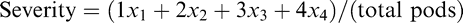Introduction
Peanut smut, caused by the fungal pathogen Thecaphora frezii Carranza & Lindquist, is an emergent disease of peanut (Arachis hypogaea L.) in South America that has significantly impacted the commercial Argentine peanut industry (Rago et al., 2017). Originally identified on wild peanut species in Brazil in 1962, peanut smut was first reported on commercial peanuts in the 1994/1995 crop year in Argentina and quickly spread to 100% of the Argentine production areas by the 2011/2012 season (Carranza and Lindquist, 1962; Marinelli et al., 1995; Rago et al., 2017). Nationwide, mean annual production losses of 3.15% have been reported, which equate to approximately US$14,151,800; yield losses of up to 35% from smut have been reported in the more heavily-infested regions of the central-northern area of Cárdoba Province. There are also global phytosanitary implications, as Argentina is currently one of the world's largest exporters of peanuts (Cazón et al., 2018; Oddino et al., 2010; Marinelli et al., 2008; Rago et al., 2017).
The management of peanut smut has been difficult, as various fungicide mixtures, seed treatments, and biocontrol measures have performed inconsistently on direct control of the pathogen (Arias et al., 2019; Cazón et al., 2018; Figueredo et al., 2017; Ganuza et al., 2017; Rago et al., 2017). Host plant resistance has shown potential as an effective tool for mitigating losses from peanut smut on commercial production, including some evidence of induced systemic resistance via inoculation by a specific strain of Bacillus spp. (Figueredo et al., 2017; Rago et al., 2017; Tonelli et al., 2011). A few commercial cultivars have historically displayed moderate resistance, including up to 52% reduced smut incidence (Farías et al., 2011; Oddino et al., 2013). One new cultivar was recently released with high levels of resistance to smut infection (A. Falco, personal communication).
For the continued development of new, smut-resistant peanut cultivars, it is necessary to identify reliable sources of resistance that can serve as useful parent material for hybridization and population development. Resistance has been identified and documented in a number of wild diploid Arachis species and Bolivian landraces (de Blas et al., 2019; Oddino et al., 2017; Soave et al., 2014). Bressano et al. (2019) and de Blas et al. (2019) demonstrated that the smut resistance trait could also be successfully introgressed into tetraploid germplasm, with relatively high transmission; Bressano et al. successfully utilized resistant A. hypogaea subsp. fastigiata landraces as parents, whereas de Blas et al. introgressed resistance from a wild, tri-species amphidiploid [(A. cardenasii × A. correntina) × A. batizocoi]4×. Little has been published, however, on the phenotypic variation of smut incidence within cultivated peanut. Identifying resistance in cultivated, tetraploid peanut genotypes could help provide more readily-useable sources of resistance for commercial cultivar development, without the necessary pre-breeding or chromosome doubling to prepare/convert diploid germplasm for hybridization with elite tetraploid germplasm. Therefore, the purpose of this study was to evaluate the incidence and severity of peanut smut on the core of the core (or "mini-core") of the U.S. peanut germplasm collection (Holbrook and Dong, 2005) in situ in Argentina.
Materials and Methods
This study was modeled after similar germplasm screenings published in cotton (Gossypium hirsutum L.) by Basal et al. (2003) and in the U.S. peanut mini-core by Dean et al. (2009); it was also arranged in the field similarly to the peanut mini-core evaluation reported by Upadhyaya (2005). Field trials were conducted with the Maniagro S.A. peanut company, at the La Riojana research farm near General Cabrera, in the Cárdoba Province of Argentina, in the 2016-2017, 2017-2018, and 2018-2019 crop years. All plots were planted on 31 Oct. 2016, 27 Oct. 2017, and 24 Oct. 2018, by hand-dribbling seed through a 2-row Monosem precision air planter (Monosem, Inc., Edwardsville, KS) at an approximate seeding rate of 3.2 seed/m and depth of 6 cm. Fresh seed for each planted mini-core accession was obtained from the USDA National Plant Germplasm System (NPGS) peanut collection in Griffin, GA for planting in 2016, therefore quantities were limited to only 25 seed per plot in the 2016-2017 season. As a result, individual plot dimensions in 2016-2017 were 1.8 m (2 rows) x 2 m. Seed harvested from each genotype in 2016-2017 was kept and replanted for evaluation in 2017-2018; therefore, plot dimensions were larger in 2017-2018 and 2018-2019, measuring 1.8 m x 6 m. Row spacing was 91 cm each year. All plots were planted such that planting began and ended in the alleys between plots, then alleys were hand-cleared of additional peanut seedlings after emergence. Additionally, all plots were planted in minimal tillage (disc-harrowed soil only) into wheat (Triticum aestivum L.) stubble every year, in a dryland production scenario with consistently heavy natural peanut smut pressure each year. Overall management throughout the growing season was conducted according to local guidelines for commercial production.
