 1, 3, 5 or 10 mg/l) for a total of 5 treatments. After 4 weeks, shoot growth, number of roots induced and lengths were recorded. The root system also was classified as normal, thickened, abnormal or abnormal stocky.
-
(iv) Survival: Rooted plantlets were rinsed gently under running tap water to remove culture medium and immediately transplanted to potting mix (Pro-Mix ‘BX’). Plants were acclimatized by gradually removing the plastic cup covering each plant. The greenhouse conditions were 28±2 C, 70% diffused natural sunlight, and 80–90% relative humidity. After 30 days, survival of the plants was assessed according to the growth of new leaves.
Data were analyzed by SAS (v.6.12, SAS Inst., 1996) using the general linear models (GLM) procedure and by the InfoStat statistical software (v.1.1, 2002). Square-root transformation of data was applied when necessary to homogenize error variances. Chi-square analyses were used for between treatment comparisons. A 5% significance level was used for the LSD Fisher test.
Overall efficiency (%) of this protocol was calculated as:
where NPlS is the number of plants that survived to soil in the greenhouse and NExpS is the number of explants cultured in the Establishment Step.
Results and Discussion
Phytohormones are important factors that can selectively induce diverse responses in plant tissue culture. Manipulation of growth regulators allows researchers to choose the best medium for each step of the induction and regeneration processes.
(i) Establishment
Explants cultured on medium with 0.1 and 1 mg/l of NAA combined with extreme cytokinin doses (1 or 10 mg/l) produced friable callus and/or roots. However, both auxin concentrations combined with 3 or 5 mg/l of BAP or KIN induced buds. High auxin dose (1 mg/l) increased bud production by 18% over the low dose. The two different cytokinins also resulted in different effects on bud induction; BAP was more efficient than KIN (Fig. 1). The treatments 0.1 mg/l NAA + 3 mg/l BAP and 1 mg/l NAA + 3 mg/l BAP resulted in the highest shoot production (19.3% and 16.2%, respectively) (Fig 1) These two treatments did not differ statistically, although the maximum number of buds per explant (1.2) was regenerated at 1 mg/l of NAA. This matches the results obtained by Mroginski et al. (1981) who reported that low NAA concentrations (0.01 and 0.1 mg/l) produce poor and erratic bud regeneration and that in high concentrations (3 and 4 mg/l) the formation of roots is stimulated, reducing the efficacy of regeneration. These results also agree with the conclusion of Sharma et al. (1990) that BAP is the most effective cytokinin in terms of number of explants producing shoots and number of shoots per explant.
(ii) Elongation
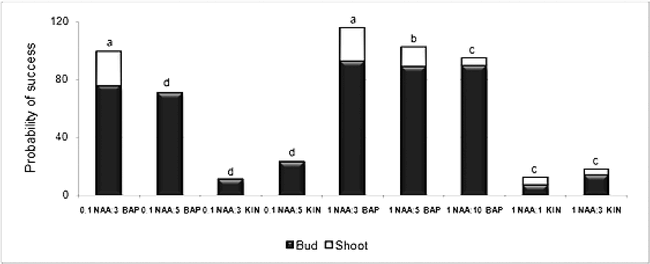
The presence of an auxin (NAA) in the elongation medium produced the expected cellular elongation effect. No significant differences were found between the two auxin concentrations assessed (0.01 and 1 mg/1), either in the number of elongated shoots or in the shoot height (Fig. 2). It was observed that a low NAA concentration was enough to stimulate the elongation of almost 91% of the shoots. This would agree with Krikorian (1995) who suggests that an auxin supplement sometimes is not necessary or it is necessary only in minimum doses due to the amounts of endogenous auxins already present in the system.
The addition of a cytokinin such as BAP to the elongation medium did not create a favorable environment for the shoots to growth in height. Treatments with BAP combined with low auxin concentration had a negative effect on elongation. The medium supplemented with 5 mg/l of BAP and 0.01 mg/l of NAA showed a 77% reduction in shoot height. In the combination of BAP with 1 mg/l of NAA, the tendency was also negative but not as marked as it was with low doses (Fig. 2). Nevertheless, shoots, instead of elongating, were stimulated to multiply whenever they were in the presence of cytokinin (data not shown). This observation was similar to the results obtained by Gagliardi et al. (2000) who showed that the use of BAP could induce the formation of multiple shoots, favoring low regeneration frequencies. Hazra et al. (2001) also observed that the presence of low BAP concentrations stimulated the axillary shoots to form multiple centers of growth, from which multiple shoots will differentiate, instead of regenerate or elongate.
(iii) Rooting and Survival
The root system developed on regenerated shoots was morphologically classified (Fig. 3). The control and medium with 1 mg/l NAA induced a “normal” root system which was characterized by long, thin (hair-like) principal adventitious roots, with abundant secondary roots and root hairs. Treatment with 3 mg/l NAA induced a root system classified as “thickened”, characterized by having slightly thickened primary roots, with a reduced number of secondary roots and root hairs, but similar in length to the normal root system. NAA at 5 mg/l induced an “abnormal” root system, with short thick primary roots, and without secondary roots and root hairs. Roots on medium with 10 mg/l NAA exhibited an “abnormal stocky” system, in which the growth of the primary roots was highly disorganized, showed presence of callus and there was a complete absence of secondary roots.
In the tests, increasing concentrations of NAA significantly increased the number of roots produced although there was no consistent behavior for the variable “length of root” which tended to decrease as the dose was increased (Table 2). This agrees with Krikorian (1995) who observed that with increasing NAA concentration, there is a tendency to produce a greater number of shorter roots, probably because auxins promote root initiation but inhibit root growth.
