Introduction
Peanut is a major food and cash crop in Ghana, especially in northern Ghana which accounts for 92% of the total national peanut production (SRID, 2004). However, average yields of 840 kg/ha obtained on farmers' fields in Ghana are low compared to yields of over 2500 kg/ha reported in developed countries such as the United States (FAO, 2002). Relatively low peanut yield in Ghana and other parts of West Africa is attributed largely to the deleterious effects of soil arthropod pests, soil and foliar disease, nematodes, and weed interference (Kishore, 2005; Umeh et al., 2001; Wightman et al., 1990). Millipedes (Myriapoda: Odontopygidae), termites (Isoptera: Termitidae), white grubs (Coleoptera: Scarabaeidae), and wireworms (Coleoptera: Elateridae) contribute to peanut yield loss in West Africa. These pests feed largely on roots, developing pods, and kernels (Umeh et al., 1999; Wightman et al., 1990). Yield loss from termite ranges from 21 to 50% in West Africa (Johnson et al., 1981; Umeh, et al. 1999). Infestation by these pests also predisposes pods to attack by disease causing organisms such as the carcinogenic fungus Aspergillus flavus (Link) (Lynch et al., 1990; Waliyar et al., 1994; Wightman et al., 1990).
Soil insecticides can reduce pest damage and increase yield in peanut (Lynch et al., 1990; Wightman et al., 1990). However, peanut farmers in northern Ghana seldom apply insecticides to control arthropods because they lack the necessary capital to purchase pesticides. Cultural practices are the only practical means to control arthropods and other pests in northern Ghana. Adjustment of planting and harvest dates and field sanitation make the environment less favorable for survival, growth, or reproduction of pests and are often essential cultural practices in pest management programs for peanut (Abate et al. 2000; van Huis and Meerman, 1997). Planting date adjustment has been effective in minimizing pest damage in peanut (Lynch and Douce, 1992; NRI, 1996; Smith and Barfield 1982). Leuck (1967) reported that peanut sown prior to mid-April in the southeastern United States usually escapes damage by the lesser cornstalk borer (Elasmopalpus lignosellus). Lynch and Douce (1992) also noted that adjusting planting date is a viable strategy for preventing late-season damage to pods by termites, especially in regions that have a distinct seasonal rainfall pattern. However, in many West African countries farmers must plant when rainfall is most plentiful to optimize yield, regardless of the impact of rainfall on pest development (Naab et al., 2005; Tsigbey et al., 2003).
Host plant resistance to insects has also shown potential for management of pests that damage peanut (Lynch, 1990; Lynch and Douce, 1992; Umeh et al., 2001). Lynch (1990) found a high degree of resistance to pod scarification by arthropods in NCAc 343 with increases in peanut pod yield in Burkina Faso compared with other less resistant lines and cultivars. Weed control early in the growing season can reduce reservoirs for arthropods, pathogens, and nematodes (Ghewande and Nandagopal, 1997; Hillocks 1998).
Defining interactions among cultural practices is important when developing effective pest management strategies for peanut. Therefore, the objective of this research was to evaluate interactions among cultivar, planting date, and weed management systems with respect to soil arthropod density, penetration and scarification of pods from arthropod feeding, foliar disease incidence, and pod yield of peanut in northern Ghana.
Materials and Methods
Experiments were conducted during 2003, 2004, and 2005 at the CSIR-Savanna Agricultural Research Institute located near Nyankpala (9° 42′ N latitude, 0° 92′ W longitude, and 184 m altitude) in the northern Ghana, West Africa. The soil was a sandy-gravelly loam with less than 3% organic matter. Two seeds were planted per hill and later thinned to one plant per hill 10 days after emergence. The experimental unit consisted of four rows (50 cm spacing) by 5 m long. Final in-row density of peanut was five plants/m. Plot ranges were separated by a 5-m alley.