At the beginning of this study, only the 104 U.S. peanut mini-core accessions registered by Chen et al. (2014) were available for public distribution in the NPGS collection. Thus, these 104 accessions were obtained and planted in 2016-2017, along with five additional accessions from the NPGS germplasm collection. Three advanced, uniform breeding lines (14-1-0066, 16-1-0033, and 16-1-0089), selected from a breeding program operated by International Peanut Group and Maniagro S.A., were used as susceptible controls. Four local Argentine cultivars were also included as commercial controls (though not resistant), including cultivars Granoleico (INASE Reg. No. 7907), MA-88 (INASE Reg. No. 17235), MA-757 (INASE Reg. No. 17240), and MA-767 (INASE Reg. No. 17234). Five of the planted mini-core accessions in 2016-2017 did not germinate and were therefore excluded from the final overall analyses. However, new seed for four of those accessions was reobtained from the NPGS collection and planted in 2017-2018. One accession (PI 429420) was planted only in 2018-2019, due to limited seed availability and poor emergence the previous years. All plots were arranged randomly each year for three subsequent years. Therefore, experimental design was a randomized complete block, blocked by year (three total blocks), with genotype as treatment.
Plots were naturally infested with peanut smut each year. The location of the field trials was in an area with historically-elevated levels of smut incidence, near two large commercial peanut shellers in General Cabrera and nearby Carnerillo. Thus, ambient soil teliospore density at the field location was approximately 4500 teliospores/g soil during the course of these evaluations, which would be classified as "highly-infested" (Oddino et al., 2010). All plots were dug on 10 April 2017, 13 April 2018, and 8 April 2019, which were 161 days after planting (DAP), 168 DAP, and 166 DAP, respectively. A 100-pod sample of each plot was randomly collected immediately after digging for determination of smut incidence and severity. All pods from each sample were individually hand-opened and visually rated for smut infection; affected pods were rated using a 0-4 visual scale developed by Astiz Gassá et al. (2008) and photographically documented by Cazán et al. (2018), de Blas et al. (2019), and Rago et al. (2017). Any pod with a scored value equal to or greater than "1" was considered affected. Smut incidence was subsequently expressed a percentage of the whole sample. Mean severity values of affected pods for each genotype were calculated using the following equation:
where xn is the number of pods with n severity grade and "total pods" is the number of total affected pods identified in a sample.
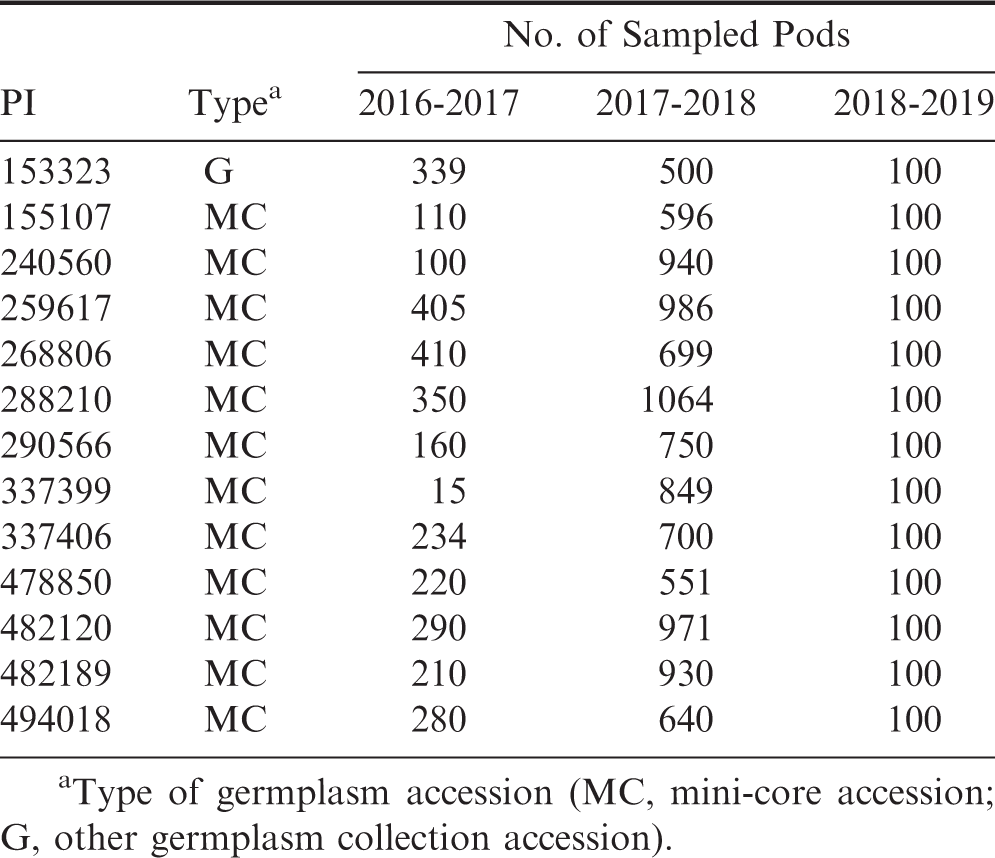
In 2016-2017, given the small plot sizes, some plots did not produce greater than 100 total pods; in these cases, all available pods were opened and rated for smut incidence and resultant incidence expressed as a percentage of the whole. There was sufficient production from plots in 2017-2018 and 2018-2019 to provide at least 100 pods per plot for analysis, including for all of the missing/non-emergent genotypes in 2016-2017 (except for PI 429420). There was sufficient production of PI 429420 in 2018-2019 to evaluate at least 100 pods. In 2016-2017 and 2017-2018, in plots with 0.0% smut incidence from the 100-pod samples, all of the remaining pods from each respective plot were opened and analyzed, to determine disease incidence in a larger sample size.
