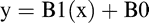Introduction
Georgia produced 51% of all U.S. peanut (Arachis hypogaea L.) in 2019 (Neal 2019). Georgia peanut growers achieve high yields by utilizing seeds that have been tested for quality and vigor. Excellent seed quality is crucial in establishing peanut stands of 20 plants/m of row (Monfort 2019), and healthy plants early in the season. Healthy emerging plant characteristics include undamaged cotyledons on a thick hypocotyl narrowing above the root, an epicotyl with more than one primary leaf, and radicle that has adventitious roots (Peterson et al. 2018). To be classified as certified peanut seed in Georgia, Tifton Seed Laboratory testing must indicate at least 70% germination (Black 2018).
Peanut seed storage techniques are an important factor in seed deterioration. The relative humidity (RH) and temperature of the storage environment are important due to influencing seed moisture and rate of seed cellular metabolism (McDonald 2004). Optimal peanut seed storage environment is 10 C at 65% RH, once peanuts have been dried to 6% moisture content (Ketring 1992). Under controlled storage conditions, Navarro et al. (1989) concluded that as humidity increased, peanut seed germination decreased. The researchers placed in-shell peanut at 15 C at 79 to 83% RH, with subsequent testing resulting in greater than 80% germination 150 d after storage initiation. When RH was increased from 85 to 89%, peanut seed germination decreased to 30% at 80 d of storage. The investigators then increased temperature to 26 C and used the same RH, this resulted in germination less than 50% at 90 d of storage for both humidity levels. Morton et al. (2008) indicated that germination rate and in-field stand establishment may be acceptable to growers, but poor seedling vigor may result in suboptimal plant performance.
To achieve high yield in peanut, proper weed control must be maintained. Weed species compete with peanut for light, nutrients, space, and water (Grichar and Dotray 2011). The critical period of weed control for maximum peanut yield will vary by broadleaf and grass species (Everman et al. 2008 a; 2008 b). Critical period is defined as the maximum time the crop and weed can compete in the early season without hindering crop performance and minimum weed-free time that will prevent yield reductions (Everman 2008b). The critical period of weed control for peanut can vary from 3 to 8 wk after planting (Everman et al. 2008 B; Webster et al. 2007). Growers maintain this period weed free through numerous techniques of integrated weed management practices. These include chemical (herbicides), physical, cultural, and biological methods (Harker and O'Donovan 2013), though herbicides are the dominant tool. Proper herbicide mode of action rotation is essential to prevent weed resistance to herbicides.
Georgia peanut growers use diclosulam and flumioxazin for weed control in peanut (Monfort et al. 2019). In 2018 according to the USDA chemical use database, 45, 14, and 25% of Alabama, Florida, and Georgia peanut hectares, respectively, were treated with diclosulam (NASS 2019). Diclosulam is an acetolactate synthase inhibitor (ALS) WSSA Group 2 herbicide belonging to the triazolopyrimidines family (WSSA). The ALS group of herbicides prevent weed growth by inhibiting the production of valine, leucine, and isoleucine amino acids. The ALS herbicides have at least 162 resistant weed biotypes across the globe (Heap 2019). The primary mode of resistance found in ALS resistant biotypes is target site mutation in which the herbicide binding position is altered preventing proper herbicide binding (Heap 2013). Wise et al. (2009) noted ALS-resistant Palmer amaranth (Amaranthus palmeri) populations in Georgia. When herbicide resistant weed populations are established, growers must switch to alternative mechanism of action for successful weed control. Flumioxazin is a protoporphyrinogen IX oxidase inhibitor, WSSA Group 14 herbicide that disrupts proper chloroplast formation by preventing protoporphyrinogen IX from oxidizing into protoporphyrin IX and produces reactive oxygen species (ROS) (Green and Owen 2011; Shaner 2014). The ROS will cause lipid peroxidation, damaging cell membrane and cell walls, resulting in cellular leaking and eventual plant death.