The cultivars Chinese (90-d maturity), Manipintar (125 to 130-d maturity), JL 24 (85 to 90-d maturity), and RLRS-11 (90-d maturity) were planted on 28 May, 11 June, 25 June, and 9 July of each year. The three weed management systems comprised weeding by hand at four weeks, six weeks, and four and six weeks after planting (WAP). The experiments were planted in a split-split-plot design. Weed management systems were used as main plots, peanut cultivars as subplots, and planting date as sub-subplots. A randomized complete block design structure with three replications was used for the main plot experimental units.
Five plants were randomly selected from the outside rows of each experimental unit at harvest to determine arthropod damage to pods and arthropod density in soil. Five soil cores (15 cm3) from each experimental unit were removed to quantify soil arthropods. At optimum pod maturity, plants in the middle two rows of each plot were harvested to determine yield. A sample of 100 mature pods from each plot was examined for pod penetration and hull scarification. The percentage of pods damaged out of the total number of pods collected from each plot was determined for each experimental unit. Weed composition by species was determined from two random quadrants (1 m2) from each experimental unit. Incidence of early and late leaf spot was rated at harvest on a scale of 1 to 10 based on visual observation on 10 randomly selected plants in each plot (Chiteka etal., 1997).
Data for arthropod density were transformed using log10 (X + 1), and percentage data were transformed using square root transformation to normalize variances (Gomez and Gomez, 1984; SAS Institute, 1998). Data for arthropod density, percent pod penetration, percent pod scarification, weed mass by species, and pod yield were subjected to analysis of variance according to the split-split-plot design pooled over years. Means of significant main effects and interactions were separated using Fisher's Protected LSD test at p ≤ 0.05 (SAS Institute, 1998). Relationships between soil arthropod pest density, percent scarified pods, percent penetrated pods, and pod yield were examined using Pearson correlation analyses (SAS Institute, 1998). Since the four soil arthropod pests inflict a common damage (pod penetration) to pods (Johnson et al., 1981; Umeh et al., 1999), cumulative numbers of pests were used in the correlation analysis. However, because only termites scarify pods (Johnson and Gumel, 1981), the relationship between termite densities and pod scarification was also examined.
Results
Soil Arthropods
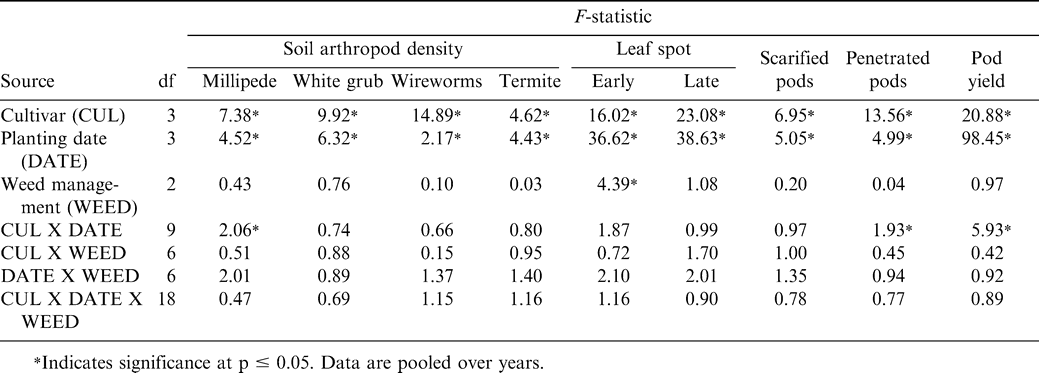
The interaction of cultivar X planting date was significant for millipede density at harvest (Table 1). Although this interaction was not significant for white grub, wireworm, or termite density, main effects of cultivar and planting date were significant (Table 1). The other main effects and interactions were not significant for these soil arthropod pests (Table 1).