Statistical analyses were conducted with R software (R Core Team, 2016), using the Agricolae package. All data were analyzed by analysis of variance and means were separated via Tukey's HSD Test (P ≤ 0.05) for the main genotype comparison and Fisher's Least Significant Difference Test (P ≤ 0.05) for the comparison of the smaller group of genotypes in 2017-2018 and 2018-2019. Pearson and Spearman correlation analyses between smut incidence and severity values were also conducted in R, using the "cor.test" function.
Results and Discussion
Significant genetic variation for both smut incidence and severity was found among the evaluated genotypes, although genotype and block were both significant in these evaluations (P ≤ 0.05; data not shown). As mentioned above, this experiment was blocked by year, so the significance in block was likely due to the natural year-to-year environmental variation inherent in utilizing naturally-infested soils. However, blocking the experiment in time minimized confounding year-to-year effects on genotype. Additionally, a significant genotype effect across varying levels of natural infestation indicates stable phenotypic expression of both resistance and susceptibility.
Smut Incidence and Severity
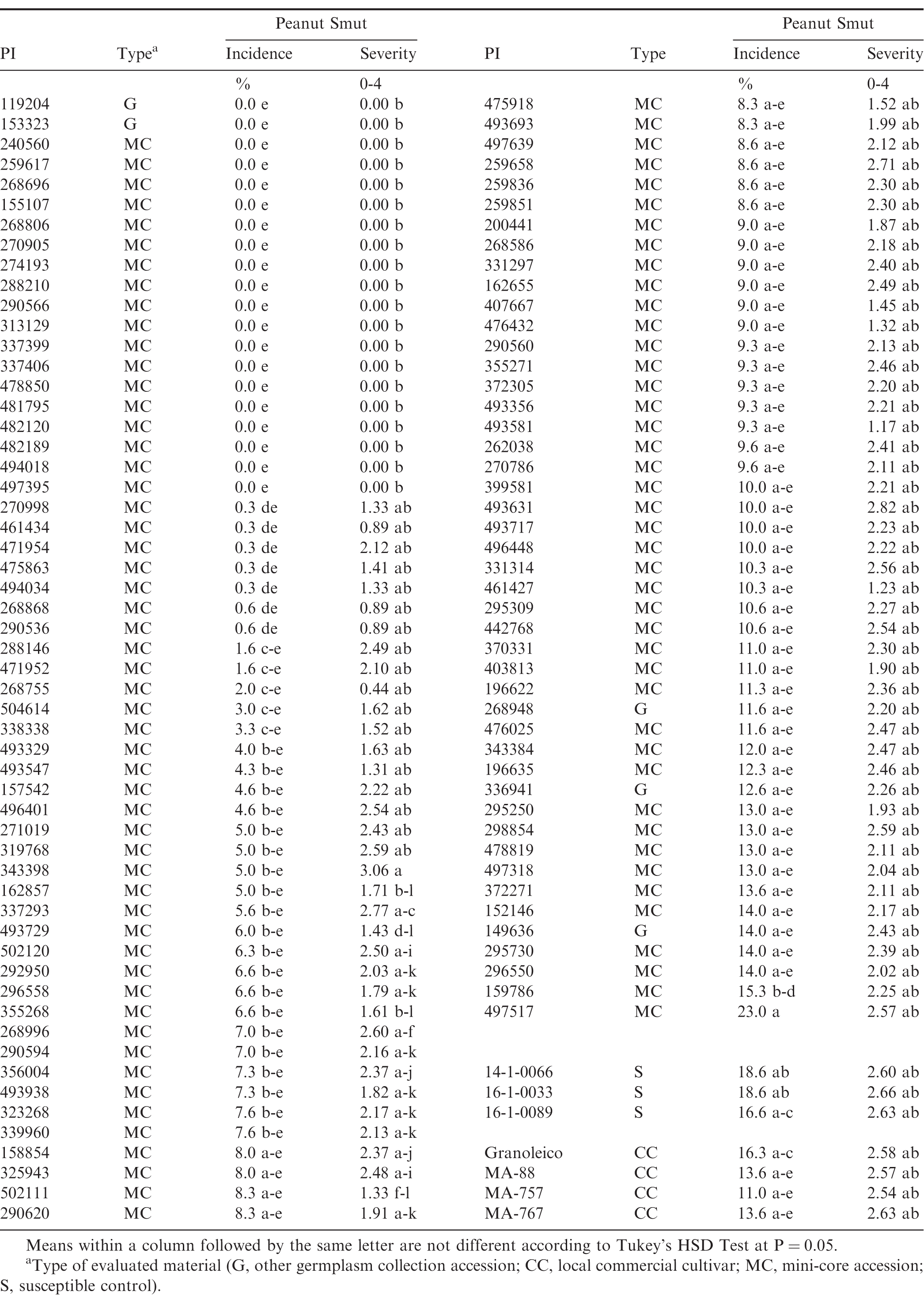
Mean smut incidence ranged 0.0 - 23.0% in the evaluated mini-core and germplasm collection accessions, whereas the susceptible controls ranged 16.6 - 18.6% and the local evaluated cultivars ranged 11.0 - 16.3% (Table 1). Approximately 20 germplasm accessions exhibited no observable incidence of smut over all three years; of those 20, two accessions (PI 119204 and PI 153323) were germplasm accessions that are not designated as part of the official mini-core collection. Additionally, all of those accessions had less smut incidence than the three susceptible controls and four commercial cultivars (P ≤ 0.05). One accession (PI 497517) exhibited