Introduction
Peanut (Arachis hypogaea L.) is an economically important crop in the southeastern United States; however, fungal diseases are responsible for substantial yield reductions annually (Nutter and Shokes, 1995; Melouk and Backman, 1995). The primary foliar diseases in this region are early leaf spot (caused by Cercospora arachidicola Hori) and late leaf spot (caused by Cercosporidium personatum Berk. & Curt.) Deighton; while southern stem rot (caused by Sclerotium rolfsii Sacc.) and Rhizoctonia limb rot (caused by Rhizoctonia solani Kuhn anastimosis group AG-4) are the most important soilborne diseases.
A recent survey of Georgia peanut producers indicated that approximately 6.2 fungicide applications are made per season for the management of the aforementioned diseases (N. B. Smith, personal communication). Chlorothalonil, a broad spectrum protectant fungicide, is an effective fungicide for the control of leaf spot diseases, and has remained the standard since the 1970s (Smith and Luttrell, 1980); however, chlorothalonil is not effective for control of soilborne pathogens. Pentachloronitrobenzene (PCNB), an organochlorine fungicide, was the first fungicide used extensively for soilborne disease control; however, high costs and inconsistent field results limited producer usage (Csinos, 1989). PCNB was applied as a granule, the logic being that granules were needed to filter down through the canopy to the soil surface for control of soilborne diseases (Csinos, 1989). This same strategy was applied to newer fungicides, such as the sterol demethylation inhibitors (DMI), as they were evaluated on peanut. Granular formulations of diniconazole and tebuconazole were examined, but results were inconsistent (Csinos, 1987); however, suppression of soilborne diseases was observed when these compounds were applied to foliage in leaf spot studies (Backman and Crawford, 1985; Csinos et al., 1987; Brenneman et al., 1991; Brenneman and Culbreath, 1994). By mixing dyes with the foliar-applied fungicides and applying irrigation, Csinos (1986) documented how these materials were delivered to the soil. He demonstrated that the architecture of the peanut plant served to funnel rain or irrigation water along the stems and increase deposition of fungicides at the plant crown and pegs.
In addition to tebuconazole, several other fungicides have been registered for foliar and/or soilborne disease control in peanuts. Flutolanil, a benzanilide fungicide, is highly effective against S. rolfsii and R. solani (Csinos, 1987; Hagan et al., 2004), but has little or no activity against the leaf spot pathogens. Therefore, tank-mixes with other fungicides, such as propiconazole or chlorothalonil, are required for leaf spot control when flutolanil is used (Kemerait et al., 2003). Azoxystrobin, a quinone outside inhibiting (QoI) fungicide, has been shown to be active against both foliar and soilborne diseases (Grichar et al., 2000; Hagan et al., 2004). Fungicide programs utilizing tebuconazole consist of a calendar-based 4-spray block of applications; whereas, flutolanil and azoxystrobin are generally applied 60 and 90 days after planting (Kemerait et al., 2003). The registration, utility, and efficacy of these products have dramatically improved control of soilborne diseases. As a result, these products have become widely accepted by producers.
All fungicides currently used for management of peanut stem rot are applied as foliar sprays, and presumably redistributed with subsequent rainfall and/or irrigation as previously described. However, this process is not well defined. To better understand this phenomenon, particularly with different fungicide classes, a method was needed to quantify residues of fungicides on various parts of the peanut plant. Previous researchers have used excised peanut stems to determine the residual activity of fungicides for the control of Sclerotinia blight, caused by the soilborne fungus Sclerotinia minor Jagger (Brenneman et al., 1988), and southern stem rot (Rideout, 2002). In addition, stem inoculations also proved useful for evaluating differences in susceptibility of plant parts as they aged (Rideout, 2002). The initial objective of the current study was to develop an inoculation method to assay fungicide residues on different plant tissues. The second objective was to use such a method to quantify concentrations of fungicides on peanut leaves and stems.