Since registration in 2001, peanut seedling injury from flumioxazin has been noted in field research. Johnson III et al. (2006) reported flumioxazin visual injury and stunting on peanut when applied 8 to 10 days after planting (DAP) at 71 g/ha at midseason. The researchers also noted that peanut treated with 107 g/ha of flumioxazin at 4 DAP were injured up to 49% and stunted at midseason. Similar conclusions were noted by Main et al. (2003) in which the researchers applied 0, 71, 105, or 211 g/ha of flumioxazin and observed stunting and visual injury. They reported that all treatments caused up to 25% stunting 28 DAP, while the 211 g/ha rate caused stunting up to 56 DAP compared to the non-treated control (NTC). In laboratory research, Williams et al. (2015) indicated that peanut radicle length decreased as flumioxazin rate increased with decreasing temperature. There were no differences noted in relation to germination and rate of flumioxazin. Peanut seedling mass was also negatively affected by both flumioxazin rate and temperature. The 2018 USDA chemical use database indicated that 62, 62, and 74% of Alabama, Florida, and Georgia peanut hectares, respectively, were treated with flumioxazin (NASS 2019), emphasizing the importance of this herbicide.
With improved tomato spotted wilt resistant cultivars, growers are now planting peanut in April (Grey et al. 2017). April peanut planting allows growers to spread out their risk associated with biotic and environmental concerns. With the acceptance of new cultivars and planting in April when soil temperatures may be highly variable, the need to ensure consistent stand establishment is always a concern. Peanut injury from flumioxazin PRE applied has not been evaluated at the initiation of seed germination and radicle development stage.
To identify the effects of flumioxazin on peanut seed germination and radicle development, laboratory experiments were conducted to test seed of the same cultivar with varying germination across a range of temperatures using a thermogradient table. Peanut seed germination and radicle development were also measured across varying rates of flumioxazin.
Materials and Methods
Experiments were conducted in 2018 using two certified seed lots of GA-16HO (Branch 2017) with initial germination of 75 and 90%, as certified by the Tifton Seed Laboratory. All peanut seed samples had been stored in shell from the September 2017 harvest until the following March 2018 in a warehouse at a constant temperature and relative humidity. After processing at a commercial facility, seed were tested by an independent laboratory to confirm viability. Multiple 22.6 kg bags were obtained from the warehouse and sub-samples taken for testing.
Peanut seed germination response to flumioxazin and temperature were then evaluated on a thermogradient table. The thermogradient table was created using solid aluminum blocks measuring 2.4 m long by 0.9 m wide and 7.6 cm in height. Each side has a 1.0 cm drilled hole allowing fluid to flow across each side of the table from either a cooling or warming unit (Anova Model A40, Anova Industries Inc., Stafford, TX) through a hose. The fluids consisted of a 1:10 ethylene glycol to water mixture flowing at a rate of 3.8 L per minute that were independent of each other. The units were set to achieve temperatures ranging between 20 C and 30 C across the entire table. Thermocouples consisted of duplex insulated PR-T-24 wire (Omega Engineering, Stamford, CT) and were placed under the table in holes measured 8 mm by 7 cm deep, allowing the couples to be within 5 mm of the table surface in 10 cm intervals. Temperature at each thermocouple was recorded every 30 min. using a Graphtec data logger (MicroDAQ.com Ltd., Contoocook, NH). The temperature was regularly checked to insure proper temperature range was achieved.
Multiple solutions of flumioxazin were prepared in 1000 mL Erlenmeyer flasks. Initially a 10,000 ppb solution was prepared by dissolving 19.7 mg of commercial flumioxazin (Valor SX, Valent Corp., Walnut Creek, CA) in 1L distilled water. A serial dilution was performed to achieve solution concentrations of 0.01, 0.10, 1.0, and 10.0 ppb, and included a nontreated control. These concentrations were selected based upon previous research, in which the researchers noted a linear decrease in radicle length as flumioxazin concentration increased from 0.01 to 10 ppm (Williams et al. 2015). Ten whole peanut seed were evenly distributed on germination paper (SDB 86 mm, Anchor Paper Co., St. Paul, MN) and placed in a 100 by 15 mm sterile Petri-dish (Fisher Scientific Education, Hanover Park, IL). Petri dishes with seed were placed on the thermogradient table within a row of cells with temperatures of 20, 23, 26, or 29 C.