the greatest level of smut incidence (23.0%) which made it greater than all other evaluated mini-core and germplasm accessions (P ≤ 0.05). Thirty-three germplasm accessions had lesser smut incidence than the susceptible controls and the evaluated cultivars, the least of which (0.0%) constituting more than a 10-fold reduction in comparative incidence than the commercial controls.
Mean severity of smut infection ranged 0.00 - 3.06 among the mini-core and germplasm collection accessions (Table 1); susceptible controls were 2.60 - 2.66 and evaluated cultivars ranged 2.54 - 2.63, but were not different (P > 0.05). The 20 mini-core and germplasm accessions with no smut incidence also had smut severity ratings of "0.00"; therefore, all of those accessions had lesser severity than the susceptible controls and the four commercial cultivars (P ≤ 0.05). Correlation analyses between smut incidence and severity were conducted only on the affected pods rated during the course of these evaluations. Severity was negatively correlated with percent incidence (P ≤ 0.05), with a Pearson correlation coefficient of - 0.148; the Spearman correlation coefficient was - 0.214. These correlation results suggest that smut severity decreases as overall incidence increases, indicating that there is varied progression of the disease throughout infected pods on a given plant. Since smut infection does not occur until peanut pegs reach the soil, this differential incidence/severity rate is likely linked to the variable pegging timing and subsequent maturity of different pods (Marraro Acuña et al., 2013). However, given the relatively low correlation coefficients for both analyses it is also likely that different genotypes have differential rates of smut severity, despite large or small levels of infestation-this would indicate that some genotypes could have a substantial incidence of smut, but at a more commercially-tolerable level.