Materials and Methods
Inoculation Techniques
In 2003, main stems of 45-day-old peanut plants, cultivar Georgia Green, were sampled from non-fungicide treated border rows collected from a field experiment at the University of Georgia Coastal Plain Experiment Station, Gibbs farm (trial 1). The experiment was repeated with 48-day-old plants obtained from non-fungicide treated border rows of a similar experiment at the University of Georgia Coastal Plain Experiment Station, Rigdon farm (trial 2). Main stems were cut at the soil line and taken to the laboratory. One leaflet and stem section (2.5 cm long) were cut from the upper canopy (at or below the second fully expanded leaf), middle canopy (at an intermediate node), and lower canopy (above the node closest to the soil line) of each plant. In all, 48 plants were used for each trial. Excised tissues were then placed in petri dishes (100 × 15 mm) containing moistened, sterile filter paper. Wound treatments consisted of wounded or non-wounded tissues, and were assigned at random to the 48 plants. A sterile dissecting needle was used to create a shallow wound in the center of each tissue. Inoculations were made on wounded or non-wounded tissues by placing hyphal plugs (1-cm diam.) of S. rolfsii isolate SR-18 mycelial side down in the middle of each tissue. Inoculum was obtained from actively growing S. rolfsii colonies on water agar (WA), potato dextrose agar (PDA), half-strength potato dextrose agar (½ PDA), and quarter-strength potato dextrose agar (¼ PDA). Treatments were arranged in a split-plot design with wounding serving as whole plots, and nutrient source as sub-plots. There were a total of six replications per treatment per trial. Petri dishes were placed in a growth chamber, and incubated in the dark at 28°C and 95% relative humidity (RH) for 96 hours. Lesion area (length × width) and lesion length were recorded at 12-hour intervals, for leaflets and stems, respectively. The area under disease progress curve (AUDPC) was calculated using methods described by Shaner and Finney (1977).
Sample Size
To determine an appropriate sample size, thirty main stems of healthy Georgia Green peanut plants were randomly collected from plots in a non-fungicide treated field study in Appling County at 50 DAP during the 2004 growing season. Tissues were inoculated with ¼ PDA plugs containing S. rolfsii SR-18 as previously described. The excised tissues in petri dishes were incubated in the dark at 28°C and 95% RH for 72-hours at which time lesions were measured.
Evaluation of Fungicides
The inoculation method previously described (¼ PDA nutrient source and non-wounded tissues) was used to quantify the response of S. rolfsii isolate SR-18 to varying concentrations of the fungicides azoxystrobin, flutolanil and tebuconazole in planta. Leaflet and stem tissues were collected from non-fungicide treated border rows of Georgia Green peanuts from field plots at the Rigdon farm and Appling County field trial during the 2004 field season at 60 DAP. Standard formulations and field rates, applied in 188 L/ha of water, of azoxystrobin (Abound 2.08F, 0.33 kg a.i./ha, Syngenta Crop Protection, Greensboro, NC), tebuconazole (Folicur 3.6F, 0.23 kg a.i./ha, Bayer CropScience, Research Triangle Park, NC), and flutolanil (Moncut 70DF, 1.0 kg a.i./ha, Gowan Co., Yuma, AZ) were used to prepare four ten-fold serial dilutions. Water containing no fungicide served as a control. Thirty μl of the surfactant Tween 20 (polyoxylethylene sorbitan monolaurate; Qbiogen Inc, Carlsbad, CA) per 100 ml was added to each solution to ensure a relatively uniform distribution of fungicides on the tissue surface. Excised tissues were randomly assigned a fungicide concentration; tissues were immersed in fungicide suspensions for 30 sec, and allowed to dry at room temperature overnight prior to inoculation. Tissues were inoculated as previously described, and lesion size was recorded after 72-hours incubation. Treatments were arranged in a randomized complete block design with five replications per trial.
Statistical Analysis
Lesion length on stems, percentage of colonization of leaflets, and AUDPC values from the inoculation studies were subjected to analysis of variance using Proc ANOVA (SAS Inst, 2005) to determine significant differences (P≤0.05) among treatments. Fisher's protected least significant differences (LSD) were calculated for mean separation within each study. All subsequent references to significant effects of factors, interactions, or differences among means indicate significance at P≤0.05.