The flumioxazin solutions were applied to each Petri dish on the table using a syringe (Fisher Scientific Education, Hanover Park, IL) beginning with the lowest (0.0 ppb) to the highest concentration (10.0 ppb). For each Petri dish with 10 peanut seed, 10 mL of solution was added initially. For each germination rate, there were four replications per concentration per temperature and replicated three times per temperature for all concentrations. During the course of the study, the Petri dish would be replenished with the respective concentration as needed to maintain constant moisture levels. Seeds of the same germination rate were tested at the same time as to not mix high germination with low germination. When applying the initial treatments, a green light was used to prevent herbicide activation. Upon trial initiation, all seed were kept in complete darkness for 5 days after initial application application (DAA). Seed were then exposed to 8 hours of light via cool, white fluorescent lamps at 10 μmol/m2/s1 for an additional 5 days, for a total of 10 days in solution. Seeds were then removed from the thermal gradient table, germination counts taken, and radicle lengths measured. Seeds were considered germinated when the radicle length was greater than 5 mm in length (Grey et al. 2011). Fresh weight was obtained by weighing all seeds per dish as well as only germinated seeds.
The experimental design was a randomized complete block with a split-plot, with flumioxazin concentration as the whole plot (blocking factor), and seedling germination and temperature as the sub plots. Flumioxazin concentration, seed germination and temperature were considered as fixed effects, whereas replication and replication x flumioxazin concentration were