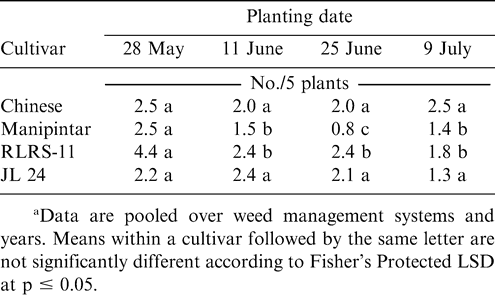
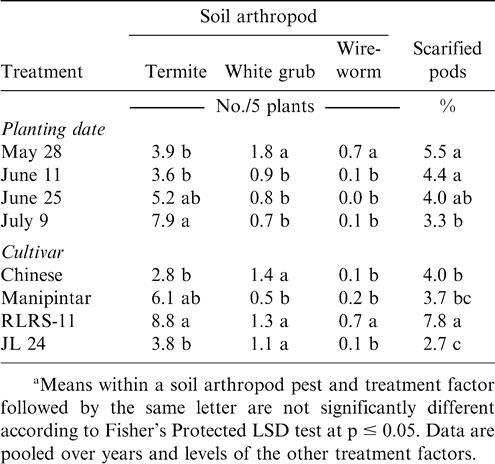
Millipede density was higher when Manipintar and RLRS-11 were planted 28 May rather than 11 or 25 June or 9 July (Table 2). Planting date did not affect millipede density of the cultivar Chinese or JL 24. The lowest millipede density for the cultivar Manipintar was noted when peanut was planted 25 June. Termite density was higher when peanut was planted 9 July compared with planting 28 May or 11 June (Table 3). In contrast, white grub and wireworm densities were the highest when peanut was planted 28 May. Variation in termite, white grub, and wireworm density was also associated with cultivar (Table 3). Density for at least one soil arthropod taxon was lower for the cultivars Chinese, Manipintar, and JL 24 than for RLRS-11.
Main effects of cultivar and planting date were significant for the percentage of scarified pods (Table 1). A higher percentage of pods were scarified when peanut was planted 28 May or 11 June compared with planting 9 July (Table 3). The cultivar RLRS-11 had more scarified pods than Chinese, Manipintar, or JL 24 (Table 3). Greater scarification was noted on pods from Chinese compared with JL 24. There was no difference in pod scarification when comparing Manipintar and JL 24. Pod penetration was highest when peanut was planted 28 May for the cultivar Manipintar (Table 4). The lowest penetration was recorded on 25 June for RLRS-11. This cultivar also had the lowest penetration across planting dates.
Cumulative soil arthropod densities were positively correlated with the percentage of scarified pods (r = 0.22, p ≤ 0.0001) and the percentage pf penetrated pods (r = 0.15, p = 0.0025) (data not shown). However, there was a nonsignificant negative relationship between cumulative pest densities and pod yield (r = -0.09, p = 0.0765) (data not shown). A positive correlation between the percentage of scarified pods and the percentage of penetrated pods was noted (r = 0.21, p ≤ 0.0001) (data not shown). The percentage of scarified pods was also correlated with pod yield (r = -0.10, p = 0.0521) (data not shown). Inconsistent results were observed for the relationship between the percentage of penetrated pods and pod yield (r = 0.19, p = 0.0001) (data not shown). When individual pest density was used in the analyses, millipede density demonstrated a positive correlation with pod scarification (r = 0.33, p ≤ 0.0001) and pod penetration (r = 0.22, p ≤ 0.001), and a negative correlation with pod yield (r = -0.18, p =0.0003) (data not shown). Termite density was positively correlated only with the percentage of scarified pods (r = 0.10, p = 0.042) (data not shown).
