Introduction
Early leaf spot, caused by Cercospora arachidicola S. Hori, (teleomorph = Mycosphaerella arachidis Deighton) (Ca) and late leaf spot, caused by Cercosporidium personatum (Berk. & M. A. Curtis) Deighton, (teleomorph = Mycosphaerella berkeleyi Jenk.) (Cp) are the most important foliar diseases affecting peanut (Arachis hypogaea L.) throughout the world (Shokes and Culbreath, 1997). The diseases often occur together or one disease may be more predominant in a given location or year. In Georgia, late leaf spot was more common in the 1980s and early 1990s, while early leaf spot became predominant in much of Georgia during the late 1990s. Late leaf spot is still predominant in Florida and lately has again increased its prevalence in Georgia. For this reason it is critical to evaluate the resistance of runner-type peanut genotypes, the type most widely grown throughout Georgia, Alabama, and Florida, to combat the menace of both Ca and Cp. Fields tests can be used for evaluation of resistance to the prevalent pathogen, but greenhouse or growth chamber evaluations should be utilized when field evaluations to both pathogens are not feasible.
There has been no complete or single-gene resistance to Ca or Cp reported in cultivated peanut. Resistance is partial and rate-reducing (Abdou et al., 1974). Partial resistance is typically a function of multiple components of resistance that contribute additively to a reduction in the rate of epidemic progress (Parlevliet, 1979). Components of resistance to early leaf spot (Foster et al., 1980; Green and Wynne, 1986; Melouk and Banks, 1984; Ricker et al., 1985) and/or late leaf spot (Chiteka et al., 1988a; Cook, 1981; Subrahmanyam et al., 1982; Walls et al., 1985; Watson et al., 1998) were described for many peanut genotypes under field and greenhouse conditions in the 1980s. However, relatively little work has been published for peanut cultivars and breeding lines developed within the past 20 years (Anderson et al., 1993; Aquino et al., 1995; Chiteka et al., 1997; Chiyembekeza et al., 1993), despite significant advances in breeding for resistance to leaf spot diseases during this time. Very little work has been accomplished to investigate the resistance of genotypes to both leaf spot pathogens (Walls et al., 1985), even though the inheritance of resistance to each pathogen appears to be independent (Wynne et al., 1991).
Variation exists for nearly all resistance components investigated for early and late leaf spots. These include infection frequency, incubation period (time from inoculation to symptom appearance), latent period (time from inoculation to first sporulating lesion), lesion size, percent necrotic leaf area, percent lesions with sporulation, spore production, and time to defoliation. Latent period, lesion size and spore production have been the components most commonly associated with genetic resistance (Chiteka et al., 1988a; Chiteka et al., 1988b; Chiyembekeza et al., 1993; Walls et al., 1985). Infection frequency is highly dependent upon temperature and relative humidity (Shew et al., 1988; Waliyar et al., 1994) and has been suggested to be an unreliable measure of resistance (Ricker et al., 1985; Waliyar et al., 1993). Examinations of multiple resistance components within resistance genotypes have shown that components are often correlated. For example, Jogloy et al. (1987) and Chiteka et al. (1988b) reported a positive genetic relationship between late leaf spot lesion size and the quantity of secondary spores produced, both of which were negatively correlated with latent period. However, no single component has been identified as a primary or consistent predictor of resistance in the field. The stability of resistance components to Ca can vary across growing regions (Chiteka et al., 1997; Waliyar et al., 1993) due to environmental interactions (Shew et al., 1988; Waliyar et al., 1994), pathogen populations (Waliyar et al., 1993) or both (Chiteka et al., 1997). However, Shew et al. (1989) reported stable response of peanut genotypes to Cp isolates from the U.S. and Thailand.
The cultivar Georgia Green (Branch, 1996) is currently the predominant runner-type cultivar grown in the southeastern U.S. (Smith, 2003). The levels of resistance to early and late leaf spots of Georgia Green are slightly better than that of Florunner (Norden et al., 1969; Branch and Culbreath, 1995), which was used as a susceptible check in studies of components of resistance of runner-type genotypes conducted in the 1980s (Aquino et al., 1995; Chiteka et al., 1988a). In a recent field study (Cantonwine et al., 2006), the field resistance to early leaf spot was assessed for Georgia Green and seven newly released cultivars or advanced breeding lines (C-99R, Hull, DP-1, GA-01R, C11-2-39, C28-305 and C34-24), all of which were developed for resistance to early and leaf spots and/or spotted wilt, caused by Tomato spotted wilt virus. Cultivar DP-1, released by the University of Florida peanut breeding program, had the best field resistance to early leaf spot among the genotypes tested (Cantonwine et al., 2006), and reportedly has better resistance to late leaf spot than any cultivar currently available in the U.S. (Gorbet, 2003). The cultivar Georganic (C.C. Holbrook, unpubl. data, 2006), previously tested as breeding line C11-2-39, had an intermediate level of resistance relative to Georgia Green and DP-1 (Cantonwine et al., 2006), and has been shown to have good field resistance to late leaf spot (Cantonwine et al., 2003). This study was conducted to describe the temporal progression of leaf spot diseases in the field and some of the components of host resistance to Ca and Cp expressed in peanut cultivars Georgia Green, DP-1, and Georganic.