To determine a reliable sample size, sample means, standard deviations, and coefficients of variability were plotted against the sample size (n). Sample size was based on the weighted mean of the variance of lesion development from each tissue layer. To account for differences in leaflet size within the canopy, a sub-sample of thirty leaflets was arbitrarily chosen from each canopy layer. Leaflets were blotted on a paper towel, and scanned using a ScanMaker 5900 48-bit color scanner (Microtek Lab Inc., Carson, CA). Leaflet area was estimated using the Assess Image Analysis Software (Lamari, 2002), and lesion size was converted to a percentage of the leaflet area colonized.
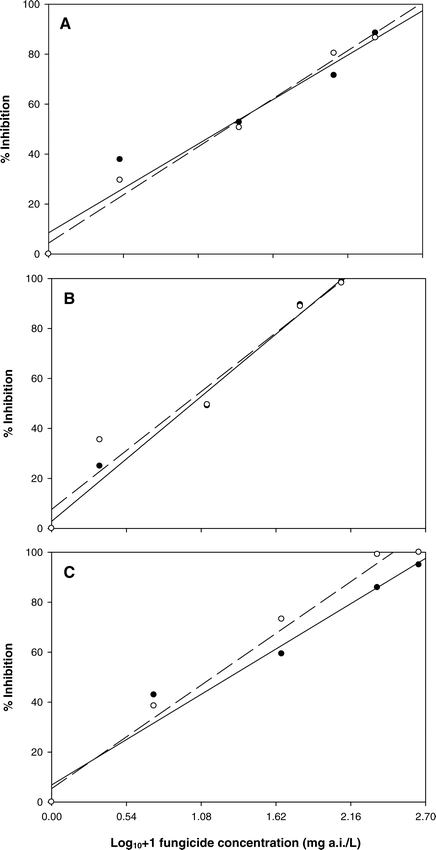
For the fungicide evaluations, the percent inhibition of lesion expansion from each treatment was determined using the equation; % inhibition = 100 − ((lesion measurement ÷ nontreated control) × 100), and values were plotted against their respective log10 transformed fungicide concentration + 1. Data were analyzed using linear regression in Sigma Plot version 9.0 (Systat Software, Inc., Point Richmond, CA). Regression equations were used to estimate fungicide concentrations for 50% inhibition of lesion expansion (EC50).
Results and Discussion
Inoculation Techniques
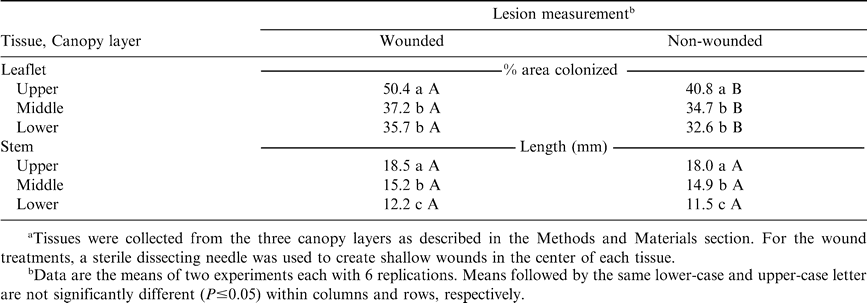
The interaction of trial by nutrient source was not significant; therefore data were pooled across the four nutrient sources to determine the effect of wounding on leaflets and stems. Experiments demonstrated that wounding was not required for infection of either tissue type. Leaflet colonization was significantly greater when wounds were made for all canopy layers; however, stem colonization did not differ between the two inoculation methods (Table 1). These findings provide additional support that S. rolfsii is capable of penetrating non-wounded tissues (Aycock, 1966; Punja, 1985).
Lesion development varied significantly according to tissue origin (Table 1). Stem tissues obtained from the upper canopy were most rapidly colonized by S. rolfsii, followed by tissues from the middle and lower canopy, respectively. Colonization of leaflets followed a similar trend, but layers from the middle and lower canopy layers were not significantly different. These results are similar to a report by Brenneman et al. (1988), in which terminal stem segments were more susceptible to infection by Sclerotinia minor than were basal segments. These results also demonstrate the importance of standardizing tissues collected from different canopy layers.