Early and Late Leaf Spot
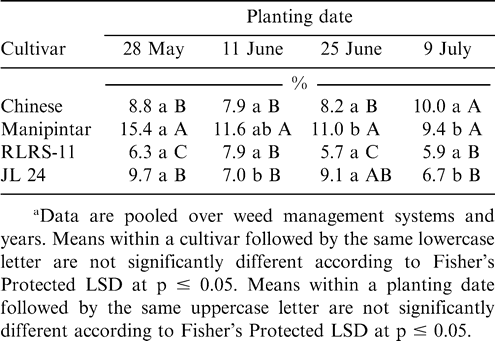
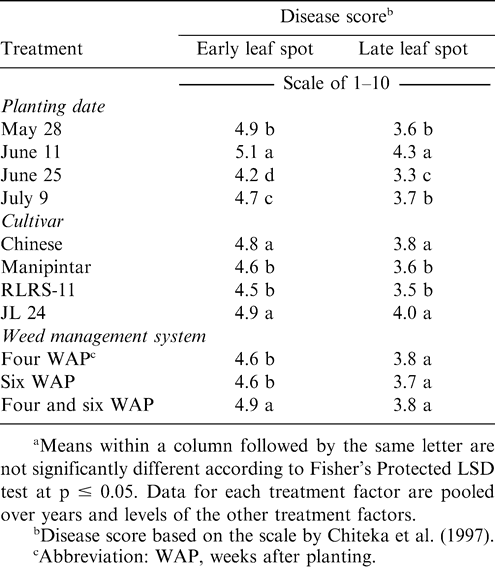
Early leaf spot incidence was affected independently by planting date, cultivar, and weed management system (Table 1). While late leaf spot incidence was affected by cultivar and planting date, weed management system had no effect on this disease. When pooled over years, early and late leaf spot incidence was highest at harvest when peanut was planted 11 June (Table 5). Early leaf spot incidence was higher when peanut was planted 28 May than 25 June or 9 July; and incidence was higher when peanut was planted 9 July compared with planting 25 June. No difference in late leaf spot incidence was noted when peanut was planted 28 May or 9 July. The lowest incidence of late leaf spot was noted when peanut was planted 25 June.
Incidence of early and late leaf spot was higher for the cultivars Chinese and JL 24 than Manipintar and RLRS-11 (Table 5). Thus, considerable host plant resistance was shown for Manipintar and RLRS-11. Although weed management system did not affect late leaf spot, incidence of early leaf spot was the highest when peanut was weeded twice at four and six WAP (Table 5). Incidence of leaf spot was higher when peanut was planted in late May or early June than when planted later.
A significant positive correlation was observed between early leaf spot and late leaf spot (r = 0.65, p = 0.0001). There was also a significant, but weak positive correlation between yield and early leaf spot (r= 0.19, p = 0.0001) or late leaf spot (r = 0.24, p = 0.0001).
Weed Effects
There were no significant effects of weed management system on arthropod pest density, pod penetration and scarring, or pod yield (Table 1). Seventeen broadleaf weeds, five grasses, and one annual sedge were documented over the three years (data not presented). Weed density was relatively low compared to many farmers' fields, with the predominant broadleaf species consisting of Hyptis sp. and Euphorbia sp. (data not presented). These observations probably explain partially the lack of significant effect of weed management system in this experiment. Weeds generally do not interfere with peanut yield when removed within six WAP (Anonymous, 2005; N'zala et al., 2002; Sibuga et al., 1989).
Peanut Yield
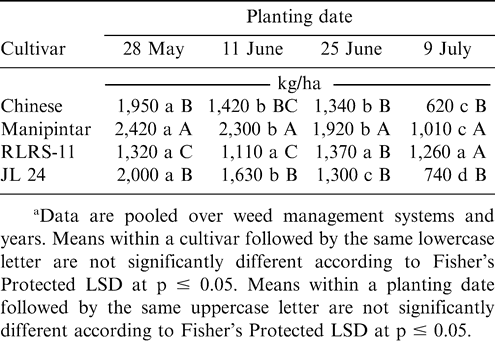
Peanut pod yield was affected by the interaction of planting date and cultivar (Table 1). Pod yield decreased as planting was delayed for the cultivars Chinese, Manipintar, and JL 24 (Table 6). In contrast, yield of RLRS-11 remained the same regardless of planting date. Yield of Manipintar was higher than yield of RLRS-11 or JL 24 when planted 28 May, 11 June, and 25 June and for all planting dates for Chinese (Table 6). The cultivar RLRS-11 yielded lower than the other cultivars when planted 28 May.
Discussion
The four soil arthropods represented in this experiment have been documented as major pests of peanut in West Africa (Wightman et al., 1990; Umeh et al., 1999, 2001). Similar to reports by Umeh et al. (1999, 2001) in other regions of Africa, densities of wireworms were relatively low compared to the other soil arthropods in peanut.