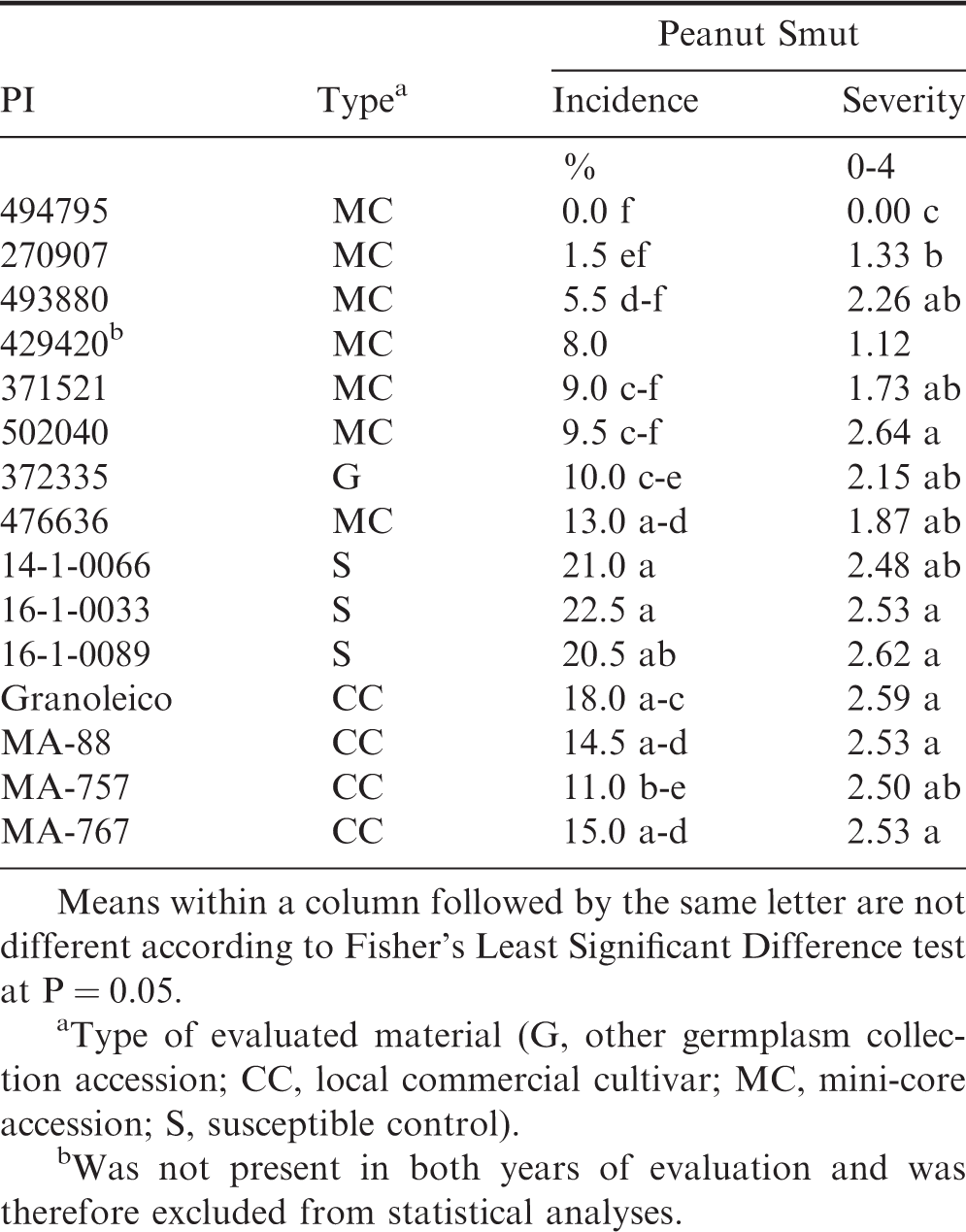
The results of the mini-core accessions that had no emergence in the 2016-2017 evaluation are presented in Table 2. One accession (PI 429420) again had no emergence in 2017-2018, so it was excluded from the statistical analyses. Genotype was significant (P ≤ 0.05) for both smut incidence and severity; block was not significant for either parameter (P > 0.05; data not shown). Smut incidence ranged 0.0 - 13.0% among the additional mini-core and germplasm accessions; the susceptible controls ranged 20.5 - 22.5% and the commercial cultivars were 11.0 - 18.0%. Of the evaluated germplasm accessions, PI 494795 was the only one to exhibit no smut incidence both years. Smut severity ranged 0.00 - 2.64; PI 494795 had a lower severity (0.00) than all of the evaluated genotypes in this group (P ≤ 0.05).
Additional evaluation (100-pod samples) of peanut smut incidence and severity on seven peanut mini-core accessions, one germplasm collection accession (which were either missing or did not emerge in the 2016-2017 evaluation), three susceptible breeding lines, and four local commercial cultivars near General Cabrera, Argentina in 2017-2019.