considered as random effects. Data was also analyzed using ANOVA in SAS 9.4 (SAS Institute Inc., Cary NC). Linear regression analysis was utilized to evaluate the parameter estimates. The intent was to determine if the response could be described by using linear regression
where y is peanut radicle length in mm, B0 is the y-intercept, B1 is the slope, and x is temperature. Where there were no interactions, data for peanut radicle length in mm was analyzed to establish the effects of concentration across temperatures. Conversely, data was further analyzed to determine temperature effects across each concentration. Data for the linear regression equations were subjected to ANOVA using the REG procedure in SAS 9.4 (SAS Institute Inc., Cary NC) with mean separation of parameter estimates. The different seed lots (75% and 90%) were not significant, therefore, data was combined for analysis with respect to seed lot.
Results and Discussion
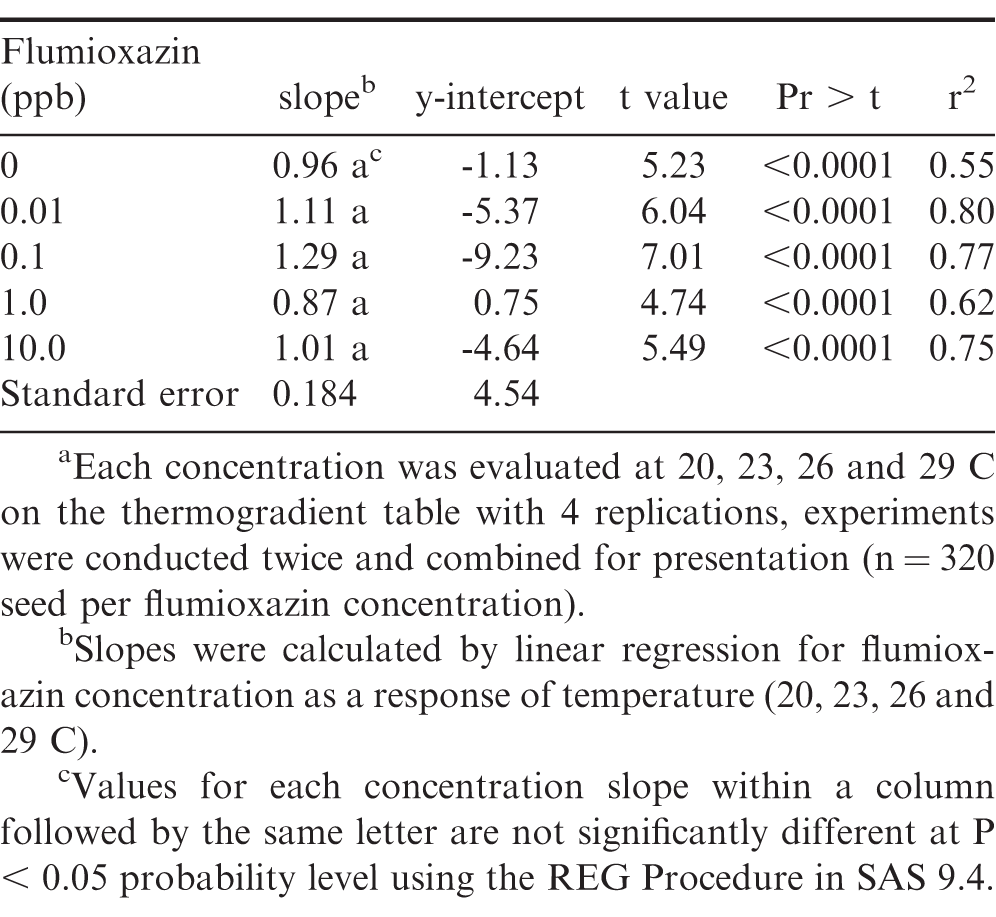
Peanut radicle development response to temperature was not affected by flumioxazin (Table 1). Comparing the slopes of each concentration regression line, there were no differences noted, with r2 values ranging from 0.55 to 0.80 (Table 1). The field rate of flumioxazin is 107 g ai/ha, in terms of soil concentration based on a hectare furrow slice (top 6 inches of soil), which corresponds to 46 ppb. Previous research indicated that the concentration applied to the field was too high for proper radicle development, when in direct contact with peanut seed (Williams et al. 2015). Due to flumioxazin being applied on the soil surface and not incorporated, the actual flumioxazin concentration contacting the seed is lower than 46 ppb as it moves through the soil profile. These results indicate that the levels of flumioxazin in the soil profile at the concentrations tested in this study, will not affect germination or radicle development up to 10 days after treatment (DAT).