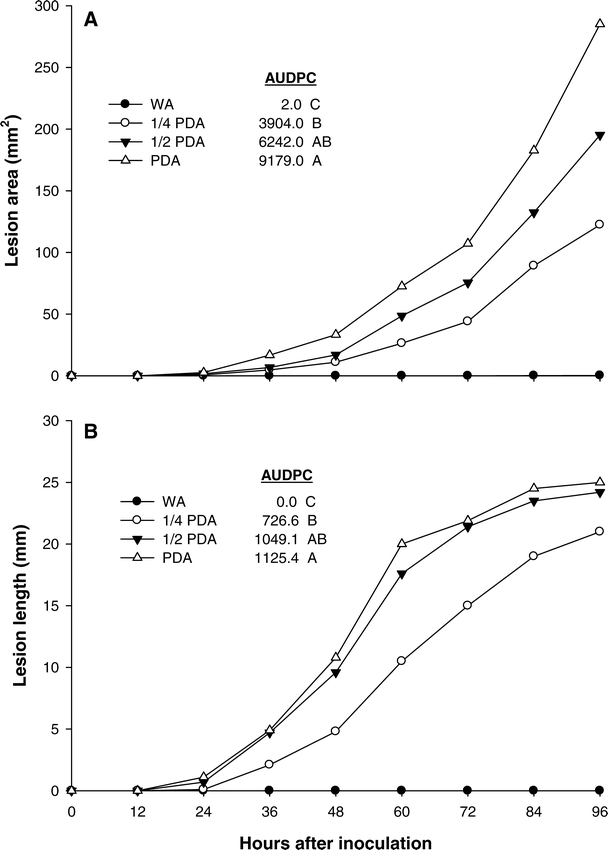
In regard to nutrient source, the analysis of variance showed no significant trial effects; therefore, results were combined over trial. Lesion development on both leaflets and stems was greatest when PDA or ½ PDA was used as a nutrient source (Figure 1). In both assays, WA was an insufficient nutrient source; whereas ¼ PDA produced intermediate lesions which were less variable than the other nutrient sources (data not shown). These results provide evidence that an exogenous nutrient source is required to initiate infection when using mycelia, and the severity of those infections is directly related to the strength of the nutrient source. Previous research has shown that mycelia from germinating sclerotia can infect host tissue without an exogenous nutrient source (Punja, 1985); however, mycelial growth is influenced by several factors including release of volatile compounds from decaying plant tissues, and nitrogenous amendments (Melouk and Backman, 1995; Punja, 1985). Lesion development was slower for non-wounded tissues inoculated with ¼ PDA plugs containing S. rolfsii mycelium, thus it was better to differentiate treatment effects. This method was used in all subsequent experiments.
Lesion development on excised peanut (A) leaflets and (B) stems inoculated with Sclerotium rolfsii grown on four nutrient sources (WA = water agar, PDA = potato dextrose agar ¼ PDA = quarter strength PDA, ½ PDA = half strength PDA). Lesions were measured every 12 hours for 96 hours. Data were used to construct disease progress curves, and the area under disease progress curve (AUDPC) was calculated (Shaner and Finney, 1977) for each treatment. Means followed by the same letter within a column are not significantly different (P≤0.05) according to Fisher's protected least significant differences test.
Sample Size
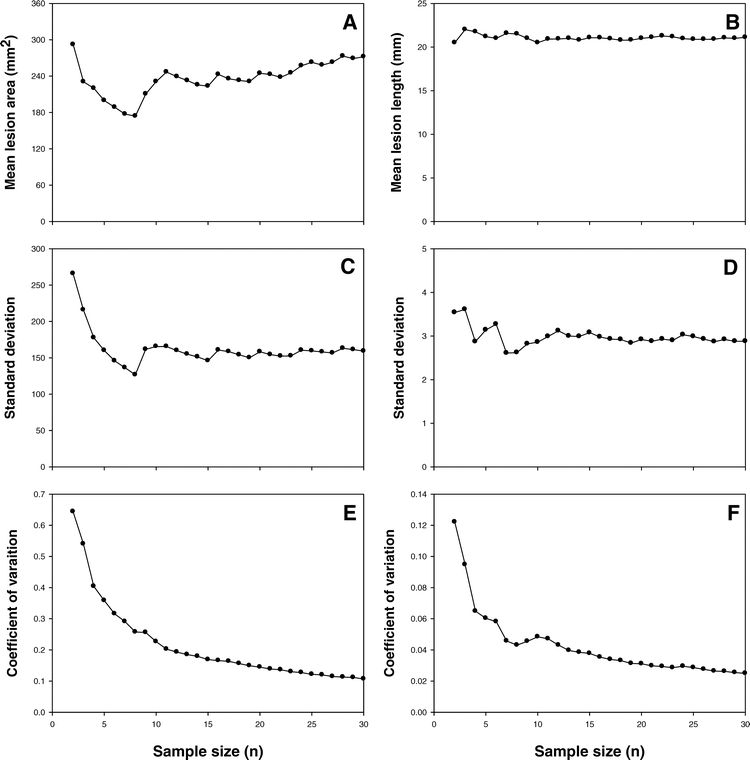
Increasing the sample size results in a more accurate estimate of parameters (Steel and Torrie, 1980). By using the sample mean, standard deviation, and coefficient of variation as a function of sample size, inferences can be made about the optimum number of samples needed to obtain the most desired level of accuracy. In these studies, mean lesion length and percent colonization differed by tissue origin, and were greatest for the tissues collected from the upper canopy and lowest for tissues collected from the middle and lower canopies, and reinforces the importance of standardizing tissues when sampling throughout the canopy.