Resistant Genotypes and Geographic Origins
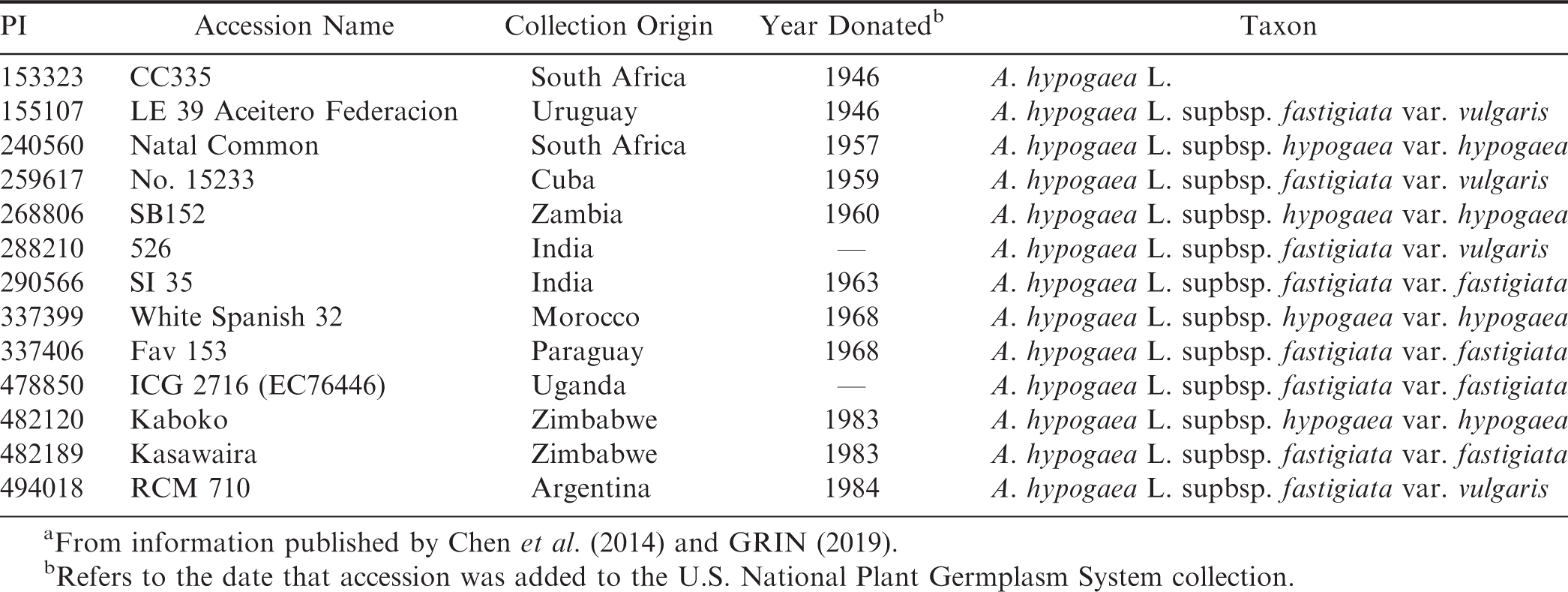
Genotypes found to have 0.0% smut incidence from both sampling methods are presented in Table 3. Again, all of the pods from the harvested plots in 2016-2017 and 2017-2018 for these genotypes were opened and found to have no smut incidence. In total, there were 13 genotypes that had 0.0% incidence in all evaluated pods: 12 mini-core accessions and a germplasm accession (PI 372335). The total number of evaluated pods per genotype ranged 15 - 1064. It is likely that these genotypes are resistant to infection by T. frezii and would be potential sources of resistance in A. hypogaea to use for introgression into breeding populations and eventually commercial cultivars.
It was hypothesized by the authors that these 13 accessions could have originated in regions of South America with significant ambient T. frezii populations, thereby suggesting possible co-development of resistant germplasm with the increased presence of the pathogen. Table 4 presents the name, geographic collection origin and date, and taxon information for the 13 resistant accessions, adapted from information published by Chen et al. (2014) and data currently available in the NPGS Genetic Resources Information Network (GRIN, 2019) for the material. The accessions were collected from a wide array of geographic origins spanning the Americas, Africa, and Asia. Only three accessions (PI 155107, PI 337406, and PI 494018) were collected from locations in South America (Uruguay, Paraguay, and Argentina, respectively). Therefore, this information does not directly suggest that these resistant accessions developed in concert with the concurrent development and proliferation of the T. frezii pathogen on peanut. However, it does suggest that mechanisms of resistance may be well-conserved across the various collected accessions within A. hypogaea. Development of resistance may have occurred in a common progenitor of these lines prior to the distribution of A. hypogaea to other continents. Alternatively, these resistant accessions may also be carrying different mechanisms of resistance. Therefore, genomic analysis and further phenotypic analysis would be necessary to elucidate the phylogenetic relationships among these resistant accessions and identify the subsequent mechanisms responsible for conferring resistance. Identifying different mechanisms of resistance to peanut smut would allow plant breeders to introgress multiple resistance loci into cultivars, thereby reducing the likelihood of T. frezii overcoming host plant resistance in the field.
Summary and Conclusions
The data presented herein suggest that the incidence and severity of peanut smut varies significantly within A. hypogaea and that there are potential sources of resistance within the U.S. peanut mini-core collection. While this was a screening of a limited subset of germplasm accessions, identifying resistant accessions within the mini-core will inform more targeted screening of related A. hypogaea groups in the larger germplasm collections worldwide. This information will be valuable for plant breeders wanting to develop smut-resistant cultivars. The identification of both resistant and susceptible germplasm will also be useful for elucidating the genetic nature of smut resistance in peanut, as well as aiding in genomic marker development.