Materials and Methods
Evaluation of Leaf Spot Epidemic Progress in the Field
A field experiment was carried out in 2002 and 2003 at the Coastal Plain Experiment Station Ridgon Farm in Tifton, GA to monitor temporal progress of naturally occurring leaf spot epidemics in plots planted with Georgia Green, DP-1, and Georganic peanuts. The experimental design was a randomized complete block with three replications of each genotype per year. Peanut seed were planted in 91-cm spaced single rows in conventionally tilled plots (1.8 × 6 m) on 17 May 2002 and 20 May 2003. No fungicides were applied. Herbicides, insecticides, and fertilizers were applied following recommendations of the University of Georgia Extension Service.
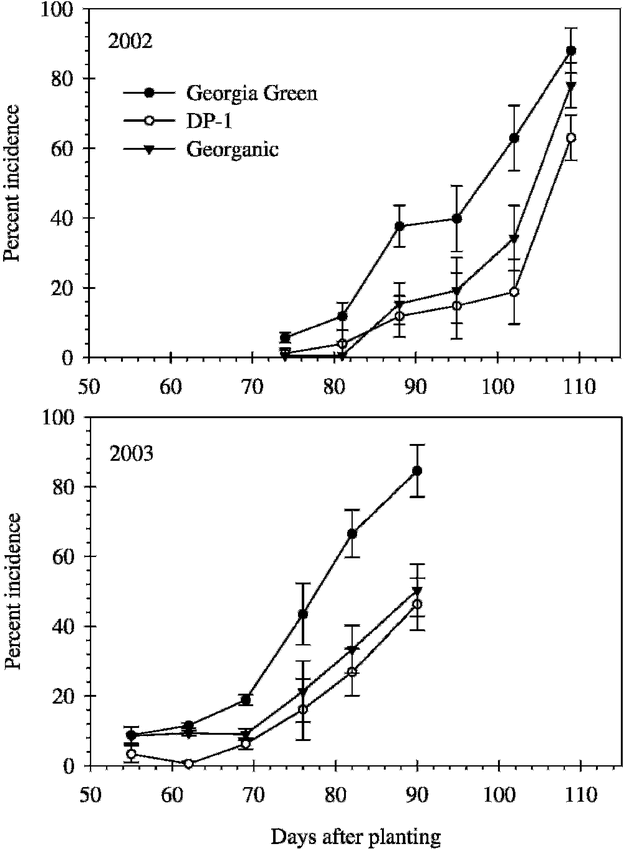
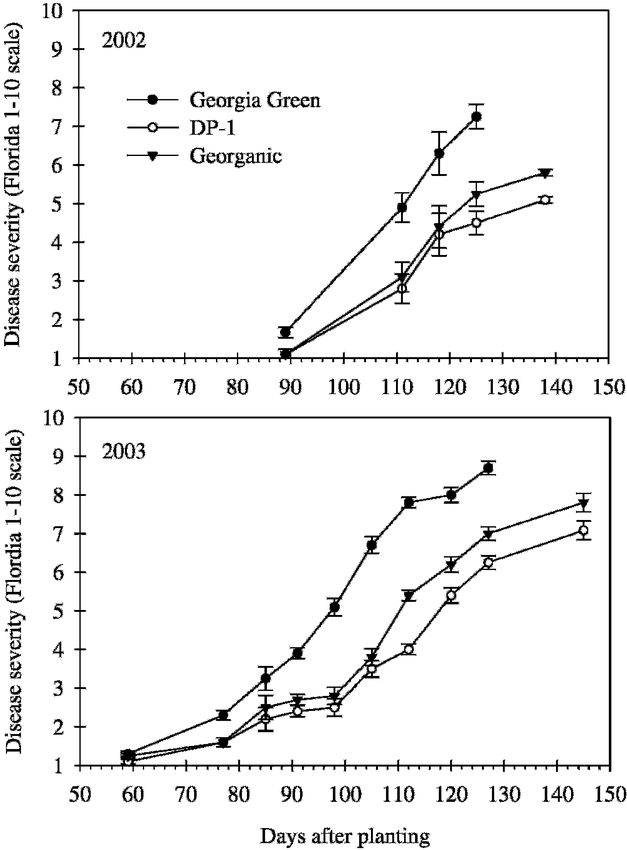
Disease incidence (the percentage of leaves with one or more lesions or defoliated leaflets) was assessed on leaves of 10 lateral branches (secondary branches 1-6) arbitrarily collected from each plot. Assessments began 78 d after planting (DAP) in 2002 and 59 DAP in 2003, and continued weekly for 6 wk. Disease severity was monitored over the season using the Florida 1 to 10 scale system, where 1 = no leaf spot; 2 = very few lesions on the leaves, none on the upper canopy; 3 = few lesions on the leaves, very few on the upper canopy; 4 = some lesions with more on the upper canopy, 5% defoliation; 5 = lesions noticeable even on upper canopy, 20% defoliation; 6 = lesions numerous and very evident on upper canopy, 50% defoliation; 7 = lesions numerous on upper canopy, 75% defoliation; 8 = upper canopy covered with lesions, 90% defoliation; 9 = very few leaves remaining and those covered with lesions, 98% defoliation; and 10 = plants completely defoliated and killed by leaf spot (Chiteka et al., 1988a). Severity assessments were made at 7 to 22 d intervals four to five times beginning 89 DAP in 2002, and nine to ten times beginning 59 DAP in 2003. One less severity assessment was taken for Georgia Green plots because Georgia Green matures earlier than DP-1 and Georganic, and the plants in these plots were inverted 11 to 13 d earlier than in the DP-1 and Georganic plots. Genotype means of disease incidence and severity were plotted individually by assessment date of each year.