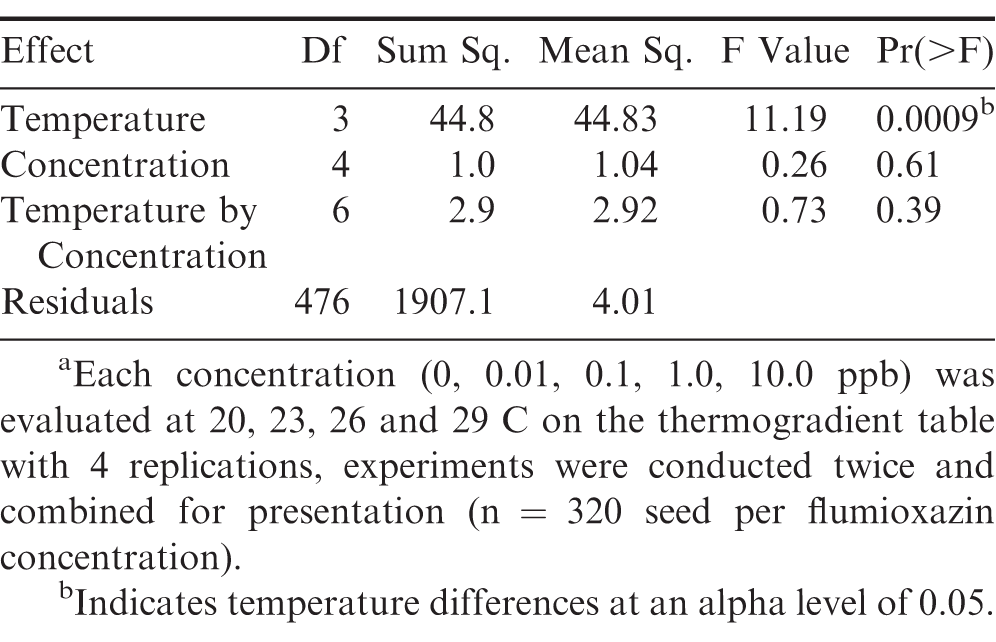
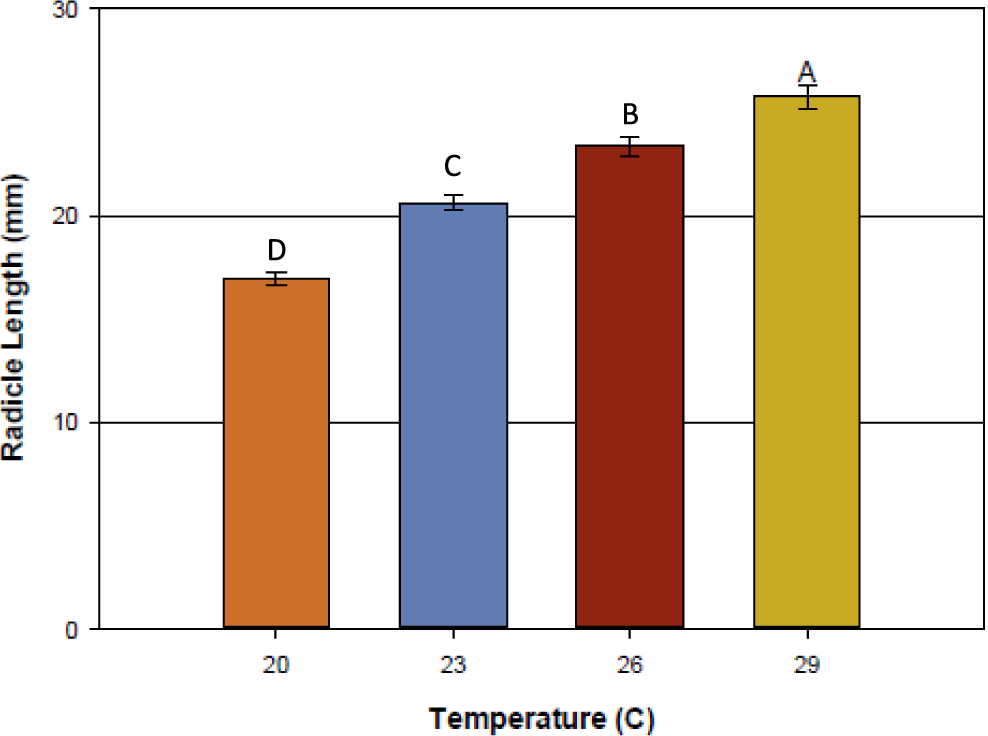
Temperature affected peanut germination more than flumioxazin. Peanut seed germination was analyzed (Table 2) and effect of temperature was observed (P=0.0009), while flumioxazin concentration and temperature by concentration had no differences in germination, P=0.61 and P=0.39, respectively. As temperatures decreased, radicle length was inhibited or development was minimal. (Figure 1). This indicates that regardless of temperature, flumioxazin did not adversely affect peanut germination and radicle development. These data are supported by Williams et al. (2015) in which the researchers noted that as temperatures decreased, both germination decreased and radicle development was reduced. It was noted that the 29 C temperature seeds had the highest germination of the studied temperatures, followed by 23, 26, and 20 C and is supported by Grey et al. (2011). Prasad et al. (2006) noted that planting early, which coincides with cooler soil temperatures, results in lower peanut seed germination; whereas the later planting dates with higher soil temperatures resulted in higher germination rates. Though flumioxazin may cause physiological injury in seedlings following emergence, this injury is not caused by root absorption. This is supported by Price et al. (2004) in which the authors concluded that root-absorbed flumioxazin is quickly metabolized and that injury is likely caused from water splashing onto green plant matter during a rain event while peanuts are emerging.