Over three years, 18 mini-core accessions and two germplasm collection accessions (PI 153323 and PI 119204) exhibited no smut incidence in a 100-pod sample, with respective severity values of "0.00". One additional mini-core accession (PI 494795) also exhibited 0.0% smut incidence in 2017-2018 and 2018-2019. There were 12 mini-core accessions and one germplasm collection accession that had no smut incidence when all harvested pods were opened and inspected in 2016-2017 and 2017-2018. These 13 accessions would likely be good sources of resistance for research and cultivar development. Identifying such resistance in A. hypogaea provides tetraploid sources for introgressing smut resistance into new, elite peanut cultivars or breeding populations, without requiring pre-breeding of resistant diploid germplasm. Additionally, using A. hypogaea sources of resistance can help reduce potential linkage drag of undesirable alleles from resistant wild Arachis species, as evidenced in other crops (Wann et al., 2017). However, depending on the source, using a tetraploid source of resistance may make it more complicated to pyramid resistance from both subgenomes in peanut, as described by de Blas et al. (2019).
The 13 resistant accessions identified in these evaluations represent both the fastigiata and hypogaea subspecies of A. hypogaea. Only three of these were collected in South America, which does not suggest potential co-development of resistant germplasm commensurate with the historic proliferation of T. frezii on peanut in South America. However, this does suggest that mechanisms of resistance to T. frezii incidence may be well-conserved across A. hypogaea germplasm-this could aid in identifying the specific mechanism(s) of smut resistance in many different groups of cultivated peanut, which would thereby increase the efficiency of selection for plant breeders in the development of resistant cultivars. Nevertheless, further research is needed to identify these specific mechanisms of resistance and to identify other groups of A. hypogaea with resistance. Additionally, more information is needed on the genetic nature of smut resistance in peanut, to better utilize the trait and maximize its stability in elite germplasm.
Acknowledgements
The authors wish to express thanks to Sergio Armando for outstanding technical assistance, which was essential to the success of this research.
Literature Cited
Arias, R.S., L.I Cazon, A.N Massa, B.E Scheffler, V.S Sobolev, M.C Lamb, M.V Duke, S.A Simpson, C Conforto, J.A Paredes, J.H Soave, M.I Buteler, and A.M Rago 2019 Mitogenome and nuclear-encoded fungicide-target genes of Thecaphora frezii causal agent of peanut smut Fung. Genomics and Bio (9)1:160
Astiz Gassó, M., R Leis, and A.D Marinelli 2008 Evaluation of incidence and severity of peanut smut (Thecaphora frezii) in artificial infections, on commercial peanut cultivars (In Spanish.) In Proceedings of the 1° Congreso Argentino de Fitopatología, Córdoba, Argentina. 28-30 May 2008. Asoc. Argentina Fitopatol., Córdoba. pp 118.
Basal, H., P Bebeli, C.W Smith, and P Thaxton 2003 Root growth parameters of converted race stocks of Upland cotton and two BC2F2 populations Crop Sci 43: 1983- 1988.
de Blas, F.J., M Bressano, I Teich, M.G Balzarini, R.S Arias, M.M Manifesto, B.P Costero, C Oddino, S.J Soave, J.A Soave, M.I Buteler, A.N Massa, and J.G Seijo 2019 Identification of smut resistance in wild Arachis species and its introgression into peanut elite lines Crop Sci 59: 1657- 1665.
M., Bressano, A.N Massa, R.S Arias, F de Blas, C Oddino, P.C Faustinelli, S Soave, J.H Soave, M.A Pérez, V.S Sobolev, M.C Lamb, M Balzarini, M.I Buteler, and J.G Seijo (2019). Introgression of peanut smut resistance from landraces to elite peanut cultivars (Arachis hypogaea L.). PLos ONE 14 ((2)): e0211920.
Carranza, J and J Lindquist 1962 Thecaphora frezii, parasitic of the Arachis sp. (In Spanish.) Bol. Soc. Argent. Bot. 10: 11- 18.
Cazán, L.I., J.A Paredes, and A.M Rago 2018 The biology of Thecaphora frezii smut and its effects on Argentine peanut production In J.N Kimatu (ed.) Advances in plant pathology, IntechOpen Ltd., London, U.K pp. 31- 46 doi:10.5772/intechopen.75837.
Chen, C.Y., N.A Barkley, M.L Wang, C.C Holbrook, and P.M Dang 2014 Registration of purified accessions for the U.S. peanut mini-core germplasm collection J. of Plant Reg 8: 77- 85.
Dean, L.L., K.W Hendrix, C.C Holbrook, and T.H Sanders 2009 Content of some nutrients in the core of the core of the peanut germplasm collection Peanut Sci 36: 104- 120.