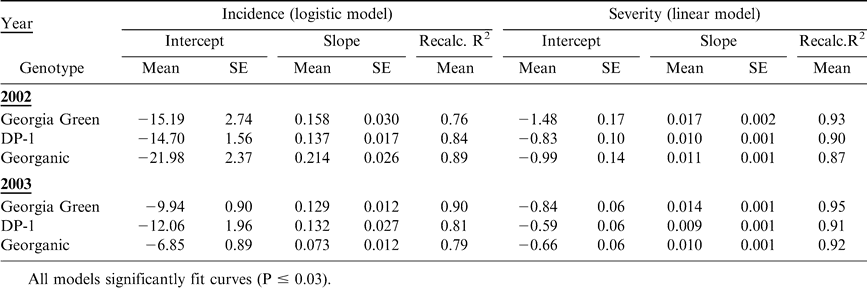
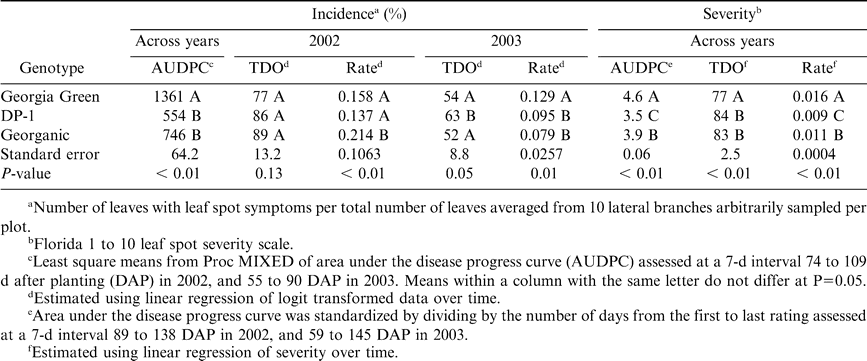
Disease incidence and severity data were analyzed separately. For each plot, area under the disease progress curve (AUDPC) was computed (Shaner and Finney, 1977). Disease assessments were converted to proportions [proportion of incidence = percent incidence/100; proportion of severity assessment = (Florida rating - 1)/9], and linearized forms of the Gompertz [-ln(-ln y)], logistic [ln(y/1-y)] and monomolecular [ln(1/1-y)] models were fit using linear regression of transformed disease intensity proportions on time (DAP), as described by Cantonwine et al., 2007. Time of disease onset (TDO) was estimated by calculating the time (DAP) when the model predicted 5% incidence or 1.5 on the Florida scale. Effects of genotype on AUDPC, TDO and the epidemic rate parameter (r) estimates were determined using the Proc MIXED procedure (SAS Version 8.3, SAS Institute, Cary, NC). When significant year by genotype interactions occurred, years were analyzed separately. Otherwise, analyses were conducted across years. Differences among genotype levels were determined using Fisher's LSD values using standard error and t-values of adjusted degrees of freedom when the main effect was significant (P < 0.05).
Evaluation of Components of Resistance to Leaf Spot Pathogens
A growth chamber experiment was carried out to assess various components of resistance of the cultivars Georgia Green, DP-1, and Georganic to infection by Ca and Cp under a controlled environment. The experimental design was a randomized complete block, with 12 replications of each of the three genotype treatments. The experiment was conducted twice beginning 29 Nov. 2003 and 21 Apr. 2004.
Plants were grown in 15-cm pots with Promix potting soil and maintained in a greenhouse for 9 wk. Three young first or second fully expanded leaves were cut at the base of the petiole from lateral branches on each of 12 plants of each genotype. The cut ends were dipped in a dry formulation of napthaleneacetamide and thiram (Rootone, Security Products Co., Atlanta, GA) and placed individually in sterile saturated sand in 100-ml beakers (Waliyar et al., 1995).