Peanut radicle length response to different temperatures across multiple flumioxazin rates when evaluated on a thermal gradient tablea. aEach temperature contains radicle length measures from flumioxazin concentrations 0, 0.01, 0.1, 1.0, 10.0 ppb with 4 replications. Error bars represent standard error. Data subjected to Tukey's HSD set at an alpha 0.05. Letters followed by each other indicate no significant differences.
McNaughton et al. (2014) reported that flumioxazin at 107 g ai/ha applied on emerged soybean cotyledons resulted up to 18% injury, primarily consisting of leaf necrosis and stunting at 7 days after treatment. At 28 days after treatment, injury from that same rate of flumioxazin was 8%. Lovell et al. (2001) noted that soybean stand counts were reduced and visible plant injury increased as temperatures decreased during crop emergence. The researchers suggested that this was due to reduced metabolism, with some varieties having up to 50% stand reduction with 107 g ai/ha flumioxazin.
Proper planting date, herbicide application, and herbicide activation timings are crucial in establishing healthy peanut stands early in the season. The herbicide label states that peanut may be severly injured if applications are made more than 2 days after planting (Anonymous 2019). Flumioxazin has been noted to cause peanut injury, but has been concluded that this injury does not come from radicle absorption, but rather through splash during a rain event. This injury is transient and typically will not affect yield. Planting peanuts at 10 com in depth when soil temperatures are >20 C for 3 consecutive days, avoids risk of future cold temperatures to achieve maximum seedling emergence (Monfort et al. 2019). These research data support the previous statement by indicating that decrease in temperature result in lower peanut germination and radicle development.
Acknowledgements
The Georgia Peanut Commission, The National Peanut Board, Georgia Seed Development, Sidney Cromer
Literature Cited
Anonymous 2019 Valor Herbicide. Valent USA LLC, 1600 Riviera Ave Suit, 200 Walnut Creek, CA 94596. http://www.cdms.net/ldat/ldEU7000.pdf Accessed: Sept 17 2019 .
Black, G.W 2018 Georgia seed law and rules and regulations. Georgia Dept. of Ag., Ag. Inputs Division, 19 M.L King Dr SW, Atlanta GA 30334.
Everman, W.J., I.C Burke, S.B Clewis, W.E Thomas, J.W Wilcut 2008 a Critical period of grass vs. broadleaf weed interference in peanut Weed Technol 22 (1): 68- 73.
Everman, W.J., S.B Clewis, W.E Thomas, I.C Burke 2008 b Critical period of weed interference in peanut Weed Technol 22 (1): 63- 67.
J.M Green, and M.D.K Owen (2011). Herbicide-resistant crops: utilities and limitations for herbicide-resistant weed management. J Agric Food Chem 59 ((11)): 5819- 5829.
Grey, T.L., J.P Beasley Jr., T.M Webster, C.Y Chen 2011 Peanut seed vigor evaluation using a thermal gradient Int. J. of Agron. Volume 2011. 7 pages.
Grey, T.L., C.Y Chen, R Nuti, W.S Monfort, G.C. III. 2017 Characterization of genotype by planting date effects on runner-type peanut seed germination and vigor response to temperature In Advances in Seed Biology Pg 103-122.
Grichar W.J and P.A Dotray (2011) Weed control and peanut tolerance with ethalfluralin-based herbicide systems Intl. J. of Agronomy Vol. 2012 8 pages.
K.N Harker, and J.T O'Donovan (2013). Recent weed control, weed management, and integrated weed management. Weed Technol 27: 1- 11.
I Heap, (2013). Global perspective of herbicide-resistant weeds. Pest Manag. Sci 70 ((9)): 1306- 1315.
Heap, I 2019 The international survey of herbicide resistant weeds Online. Accessed August 14th, 2019. http://weedscience.com/Summary/MOA.aspx.