Although differences in lesion size were observed among tissues from different canopy layers, no treatment × canopy layer interaction was observed; therefore, data from all three layers were combined for analysis. Variance for both the leaflet, and stem assay were similar. For the leaflet assay, the sample mean, standard deviation, and coefficient of variation stabilized at sample sizes of 11, 9, and 10, respectively (Figure 2 a, c, and e). For the stem assay, reliable sample size based on the mean, standard deviation, and coefficient of variation were found to be 8, 10, and 11, respectively (Figure 2 b, d, and f). Based on these results maximium sample sizes of 10 observations are needed to obtain reliable parameter estimates for leaflet or stem segments. However, other factors such as the availability of space or materials may influence sample sizes.
Evaluation of Fungicides
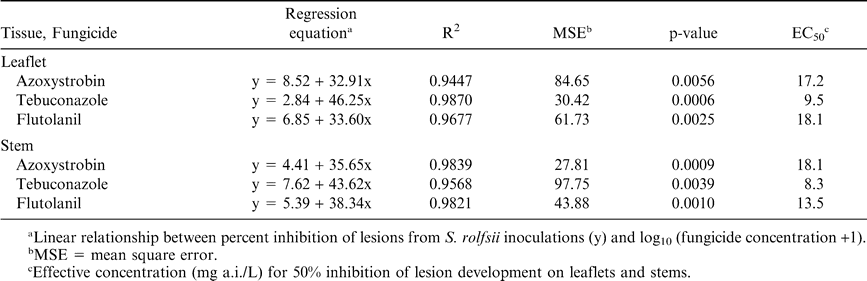
There were no canopy layer × concentration interactions for lesion development in either assay; therefore, data were pooled across canopy layer. Lesion development was significantly reduced for all concentrations of azoxystrobin, flutolanil, and tebuconazole when compared to the non-fungicide amended control (data not shown). Fungicide performance was similar on both leaflet and stem tissues for all three compounds (Table 2, Figure 3). A positive linear relationship was found between the log transformed fungicide concentration+1 and inhibition of lesion development. The percent inhibition increased linearly as fungicide concentration increased. Linear relationships were significant for all fungicides and tissues (Table 2). In general, the inhibition of lesions was greater in the stem assay than in leaflet assay, which may have been due to differences in susceptibility between the two tissues. Similar trends in the EC50 values were observed for azoxystrobin, flutolanil, and tebuconazole in both tissue types (Table 2). Field studies have demonstrated that azoxystrobin, tebuconazole, and flutolanil are highly efficacious against stem rot (Csinos, 1987; Brenneman and Culbreath, 1994; Grichar et al., 2000; Hagan et al., 2004), and those findings are supported in this study. Isolate SR-18 was more sensitive to tebuconazole than azoxystrobin or flutolanil in both assays based on the lower EC50 values (Table 2).