In order to choose an optimum rooting medium, the survival of the plants during transplanting was considered. This analysis assessed whether the root system developed in the rooting media was sufficiently normal and functional to allow the plant to survive transplanting. Karhu (1997) also considers that the production of a high number of in vitro-formed roots is not strictly associated with a high percentage of ex vitro survival.
Plants rooted without auxins or treated with auxins at 1 or 3 mg/l NAA in the rooting media showed no significant difference in survival rate, which was higher than 88% (Table 2). However, among these three treatments compared with 5 and 10 mg/l NAA there was a significant difference.
Conclusions
To regenerate peanut cv. Florman INTA via organogenesis, we suggest the protocol represented in Fig. 4 and summarized below:
-
Induce shoots from immature leaves in MS + 1 mg/l NAA + 3 mg/l BAP
-
Elongate shoots in MS + 0.01 mg/l NAA (without BAP)
-
Root shoots in medium with 3 mg/l NAA
The general efficiency of the system was 15% which means that out of 25 explants about 4 plants survived transfer to the greenhouse. These regenerated plants were normal and produced flowers and seeds.
In conclusion, the present study has aimed at demonstrating a successful regeneration protocol for the peanut variety Florman INTA with the attainment of normal and fertile plants from leaflets. Its use in future transformation systems could be limited due to the relatively low efficiency of regeneration from leaflets of this genotype compared with leaflets of New Mexico Valencia A and cotyledon organogenic or embryogenic regeneration systems that have been used for transformation of multiple genotypes (reviewed in Ozias-Akins 2007).
Literature cited
Gagliardi R. F. , Pacheco G. P. , Coculilo S. P. , Valls J. F. M. , and Mansur E. 2000 In vitro plant regeneration from seed explants of wild groundnut species (Genus Arachis, Section Extranervosae). Biodiversity and Conservation 9 : 943 – 951 .
Galgaro L. , Valls J. F. M. , and Romero Lopes C. 1997 Study of the genetic variability and similarity among and within Arachis villosulicarpa, A. pietrarellii and A. hypogaea through isoenzyme analysis. Genet. Res. Crop Evol 44 : 9 – 15 .
Georgalos A. H. 1999 Georgalos Peanut World 3 : 18 – 99 .
Gorgas J. A. , Lovera E. , Dardanelli J. , Bosnero E. A. , Grimoldi A. , and Salas H. P. 1997 Aptitud edáfica y agroclimática de Córdoba para el cultivo del maní. EEA INTA Manfredi 11 .
Hazra S. , Agrawal D. C. , Banerjee A. K. , Krishnamurthy K. V. , and Nalawade S. M. 2001 Induction of multiple shoots and plant regeneration from “accessory buds” of nodal segments from field-grown mature cotton plants (Gossypium hirsutum L.). In vitro Cell. Dev. Biol.-Plant 37 : 830 – 834 .
InfoStat version 1.1/Profesional 2002 Grupo InfoStat, Facultad de Ciencias Agropecuarias, Universidad Nacional de Córdoba, Argentina, 268 .
Karhu S. T. 1997 Rooting of blue honeysuckle microshoots. Plant Cell, Tissue and Organ Culture 48 : 153 – 159 .
Krikorian A. D. 1995 Hormones in tissue culture and micropropagation. En: Plant Hormones Davies P. J. Kluwer Academic Publishers Dordrecht, Netherlands 774 – 796 .
Mroginski L. A. , Kartha K. K. , and Shyluk J. P. 1981 Regeneration of peanut (Arachis hypogaea) plantlets by in vitro culture of immature leaves. Canadian Journal of Botany 59 5 : 826 – 830 .
Murashige T. and Skoog F. 1962 A revised medium for rapid growth and bioassays with tabacco tissue cultures. Physiol. Plant 15 : 473 – 497 .
Ozias-Akins P. 2007 Peanut.In: Biotechnology in Agriculture and Forestry 61 – Transgenic Crops VI Pua E. C. and Davey M. R. Springer Heidelberg 81 – 105 .
Ozias-Akins P. and Gill R. 2001 Progress in the development of tissue culture and transformation methods applicable to the production of transgenic peanut. Peanut Science 28 : 123 – 131 .
Ozias-Akins P. , Anderson W. F. , and Holbrook C. C. 1992 Somatic embryogenesis in Arachis hypogaea L.: genotype comparison. Plant Science 83 : 103 – 111 .
Rey H. Y. , Scocchi A. M. , Gonzalez A. M. , and Mroginski L. A. 2000 Plant Cell Reports 19 : 856 – 862 .
SAS Institute Inc 1996 User's Guide: Stadistic. Release 6.03 Edition SAS Institute Inc Cary, NC.
Sharma K. K. , Bhojwani S. S. , and Thorpe T. A. 1990 Factors affecting high frequency differentiation of shoots and roots from cotyledon explants of Brassica juncea (L.) Czern. Plant Science 66 : 247 – 253 .
Notes
1Agric. Engineer (M.Sc.) Faculty of Agricultural Sciences, Catholic University of Córdoba, Córdoba, Argentina.
2Agric. Engineers. Institute of Phytopathology and Plant Physiology (IFFIVE)-INTA, Córdoba, Argentina.
3Agric. Engineer (M.Sc.) Faculty of Agricultural Sciences, National University of Córdoba, Córdoba, Argentina.
4Professor. Department of Horticulture, University of Georgia Tifton Campus, Tifton, GA 31794.
*Corresponding author (email: pfaustinelli@gmail.com)