W.C., Johnson E.P Prostko, B.G Mullinix (2006). Phytotoxicity of delayed applications of flumioxazin on peanut (Arachis hypogaea). Weed Technol 20: 157- 163.
S.T., Lovell, L.M Wax, R Nelson (2001). Phytotoxic response and yield of soybean (Glycine max) varieties treated with sulfentrazone or flumioxazin. Weed Technol 15: 95- 102.
D.L Ketring, ((1992)). Physiology of oil seeds. X. Seed quality of peanut genotypes as affected by ambient storage temperature. Peanut Sci 19 ((2)): 72- 77.
C.L., Main, J.T Ducar, E.B Whitty, G.E MacDonald (2003). Response of three runner-type peanut cultivars to flumioxazin. Weed Technol 17: 89- 93.
McDonald M.B 2004 Orthodox seed deterioration and its repair In: Benech-Arnold, R.L and Sánchez, R.A., Eds., Handbook of Seed Physiology P 273- 298.
K.E., McNaughton C Shropshire, D Robinson, P.H Sikkema (2014). Soybean (Glycine max) tolerance to timing applications of pyroxasulfone, flumioxazin, and pyroxasulfone + flumioxazin. Weed Technol 28 ((3)): 494- 500.
Monfort, W.S., E.P Prostko, R.S Tubbs, G Harris, M Abney, W.M Porter B Kemerait 2019 UGA Peanut Production 2019 Quick Reference Guide. University of Georgia. Annual Publication: AP-118.
B.R., Morton, B.L Tillman, D.W Gorbet, K.J Boote (2008). Impact of seed storage environment on field emergence of peanut (Arachis hypogaea) cultivars. Peanut Sci 35: 108- 115.
[NASS] National Agricultural Statistics Service (2019) United States Department of Agriculture https://www.nass.usda.gov/Surveys/Guide_to_NASS_Surveys/Chemical_Use/ Accessed 21 Aug 2019.
Neal, S 2019 USDA crop production. United States Department of Agriculture: National Agricultural Statistics Service: Washington D.C., United States of America ISSN:1936-3737.
Peterson J., K Polzin, K Fogarty, K Albers, L Biondo 2018 AOSA Rules for Testing Seeds: Principles and Procedures. Association of Official Seed Analysts, Inc Pg 57-60.
P.V.V., Prasad, K.J Boote, J.M.G Thomas, L. H Allen D. W Gorbet (2006). Influence of soil temperature on seedling emergence and early growth of peanut cultivars in field conditions. J. Agron. and Crop Sci. 192: 168- 177.
A.J., Price, J.W Wilcut, J.R Cranmer (2004). Physiological behavior of root-absorbed flumioxazin in peanut, ivyleaf morningglory (Ipomoea hederaceae), and sicklepod (Senna obtusifolia). Weed Sci 55 ((2)): 718- 724.
SAS 2019 SAS Institute Inc., Cary, North Carolina 27513-2414.
Shaner, D.L 2014 Pages 212 - 213 In Herbicide Handbook 10th ed Weed Sci. Soc. Amer.
T.M., Webster, W Faircloth, T Flanders, and T.L Grey (2007). Critical period of Benghal dayflower control in peanut (Arachis hypogaea). Weed Sci 55: 359- 364.
A.N., Williams T.L Grey, R.S Tubbs, S.R Cromer (2015). The interaction effects of herbicide and temperature on peanut germination. In American Peanut Research & Education Society Abstracts 47: 92.
A., Wise, T.L Grey, E.P Prostko, W.K Vencill, and T.M Webster (2009). Establishing the geographical distribution and level of acetolactate synthase resistance of Palmer amaranth (Amaranthus palmeri) accessions in Georgia. Weed Technol 23: 214- 220.
Notes
Author Affiliations
1Graduate Research Assistant, Professor, Assistant Professor, Associate Professor, Extension Agronomist, respectively. Department of Crop and Soil Sciences, The University of Georgia, 2360 Rainwater Rd., Tifton, GA 31793.