One potential explanation for the observed differences in fungicide activity could be related to protectant or systemic activity of the compounds evaluated. Fungicides within a chemical class can have very different physiochemical properties; therefore one would expect even greater differences among classes than among fungicides within the same class. Araki (1985) reported that flutolanil is absorbed by roots, and translocated acropetally in rice (Oryza sativa L.); however, little information regarding the systemic activity of flutolanil in peanut is available. Although comparisons in absorption between leaflet and stem tissues were not made in the current study, it appears that flutolanil was more readily absorbed into stems than leaflets. This effect could simply be an artifact of differences between the two tissue types; therefore, additional studies may be required to determine differences in the absorption of fungicides between peanut tissues.
The systemic properties of DMI and QoI fungicides are far more defined than those of flutolanil. Studies conducted by Tsuda et al. (2004) documented the translaminar (movement from the upper leaf surface to the lower leaf surface) and transcuticular (movement through the cuticle) activity of several DMI fungicides, including tebuconazole. They found that fungicide efficacy against powdery mildew (Sphaerotheca cucurbitae (Jaczewski) Zhao) on cucumber (Cucumis sativus L.) seedlings resulted from permeation and dissipation of the fungicides within leaves. The foliar uptake of azoxystrobin is a gradual process with relatively low amounts of applied material being absorbed within 24 hours of application (Bartlett, et al., 2002). Furthermore, tissue type and age have also been shown to influence uptake of azoxystrobin in a broad range of commercial field crops (Bartlett, et al., 2002). The effect of tissue age on uptake of azoxystrobin could not be ascertained, due to the confounding effects of tissue susceptibility from the three canopy layers.
Conclusions
Various assays have been developed using excised peanut tissues to study different aspects of peanut disease management. Detached lateral branches were used to evaluate cultivar susceptibility, and isolate virulence of Sclerotinia minor (Brenneman, et al., 1988). Detached shoots, limbs, leaves or leaflets have been used to successfully screen cultivars for resistance to several peanut pathogens (Franke and Brenneman, 2001; Hollowell and Shew, 2003; Melouk et al., 1992). Such methods also have been used to determine the residual activity of fungicides used to control peanut diseases (Brenneman et al., 1988; Rideout, 2002).
Methods using excised tissue are efficient relative to traditional field studies in that they require relatively small amounts of plant material, reduce labor, and can be conducted in less time under controlled environmental conditions. The method developed in this study proved to be useful for determining the susceptibility of peanut tissues from different canopy layers to S. rolfsii. Using this method, we demonstrated that tissues from the upper canopy are more susceptible to infection than those from the middle and lower canopy. These differences must be considered in subsequent studies where samples are taken within the peanut canopy. These findings also indicate that inoculation with S. rolfsii is a sensitive means of estimating concentrations of fungicide residues on/in peanut tissues, and that the method described can be used to determine the deposition and redistribution of foliar-applied fungicides.
Acknowledgements
This project was funded in part by a grant from the Georgia Agricultural Commodity Commission for Peanuts. Appreciation is expressed to K. Mullis, P. Hilton and E. Jackson for technical assistance. We also thank R. Kemerait, A. Cubreath, and K. Stevenson for providing helpful reviews of the manuscript prior to submission.
Literature Cited
Araki F. 1985 Moncut (Flutolanil), a new systemic fungicide. Japan Pesticide Info. No 47 : 23 – 25 .
Aycock R. A. 1966 Stem rot and other diseases caused by Sclerotium rolfsii. N. C. Agric. Exp. Sta. Tech. Bull 174 .
Backman P. A. and Crawford M. A. 1985 Effects of triazole fungicides on soilborne diseases of peanuts. Proc. Am. Peanut Res. and Ed. Soc 17 : 42 (abstr.) .
Bartlett D. W. , Clough J. M. , Godwin J. R. , Hall A. A. , Hamer M. , and Parr-Bobrzanski B. 2002 Review: The strobilurin fungicides. Pest. Manag. Sci 58 : 649 – 662 .
Brenneman T. B. and Culbreath A. K. 1994 Utilizing a sterol demethylation inhibiting fungicide in an advisory to manage foliar and soilborne pathogens of peanut. Plant Dis 78 : 866 – 872 .
Brenneman T. B. , Murphy A. P. , and Csinos A. S. 1991 Activity of tebuconazole on Sclerotium rolfsii and Rhizoctonia solani, two soilborne pathogens of peanut. Plant Dis 75 : 744 – 747 .