A positive response to early planting even though pod penetration and scarification from arthropod feeding was higher may have been associated with rainfall patterns. A distinct seasonal rainfall pattern is noted in northern Ghana, and planting in early May results in a higher percentage of the growing season occurring during adequate soil moisture (Naab et al., 2005; Tsigbey et al., 2003). Although damage from arthropods is generally lower with later plantings, soil moisture is also limited during that period of the year. Consequently, moisture is a more yield limiting factor than arthropod damage (Crosthwaite, 1994; Pilcher and Rice, 2001; Rogers et al., 2005; Wright and Nageswara Rao, 1994). Naab et al. (2004) reported a higher simulated yield loss due to water deficit for late-planted peanut than for early planting dates in northern Ghana. Our results agree with those of Naab et al. (2004, 2005), who reported that early planting of peanut resulted in higher peanut pod yield than later plantings.
The interaction of planting date and cultivar demonstrated that pods from Manipintar had more penetrated pods when planted at the first planting date compared to later plantings. As a full season cultivar (125-d maturity), Manipintar probably suffered higher pod penetration because of longer exposure to arthropods than the other cultivars in this experiment that reach optimum maturity by 85 to 95 d after planting. Pod yield, however, was greatest in Manipintar than the three early maturing cultivars. The reason for this high yield may have been due to the longer pod-filling period for Manipintar compared to the short duration cultivars or the inherent yield potential of this cultivar. Naab et al. (2005) also reported that the long duration cultivar F-mix gave higher yield than the short maturity cultivar Chinese. The interaction of cultivar and planting date also demonstrated that Chinese and JL 24 yielded the lowest when planted in July compared with earlier plantings. These cultivars are early maturing and are probably best planted at the beginning of the cropping season.
Cumulative pest densities were positively correlated with the percentage of scarified and penetrated pods. However, the percentage of scarified pods was the only parameter that negatively affected pod yield. Damaged pods are also predisposed to infection by secondary organisms some of which pose hazards to human health (Wightman and Wightman, 1994). Umeh et al. (2001) reported that arthropod-damaged pods are often infected by Aspergillus flavus which produces mycotoxins that can adversely affect human health. There was also a significant positive correlation between numbers of scarified pods and penetrated pods, suggesting that pod scarification by termites weakened pods and made it easier for penetration by other soil pests (Johnson and Gumel, 1981).
Results from this research confirm earlier findings demonstrating that peanut planted early in the season suffer greater leaf spot severity than those planted late (Naab et al., 2005). Weather conditions earlier in the season are often conducive to disease development because of rainfall patterns. Ringer and Grybauskas (1995) found that rainfall early in the season was positively correlated with levels of gray leaf spot on maize. Jensen and Boyle (1966) and Smith and Crosby (1973) also reported rapid increases in peanut leaf spot severity with periods of high relative humidity shortly after the onset of rainfall. Naab et al. (2005), however, observed that main-stem defoliation of peanut due to leaf spot was lower when peanut was planted early. Mainstem defoliation was not measured in the present study.
Higher incidence of early leaf spot when peanut was weeded twice may have been associated with movement of soil on vines by the additional weed removal procedure. Movement of pathogen inoculum from soil may have been exacerbated by making two trips across the field for weed control. Cultivation is often discouraged in peanut because of the potential for moving soil onto peanut vines (Wilcut et al., 1995).
Results from these experiments demonstrate that millipedes, termites, and white grubs as well as early and late leaf spots are important pests in northern Ghana. Additionally, weeding within six weeks after planting most likely minimized interference and reduced contributions to soil arthropod pests from weeds serving as alternate hosts. However, it is unknown as to whether or not weeds present in the field after six weeks would affect soil arthropod pest density and distribution associated with peanut. Arthropod pest density and pod damage as well as incidence of early and late leaf spot were generally higher in early planted than in later planted peanut. However, pod yields were higher when peanut was planted earlier. This suggests that soil arthropod pests and disease effects on peanut are less critical than planting during a period of time that optimizes peanut exposure to soil moisture.