For inoculations, conidia of Cp were acquired by agitating leaf discs of sporulating late leaf spot lesions from Georgia Green leaves that were collected in Tifton, GA and stored at 10°C for 3 to 9 mo in a solution of 0.005% Tween 20. Conidia of Ca were acquired by rinsing conidia from single-conidium cultures isolated from infected Georgia Green peanut leaves collected in Tifton, GA the previous growing season using a technique modified from Lu et al. (2003) and described by Cantonwine et al. (2007) using a 0.005% Tween 20 rinse. Conidial suspensions were standardized to a concentration of 1.0 × 104 conidia/ml and each leaf was inoculated by spraying for 1 sec with an aerosol spray bottle. A solution of 0.005% Tween 20 without conidia was the control. Viability of conidia from inoculation suspensions was verified by measuring spore germination on 2% water agar plates after 24 hr incubation at 22 C under incandescent light.
Leaves in beakers were randomly positioned on trays and placed within a transparent enclosure in a growth chamber at 24 C, 90% RH, and 12-hr photoperiod to provide optimal conditions for Ca germination and infection (Waliyar et al., 1995). The enclosure (1.3 × 0.7 × 0.6 m) was constructed using PVC pipe for the frame, clear plastic for the sides, and overlapping sheets of glass for the top. The enclosure did not affect air temperature. Relative humidity was supplemented by maintaining standing water in trays and with two humidifiers (PersonalMist ultrasonic humidifier, Kaz, Inc, Hudson, NY) evenly spaced within the enclosure. Water was added to beakers and trays as needed. Severely wilted leaves were excluded from measurements.
Lesion numbers per leaf were counted at 17 and 30 d after inoculation (DAI). The percent incubation period 17 DAI was computed by dividing the number of lesions from the 17 DAI count by the 30 DAI count and multiplying by 100. Latent period, defined as DAI until one lesion sporulated, was determined by inspecting spots daily with a 20X magnification lens beginning 17 DAI. After sporulation was observed on one lesion per leaf, or at 30 DAI if no sporulation was apparent, leaves were removed from the growth chamber and placed in a light box at 100% RH chamber for 72 hr, after which the percentage of spots with sporulation was determined. Latent period was not recorded for leaves if sporulation was not apparent 33 DAI. Three sporulating lesions per leaf were randomly selected, excised with a razor blade and placed in a 1-ml tube with 0.1 ml of 0.005% Tween 20 for spore quantification. The necrotic area of each selected lesion was measured. Number of spores per lesion, per unit sporulating lesion area, and per unit leaf area were determined. In cases where no sporulation occurred, the number of stroma was assessed. Percent necrotic area per leaf was estimated for a sub-sample of 10 to 50 randomly selected lesions per leaf. Infection frequency at 17 and 30 DAI was determined by dividing the number of lesions at each DAI by leaf area.
Effects of genotype on resistance variables and square root transformations of variables were tested using the Proc MIXED procedure. Due to unbalanced data, whenever the standard errors were reasonably similar, the largest standard error was used for means comparisons rather than computing a weighted standard error. Fisher's LSD values were computed using standard errors and t-values of adjusted degrees of freedom when the main effect was significant (P < 0.05). Comparisons of untransformed means of transformed variables were determined based on analysis of transformed variables, since variances cannot be transformed (Steel and Torrie, 1980).
Results
Evaluation of Leaf Spot Epidemic Progress in the Field
Early leaf spot was the predominant disease in the field study. Late leaf spot occurred late in the season (>100 DAP) in both years, but the disease never exceeded 10% of the leaf spots combined. For this reason, the field epidemics will be referred to as early leaf spot.
Early leaf spot epidemics began later and were less severe in 2002 than in 2003 (Fig. 1–2). Disease progress curves for early leaf spot incidence varied by genotype (Fig. 1) and were best described by the logistic model (P ≤ 0.03) (Table 1). Disease progress curves for severity also varied by genotype (Fig. 2) and were best described by the linear model (P ≤ 0.01) (Table 1).
Across years, early leaf spot epidemics, measured as AUDPC based on incidence (AUDPC-I) and severity (AUDPC-S), were more severe for Georgia Green, than DP-1 and Georganic (Table 2). AUDPC-I was comparable between the two resistant genotypes, while AUDPC-S was significantly lower for DP-1 than Georganic. There was a significant interaction between year and genotype for incidence based estimates of TDO (TDO-I) and epidemic rate (rate-I). Estimates of TDO-I were 9 d later for DP-1 than Georgia Green both years, but this difference was only statistically significant in 2003 (Table 2). In 2002, TDO-I of Georganic was more similar to TDO-I of DP-1 than Georgia Green, while in 2003, TDO-I of Georganic was more similar to TDO-I of Georgia Green than DP-1. Estimates of rate-I were lower for Georganic than Georgia Green and DP-1 in 2002, and lower for DP-1 and Georganic than Georgia Green in 2003. Across years, severity based estimates of TDO were statistically later for DP-1 and Georganic than Georgia Green, and severity based epidemic rates were highest for Georgia Green and lowest for DP-1 (Table 2).