Farías, A., J Baldessari, A.F Marraro, and P.H Mazzini 2011 Evaluation of high-oleic peanut cultivars in the presence of peanut smut (Thecaphora frezii) (In Spanish.) In 26th° Jornada Nacional del Maní, General Cabrera, Cárdoba, Argentina pp. 20- 22.
Figueredo, M.S., M.L Tonelli, F Ibáñez, F Morla, G Cerioni, M.C Tordable, and A Fabra 2017 Induced systemic resistance and symbiotic performance of peanut plants challenged with fungal pathogens and co-inoculated with the biocontrol agent Bacillus sp. CHEP5 and Bradyrhizobium sp. SEMIA6144 Microbiol. Res 197 (2017): 65- 73.
Ganuza, M., N Pastor, J Erazo, J Andrés, M.M Reynoso, M Rovera, and A.M Torres 2017 Efficacy of the biocontrol agent Trichoderma harzianum ITEM 3636 against peanut smut, an emergent disease caused by Thecaphora frezii. Europ J. Plant Path 151: 257- 262.
[GRIN] Genetic Resources Information Network 2019 U.S. Dept. of Agric. National Plant Germplasm System. Available at https://npgsweb.ars-grin.gov/gringlobal/ crop.aspx?id=86 (verified 21 Jan. 2020).
Holbrook, C.C and W Dong 2005 Development and evaluation of a mini core collection for the U.S. peanut germplasm collection Crop Sci 45: 1540- 1544.
Marinelli, A.D., G March, and A.M Rago 1995 Peanut smut (Thecaphora frezii) on Arachis hypogaea L (In Spanish.) In Proceedings of the VII Congreso de Micología y XVII Jornadas Argentinas de Micología, Rosario, Santa Fé, Argentina. 14-16 June 1995. Asoc. Argentina Micol., Buenos Aires. pp. 134.
Marinelli, A., G.J March, and C Oddino 2008 Biological and epidemiological aspects of peanut smut (Arachis hypogaea L.) caused by Thecaphora frezii Carranza & Lindquist AgriScientia 25 (1): 1- 5.
Marraro Acuña, F., M Cosa, and A Wiemer 2013 Peanut smut: Histology, incidence, and severity (In Spanish.) In 28° Jornada Nacional del Maní, General Cabrera, Cárdoba, Argentina pp. 26- 27.
Oddino, C., A Marinelli, G March, J García, L Tarditi, L.Y D'Eramo, and S Ferrari 2010 Relationship between inoculum potential of Thecaphora frezii, peanut smut intensity, and cultivar yield (In Spanish.) In 25° Jornada Nacional del Maní, General Cabrera, Cárdoba, Argentina pp. 24- 26.
Oddino, C., J Soave, S Soave, A Moresi, C Bianco, M Buteler, P Faustinelli, and D Torre 2013 Genetic advances in the tolerance of peanut to smut caused by Thecaphora frezii (In Spanish.) In 28° Jornada Nacional del Maní, General Cabrera, Cárdoba, Argentina pp. 28- 30.
Oddino, C.M., J.A Soave, S.J Soave, M.I Buteler, A Moresi, F.J de Blas, et al 2017 Sources of smut resistance in peanut wild species and Bolivian landraces In Advances in Arachis through genomics and biotechnology. Cordába, Argentina. 14-17 March 2017. pp. 8.
R Core Team 2016 R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria Available at: http://www.R-project.org (verified 16 Jan. 2020).
Rago, A.M., L.I Cazán, J.A Paredes, J.P.E Molina, E.C Conforto, E.M Bisonard, and C Oddino 2017 Peanut smut: from an emerging disease to an actual threat to Argentine peanut production Plant Disease 101: 400- 408.
Soave, J.A., C Bianco, R Burgoa, R Montaño, A.M Rago, L.I Cazán, et al 2014 First detection of peanut smut (Thecaphora frezii) in Bolivia. (In Spanish.) In L.D Ploper (ed.) Proceedings of the 3° Congreso Argentino de Fitopatología, San Miguel de Tucumán, Argentina. 4-6 June 2014. Asoc. Argentina Fitopatol., Cárdoba, Argentina. pp. 211.
Tonelli, M.L., A Furlán, T Taurian, S Castro and A Fabra 2011 Peanut priming induced by biocontrol agents Physiol. Mol. Plant Pathol., 75 (2011): 100- 105.
Upadhyaya, H.D 2005 Variability for drought resistance related traits in the mini core collection of peanut Crop Sci 45: 1432- 1440.
Wann, D.Q., J.K Dever, M.D Arnold, and H.D Elkins 2017 Genetic analysis and gain from selection of thrips resistance in cotton Euphytica 213, 70(2017) doi:10.1007/s10681-017-1861-0.