Acknowledgements
The study was made possible through support provided by the United States Agency for International Development (USAID) Peanut CRSP (LAG-G-00-96-90013-00). Appreciation is expressed to D. Opare-Atakora, Issifu Ramat, and Fred Anaman for their technical assistance in the field.
Literature Cited
Abate T. , van Huis A. , and Ampofo J. K. O. 2000 Pest management strategies in traditional Agriculture: An African perspective. Ann. Rev. Entomol 45 : 631 – 659 .
Anonymous 2005 http://www.ikisan.com/links/ap_peanutWeed20Management.html
Chiteka Z. A. , Gorbet D. W. , Shokes F. M. , and Kucharek T. A. 1997 Components of resistance to early leaf spot in peanut - genetic variability and heritability. Soil Crop Sci. Soc. Fl. Proc 56 : 63 – 68 .
Crosthwaite I. 1994 Peanut growing in Australia. Brisbane QDPI Queensland Department of Primary Industries Information Series Q194019. 93 .
FAO 2002 Food and Agricultural Organization Crop Production Statistics. Rome, Italy .
Ghewande M. P. and Nandagopal V. 1997 Integrated pest management in peanut (Arachis hypoaea L.) in India. Integrated Pest Management Reviews 2 : 1 – 15 .
Gomez K. A. and Gomez A. A. 1984 Statistical procedures for agricultural research. 2nd edition NY John Wiley and Sons, Inc .
Hillocks R. J. 1998 The potential benefits of weeds with reference to small holder agriculture in Africa. Integrated Pest Management Reviews 3 : 155 – 167 .
Jensen R. E. and Boyle L. W. 1966 A technique for forecasting leafspot on peanuts. Plant Dis 50 : 810 – 814 .
Johnson R. A. and Gumel M. H. 1981 Termite damage and crop loss studies in Nigeria- the incidence of termite-scarified peanut pods and resulting kernel contamination in field and market samples. Tropical Pest Management 27 : 343 – 350 .
Johnson R. A. , Lamb R. W. , and Wood T. G. 1981 Termite damage and crop loss studies in Nigeria – a survey of damage to peanut. Tropical Pest Management 27 : 325 – 342 .
Kishore G. K. and Pande S. 2005 Integrated management of late leaf spot and rust diseases of peanut (Arachis hypogeae L.) with Prosopis juliflora leaf extract and chlorothalonil. Int'l J. Pest Management 51 : 325 – 332 .
Leuck D. B. 1967 Lesser cornstalk borer damage to peanut plants. J. Econ. Entomol 60 : 1549 – 1551 .
Lynch R. E. , Ouedraogo A. P. , and Some S. A. 1990 Effect of harvest date and termite-resistant varieties on termite and millipede damage to peanut in Burkina Faso. 87 – 90 In Summary Proceedings of the First ICRISAT Regional Peanut Meeting for West Africa, 13–16 September 1988 Niamey, Niger ICRISAT Sahelian Center .
Lynch R. E. 1990 Resistance in peanut to major arthropod pests. Florida Entomol 73 : 422 – 455 .
Lynch R. E. and Douce G. K. 1992 Implementation of integrated pest management in peanut: current status and future direction. 267 – 278 In Nigan S. N. Peanut – a global perspective: proceedings of an international workshop, 25–29 November 1991 India ICRISAT Center .
N'Zala D. , Naadjidjim J. , and Ngaka A. 2002 Weed population dynamics during the growth cycle in the wet tropical zone of Kombe (Congo). Weed Research 42 : 100 – 106 .
Naab J. B. , Tsigbey F. K. , Prasad P. V. V. , Boote K. J. , Bailey J. E. , and Brandenburg R. L. 2005 Effect of sowing date and fungicide application on yield of early and late maturing peanut cultivars grown under rainfed conditions in Ghana. Crop Protection 24 : 325 – 332 .