Evaluation of Components of Resistance to Leaf Spot Pathogens
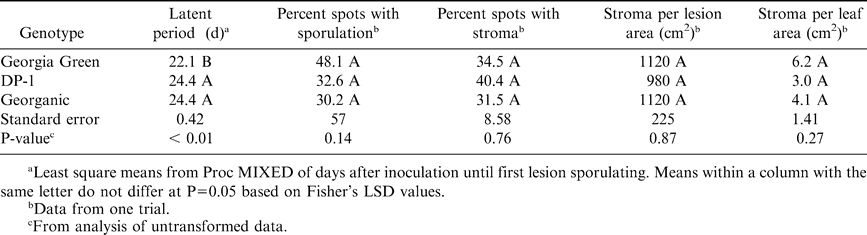
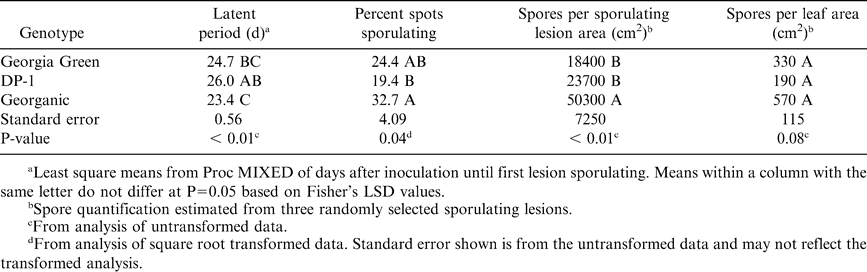
In the growth chamber experiment, leaf spot symptoms were observed on all inoculated leaves and none were observed on control leaves. After 24 hr, germination of Ca and Cp conidia of the inoculum suspensions were approximately 87 and 65%, respectively, in both trials. Across trials and pathogen exposures, five to seven leaves of each genotype were excluded from analyses due to severe wilting that appeared to impede lesion development. The pattern of wilting appeared to be random and the cause was not evident. Infection frequency of Ca did not differ among genotypes 17 DAI, but was higher for Georgia Green 30 DAI (Table 3). Percent incubation period 17 DAI did not differ among genotypes (Table 3). Lesion diameter was 16–21% greater for Georgia Green than for the other genotypes, while percent necrotic area was 47 and 60% greater for Georgia Green than for DP-1 and Georganic, respectively (Table 3). The latent period was 2 d longer for DP-1 and Georganic compared to Georgia Green, but no other differences in early leaf spot reproductive patterns were detected among genotypes (Table 4).
Infection frequency of Cp was significantly lower for DP-1 at 17 DAI, and for DP-1 and Georganic at 30 DAI compared to Georgia Green (Table 5). Percent incubation period at 17 DAI was lower for DP-1 than Georganic (Table 5). Lesion diameter for Georgia Green was 15% greater than Georganic and DP-1, while percent necrotic area was approximately 46% greater for Georgia Green than the other genotypes (Table 5). The latent period was more than 2 d shorter and percent sporulating lesions was greater for Georganic than DP-1 (Table 6). Spores per sporulating lesion area were significantly higher for Georganic than the other genotypes, but spores per leaf area were similar for all genotypes (Table 6).
Discussion
The field study provided a suitable setting to characterize field resistance of Georgia Green, DP-1 and Georganic to Ca but not to Cp, since early leaf spot was the predominant disease both years. Differences in the onset and severity of early leaf spot epidemics by year can be attributed in part to 60% less rainfall during the growing season in Tifton, GA in 2002 than in 2003. Genotype ranking of field resistance to early leaf spot was Georgia Green most susceptible, Georganic moderately resistant, and DP-1 most resistant. These results corroborate those observed in previous field trials (Cantonwine et al., 2003, 2006). The observed delay of TDO for DP-1 and Georganic compared to Georgia Green was not always consistent, but it suggests that fewer initial infections may have occurred for these genotypes. Although it is likely that the TDO differences detected for the selected disease intensities (5% incidence and 1.5 on Florida scale) were confounded by differences in the rate of disease increase, data from the growth chamber studies suggest there might be a true delay in TDO for DP-1 and Georganic since infection frequency was shown to be reduced by these genotypes. A preliminary experiment to evaluate germination rates of Ca and Cp on leaf surfaces of these genotypes suggest that germination rates of Ca may be suppressed on DP-1 leaves compared to leaves of Georgia Green or Georganic (Cantonwine, 2005).
Reduced infection frequencies likely also contributed to the reduced rates of epidemic progress that were observed for Georganic and DP-1. Genotype ranking by the epidemic rate parameter estimate corresponded more closely to the ranking observed by AUDPC of incidence and severity than did the ranking by disease onset. This is not surprising since the length of time that resistance factors affect disease intensity is greater for measures of rate of increase and AUDPC than disease onset. This result also supports the concept that partial resistance primarily suppresses epidemics by causing a reduction to the rate of disease increase (Parlevliet, 1979).