Brenneman T. B. , Phipps P. M. , and Stipes R. J. 1988 A rapid method for evaluating genotype resistance, fungicide activity, and isolate pathogenicity of Sclerotinia minor in peanut. Peanut Sci 15 : 104 – 107 .
Csinos A. S. 1986 Aiming the magic bullet for Sclerotium rolfsii. Proc. Am. Peanut Res. Educ. Soc 18 : 58 (abstr.) .
Csinos A. S. 1987 Control of southern stem rot and Rhizoctonia limb rot of peanut with flutolanil. Peanut Sci 14 : 55 – 58 .
Csinos A. S. 1989 Targeting fungicides for control of southern stem rot on peanut. Plant Dis 73 : 723 – 726 .
Csinos A. S. , Kvien C. S. , and Littrell R. H. 1987 Activity of diniconazole on foliar and soilborne diseases of peanut. Appl. Agric. Res 2 : 111 – 116 .
Franke M. D. and Brenneman T. B. 2001 Evaluation of detached shoot and leaflet inoculation techniques to screen peanut genotypes for resistance to Rhizoctonia limb rot. Peanut Sci 28 : 24 – 28 .
Grichar W. J. , Besler B. A. , and Jaks A. J. 2000 Use of azoxystrobin for disease control in Texas peanut. Peanut Sci 27 : 83 – 87 .
Hagan A. K. , Rivas-Davila M. E. , Bowen K. L. , and Wells L. 2004 Comparison of fungicide programs for the control of early leaf spot and southern stem rot on selected peanut cultivars. Peanut Sci 31 : 22 – 27 .
Hollowell J. E. and Shew B. B. 2003 Evaluating isolate aggressiveness and host resistance from peanut leaflet inoculations with Sclerotinia minor. Plant Dis 87 : 402 – 406 .
Kemerait R. C. , Brenneman T. B. , and Culbreath A. K. 2003 Peanut disease control. 114 – 115 In Guillebeau P. Georgia Pest Control Handbook Athens Univ. of Georgia Special Bull. 28, .
Lamari L. 2002 Assess Image Analysis Software for Plant Disease Quantification St. Paul, MN APS Press .
Melouk H. A. , Akem C. N. , and Bowen C. 1992 A detached shoot technique to evaluate the reaction of peanut genotypes to Sclerotinia minor. Peanut Sci 19 : 58 – 62 .
Melouk H. A. and Backman P. A. 1995 Management of soilborne fungal pathogens. 75 – 82 In Melouk H. A. and Shokes F. M. Peanut Health Management St. Paul, MN American Phytopathological Society Press .
Nutter F. W. and Shokes F. M. 1995 Management of foliar diseases caused by fungi. 65 – 74 In Melouk H. A. and Shokes F. M. Peanut Health Management St. Paul, MN American Phytopathological Society .
Punja Z. K. 1985 The biology, ecology, and control of Sclerotium rolfsii. Annu. Rev. Phytopathol 23 : 97 – 127 .
Rideout S. L. 2002 Influence of environment and host growth for improved fungicide applications for control of southern stem rot of peanut Ph.D. dissertation, Athens Univ. of Georgia .
SAS Instituite Inc 2005 SAS User's Guide to the Statistics Analysis System Cary, NC Version 9.1, .
Shaner G. and Finney P. E. 1977 The effect of nitrogen fertilizer on expression of slow mildewing resistance in Knox wheat. Phytopathol 67 : 1051 – 1056 .
Smith D. H. and Luttrell R. H. 1980 Management of peanut foliar diseases with fungicides. Plant Dis 64 : 356 – 361 .
Steel R. G. B. and Torrie J. H. 1980 Principles and Procedures of Statistics New York McGraw-Hill .
Tsuda M. , Itoh H. , and Kato S. 2004 Evaluation of the systemic activity of simeconazole in comparison with that of other DMI fungicides. Pest. Manag. Sci 60 : 875 – 880 .
Notes
- Former Graduate Student and Professor, respectively, Dept. of Plant Pathology, The University of Georgia, Coastal Plain Experiment Station, Tifton, GA 31793 [^] * Corresponding author (email: jewoodward@ag.tamu.edu)
Author Affiliations