Infection frequency, reported as an unreliable resistance component for Ca (Ricker et al., 1985; Waliyar et al., 1993), appeared to be an important component of resistance for comparison of genotypes in this study. It is possible that the environmental conditions selected for the growth chamber study, 24 C and high RH, were more suitable to measure differences of infection frequency than a more variable greenhouse environment, or different regime. The frequency and variation of conidial germination rates and infections was found to be greater for Ca under 24/24 C and 26/20 C day/night regimes, than 32/26, 38/26, and 38/32 C regimes (Waliyar et al., 1994, 1995), and for Cp at 20, 24 and 28 C, compared to 32 C (Shew et al., 1988). In addition to lower infection frequencies, components of resistance to Ca measured for DP-1 and Georganic were smaller lesion diameters, reduced percent necrotic areas, and prolonged latent periods. Inconsistencies in measurement techniques throughout the literature and lack of a consistent reference cultivar make comparisons of these results to others difficult. However, our results appear to corroborate those reported previously. The mean size of early leaf spot lesions in this study was similar to the range of diameters of Ca lesions plus necrotic areas surrounding lesions (halo) (approximately 1.7 to 1.8 mm) for the most resistant virginia-type peanut genotypes observed by Waliyar et al. (1994) under the same temperature regime. The means of early leaf spot latent period for DP-1 and Georganic were equal to the T2 latent period (days until two Ca lesions sporulated) observed for NC 3033, the virginia-type genotype with the longest T2 latent period of the genotypes investigated by Ricker et al. (1985). In this study, it was common to find 2 or more lesions with sporulation per leaf on the first day that sporulation was noticed. Measurements of components affecting secondary reproduction of Ca did not differ among genotypes. However, various obstacles to the collection of these data may have inhibited our ability to detect differences for these variables. Sporulation did not occur in trial 1. Instead lesions with stroma and total number of stroma per lesion were counted. Stroma formation is a precursor to sporulation of Ca and Cp lesions (Abdou et al., 1974). Abdou et al. (1974) noticed less defined stroma development within lesions from moderately susceptible Arachis species than highly susceptible species. Estimates of stroma per leaf area did not differ among genotypes, even though the means were 50 and 33% greater for Georgia Green than for DP-1 and Georganic, respectively. In trial 2, sporulation was common for early leaf spots, but spore concentrations were not measured due to contamination of spore quantification vials that resulted in degradation of spores.
Although not assessed in this study, Georganic and DP-1 have been shown to have more field resistance to late leaf spot than Georgia Green (A.K. Culbreath, unpubl. data, 2001; C.C. Holbrook, unpubl. data, 2006). The growth chamber study suggests their resistance is due to reduced infection frequency, smaller lesion size and less percent necrotic area (Tables 5,6). The mean Cp lesion size for all genotypes in this study was smaller than for the most resistant genotypes reported by Chiteka et al. (1988a) or Aquino et al. (1995). Since neither of these earlier studies included Georgia Green, it is unclear whether genetic variation is responsible for the lesion diameter discrepancy observed between the genotypes tested in our study and those tested by Chiteka et al. (1988a) and Aquino et al. (1995), since factors other than genetics have been shown to influence lesion size. Temperature does not appear to be the cause of the lesion diameter discrepancy since the temperatures in Chiteka's greenhouse trials (19.8 to 30.8 C in one trial, mean daily temperatures 27 to 34 C in a second trial) may have been less favorable for lesion expansion than 24 C in this study (Shew et al., 1988). It is possible that the high mean Cp-induced lesions per leaf in this study, which ranged from 55.5 to 101.2 per leaf, limited the expansion potential of Cp lesions. This type of negative interaction between lesion number and lesion size interaction was reported for Ca (Johnson et al., 1986). The mean number of Cp lesions per leaf in Chiteka's study ranged from 8.6 to 13.0, but was not reported by Aquino et al. (1995). The latent periods for the genotypes in this study fell within the range observed by Chiteka et al. (1988a), 12 to 35.0 DAI, and Aquino et al. (1995), 21.8 to 30.8 DAI. Ricker et al. (1985) reported that percent sporulating lesions was a component of resistance for Ca, but that it did not appear to be a resistance component for Cp. We found the opposite trend with the genotypes in our study, but it is important to point out that Ca sporulation data were only recorded for the second trial, and therefore was not repeated.
Overall, the reproduction potential of Cp was greatest for Georganic of the genotypes tested. Although Cp spore production per unit leaf area was not statistically different among genotypes (P = 0.07), they were more similar for Georganic and Georgia Green than for DP-1 (Table 6). Typically, as lesion size decreases in response to host genetics, spore production decreases and latent period increases (Chiteka et al., 1988a; Jogloy et al., 1987). This trend was observed for all genotypes for Ca, and for Georgia Green and DP-1 for Cp, but Georganic presented relatively small Cp lesions, more Cp sporulation and short Cp latent periods. It is possible that the temperature regime chosen in this study was more conducive for sporulation of Cp on Georganic than the other genotypes. Interactions between genotype and temperature have been observed with early leaf spot for incubation period, infection frequency, lesion diameter and latent period (Shew et al., 1988; Waliyar et al., 1994), although no genotype and temperature interactions were observed in the cited studies for spore production. These results suggest that the relative field resistance of Georganic to late leaf spot, compared to Georgia Green or DP-1, may interact with time of disease onset (time available for secondary infection cycles). Information on timing and quantification of secondary inoculum production under field conditions could help assess the precision of the results observed in the growth chamber study, and their effect in the field.
In conclusion, results from the growth chamber study support the field results that DP-1 and Georganic have greater resistance to Ca than Georgia Green. The components of resistance to Ca that were assessed in this study did not distinguish the resistance of DP-1 from Georganic, despite resistance differences observed in the field for these genotypes. Although not directly comparable, the resistance components measured in this study were within the ranges reported for the most resistant peanut genotypes previously reported. This suggests that the partial resistance to Ca and Cp in DP-1 and Georganic is at least in part a function of resistance components that have been previously reported.
Acknowledgements
The authors wish to thank Y.J. Lu for assistance with culture preparation, B. Wilson for assistance with the growth chamber, M. Heath, S. Gremillion, K. Parrish, S. Carter and A. McKeown for general assistance, and B. Mullinix, Jr. for statistical help.
Literature Cited
Abdou Y. A-M. , Gregory W. C. , and Cooper W. E. 1974 Sources and nature of resistance to Cercospora arachidicola Hori and Cercosporidium personatum (Beck and Curtis) Deighton in Arachis species. Peanut Sci 1 : 6 – 11 .
Anderson W. F. , Holbrook C. C. , and Brenneman T. B. 1993 Resistance to Cercosporidium personatum within peanut germplasm. Peanut Sci 20 : 53 – 57 .
Aquino V. M. , Shokes F. M. , Gorbet D. W. , and Nutter F. W. 1995 Late leaf spot progression on peanut as affected by components of partial resistance. Plant Dis 79 : 74 – 78 .
Branch W. D. 1996 Registration of 'Georgia Green' peanut. Crop Sci 36 : 806 .
Branch W. D. and Culbreath A. K. 1995 Combination of early maturity and leaf spot tolerance within an advanced Georgia peanut breeding line. Peanut Sci 22 : 106 – 108 .
Cantonwine E. G. , Culbreath A. K. , Holbrook C. C. , Branch W. D. , and Gorbet D. W. 2003 Field response of runner-type genotypes to multiple diseases under conventional and strip-tillage, 2002. Biol. and Cult. Tests for Control of Plant Dis. (online.) Report 18 . P009. DOI: 10.1094/BC18 The Amer. Phytopath. Soc., St. Paul, MN.
Cantonwine E. G. 2005 The use of genetics and cultural practices to suppress foliar diseases of peanut and reduce fungicide requirements. Ph.D. thesis, Athens University of Georgia .
Cantonwine E. G. , Culbreath A. K. , Stevenson K. L. , Kemerait R. C. , Brenneman T. B. , Smith N. B. , and Mullinix B. G. 2006 Integrated disease management of leaf spot and spotted wilt of peanut. Plant Dis 90 : 493 – 500 .
Cantonwine E. G. , Culbreath A. K. , and Stevenson K. L. 2007 Characterization of early leaf spot suppression by strip-tillage in peanut. Phytopathology 97 : 187 – 194 .
Chiteka Z. A. , Gorbet D. W. , Shokes F. M. , Kucharek T. A. , and Knauft D. A. 1988a Components of resistance to late leaf spot in peanut I. Levels of variability-implications for selection. Peanut Sci 15 : 25 – 30 .
Chiteka Z. A. , Gorbet D. W. , Knauft D. A. , Shokes F. M. , and Kucharek T. A. 1988b Components of resistance to late leafspot in peanut II. Correlations among components and their significance in breeding for resistance. Peanut Sci 15 : 76 – 81 .
Chiteka Z. A. , Gorbet D. W. , Shokes F. M. , and Kucharek T. A. 1997 Components of resistance to early leaf spot in peanut-genetic variability and heritability. Soil Crop Sci. Soc. Florida Proc 56 : 63 – 68 .
Chiyembekeza A. J. , Knauft D. A. , and Gorbet D. W. 1993 Comparison of components of resistance in peanut to late leafspot in different environments. Crop Sci 33 : 994 – 997 .
Cook M. 1981 Susceptibility of peanut leaves to Cercosporidium personatum. Phytopathology 71 : 787 – 791 .
Foster D. J. , Beute M. K. , and Wynne J. C. 1980 Spore production and latent period as mechanisms of resistance to Cercospora arachidicola in four peanut genotypes. Peanut Sci 7 : 88 – 89 .
Gorbet D. W. 2003 New University of Florida peanut varieties for 2003. Marianna 03-2, Marianna NFREC Research Report, .
Green C. C. and Wynne J. C. 1986 Field and greenhouse evaluation of the components of partial resistance to early leafspot in peanut. Euphytica 35 : 561 – 573 .
Jogloy S. , Wynne J. C. , and Beute M. K. 1987 Inheritance of late leafspot resistance and agronomic traits in peanut. Peanut Sci 14 : 86 – 90 .
Johnson C. S. , Beute M. K. , and Ricker M. D. 1986 Relationship between components of resistance and disease progress of early leaf spot on virginia-type peanut. Phytopathology 76 : 495 – 499 .
Lu Y. J. , Shew B. B. , and Ypema H. L. 2003 In vitro sporulation of Cercospora arachidicola from peanut and sensitivity of spore germination to pyraclostrobin. Published online at http://www.apsnet.org/meetings/2003/abstracts/a03ma394.htm.
Melouk H. A. and Banks D. J. 1984 Assessment of resistance to Cercospora arachidicola in peanut genotypes in field plots. Plant Dis 68 : 395 – 397 .
Norden A. J. , Lipscomb R. W. , and Carver W. A. 1969 Registration of 'Florunner' peanut. Crop Sci 9 : 850 .
Parlevliet J. E. 1979 Components of resistance that reduce the rate of epidemic development. Annu. Rev. Phytopathol 17 : 203 – 222 .
Ricker M. D. , Beute M. K. , and Campbell C. L. 1985 Components of resistance in peanut to Cercospora arachidicola. Plant Dis 69 : 1059 – 1064 .
Shaner G. and Finney R. E. 1977 The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat. Phytopathology 67 : 1051 – 1056 .
Shew B. B. , Beute M. K. , and Wynne J. C. 1988 Effects of temperature and relative humidity on expression of resistance to Cercosporidium personatum in peanut. Phytopathology 78 : 493 – 498 .
Shew B. B. , Sommartya T. , and Beute M. K. 1989 Components of partial resistance in peanut genotypes to isolates of Cercosporidium personatum from the United States and Thailand. Phytopathology 79 : 136 – 142 .
Shokes F. M. and Culbreath A. K. 1997 Early and late leaf spots. 17 – 20 In Kokalis-Burelle N. , Porter D. M. , Rodriguez-Kabana R. , Smith D. H. , and Subrahmanyam P. Compendium of Peanut Diseases, 2nd Ed APS Press St. Paul, MN .
Smith N. B. 2003 Peanut production survey state results, 2003. Univ. Georgia. Coop. Ext. Publ Econ. College of Agricultural and Environmental Sciences .
Steel R. G. B. and Torrie J. H. 1980 Principles and Procedures of Statistics. New York McGraw-Hill .
Subrahmanyam P. , McDonald D. , Gibbons R. W. , Nigam S. N. , and Nevill D. J. 1982 Resistance to rust and late leafspot diseases in some genotypes of Arachis hypogaea. Peanut Sci 9 : 6 – 10 .
Waliyar F. , McDonald D. , Subba Rao P. V. , and Reddy P. M. 1993 Components of resistance to an Indian source of Cercospora arachidicola in selected peanut lines. Peanut Sci 20 : 93 – 96 .
Waliyar F. , Shew B. B. , Stalker H. T. , Isleib T. G. , Sidahmed R. , and Beute M. K. 1994 Effect of temperature on stability of components of resistance to Cercospora arachidicola in Peanut. Phytopathology 84 : 1037 – 1043 .
Waliyar F. , Shew B. B. , Sidahmed R. , and Beute M. K. 1995 Effects of host resistance on germination of Cercospora arachidicola on peanut leaf surfaces. Peanut Sci 22 : 154 – 157 .
Walls S. B. , Wynne J. C. , and Beute M. K. 1985 Resistance to late leafspot of peanut of progenies selected for resistance to early leafspot. Peanut Sci 12 : 22 – 27 .
Watson G. R. , Kucharek T. A. , and Shokes F. M. 1998 Components of resistance in peanut to late peanut leaf spot. Soil and Crop Sci. Soc. Florida Proc 57 : 87 – 91 .
Wynne J. C. , Beute M. K. , and Nigam S. N. 1991 Breeding for disease resistance in peanut (Arachis hypogaea L.). Annu. Rev. Phytopathol 29 : 279 – 303 .
Notes
- Department of Biology, Valdosta State University, Valdosta GA 31698, egcantonwine@valdosta.edu [^]
- Department of Plant Pathology, University of Georgia, Tifton, GA 31793-0748 [^]
- USDA-ARS, Crop Genetics and Breeding Research Unit, Tifton, GA 31793-0748 [^]
- Department of Agronomy, University of Florida NFREC, Marianna, FL 32446-7906 [^] * Corresponding author - Emily Cantonwine
Author Affiliations