Naab J. B. , Singh P. , Boote K. J. , Jones J. W. , and Marfo K. O. 2004 Using the CROPGROW-Peanut model to quantify yield gaps of peanut in the Guinea Savanna zone of Ghana. Agronomy J 96 : 1231 – 1242 .
NRI 1996 Natural Resources Institute Peanut. Pest Control Series 348 .
Pilcher C. D. and Rice M. E. 2001 Effect of planting date and Bacillus thuringiensis corn on the population dynamics of European corn borer (Lepidoptera: Crambidae). J. Econ. Entomol 94 : 730 – 742 .
Ringer C. E. and Grybauskas A. P. 1995 Infection cycle component of disease progress of gray leaf spot on field corn. Plant Dis 97 : 24 – 28 .
Rogers D. J. , Ward A. L. , and Wightman J. A. 2005 Damage potential of two scarab species on groundnut. Int'l. J. Pest Management 51 : 305 – 312 .
SAS Institute 1998 SAS User's Guide, 9th Ed Cary, North Carolina SAS Institute .
Sibuga K. P. , Bwana E. N. , and Mwakitwange F. E. 1989 Effect of time of weeding on peanut yield. 213 – 217 In Proceedings of the Third Regional Peanut Workshop for Southern Africa, 13–18 March 1988 International Crops Research Institute for the Semi-Arid Tropics .
Smith J. W. and Barfield C. S. 1982 Management of preharvest insects. 250 – 325 In Pattee H. E. and Young C. T. Peanut Science and Technology Yoakum, TX American Peanut Research and Education Society .
Smith D. H. and Crosby F. L. 1973 Aerobiology of two leafspot fungi. Phytopathol 63 : 703 – 707 .
SRID 2004 Production of major crops in Ghana, 2003. Statistics, Research and Information Directorate (SRID). Ministry of Food and Agriculture .
Tsigbey F. K. , Brandenburg R. L. , and Clottey V. A. 2003 Peanut production methods in northern Ghana and some disease perspectives. World Geography pf Peanut. USAID Peanut CRSP. Web page: http://www.lanra.uga.edu/peanut/knowledgebase/tsigbey1.cfm .
Umeh V. C. , Waliyar F. , Traore S. , and Egwurube E. 1999 Soil pests of peanut in West Africa - species diversity, damage and estimation of yield losses. Insect Sci. Applic 19 : 131 – 140 .
Umeh V. C. , Yuom O. , and Waliyar F. 2001 Soil pests of peanut in sub-Saharan Africa – A Review. Insect Sci. Applic 21 : 23 – 32 .
van Huis A. and Meerman F. 1997 Can we make IPM work for resource-poor farmers in sub-Saharan Africa. Int'l. J. Pest Management 43 : 313 – 320 .
Waliyar F. , Ba A. , Hamma H. , Bonkoungou S. , and Bosc J. P. 1994 Sources of resistance to Aspergillus flavus and aflatoxin contamination in peanut genotypes in West Africa. Plant Disease 78 : 704 – 708 .
Wightman J. A. and Wightman A. S. 1994 An insect, agronomic and sociological survey of peanut fields in southern Africa. Agric. Ecosystem Environ 51 : 311 – 331 .
Wightman J. A. , Dick K. M. , Ranga Rao G. V. , Shanower T. G. , and Gold C. G. 1990 Pests of peanut in the semi-arid tropics. 243 – 322 In Singh S. R. Insect Pests of Legumes New York, New York Longman and Sons Ltd .
Wilcut J. W. , York A. C. , Grichar W. J. , and Wehtje G. R. 1995 The biology and management of weeds in peanut (Arachis hypogaea). 207 – 244 In Pattee H. E. and Stalker H. T. Advances in Peanut Science Stillwater, OK Am. Peanut Res. and Education Soc .
Wright G. C. and Nageswara Rao R. C. 1994 Peanut water relations. 281 – 335 In Smart J. The peanut Crop - A scientific Basis for Improvement London, UK Chapman and Hall Ltd .
Notes
- CSIR-Savanna Agricultural Research Institute, P. O. Box 52, Tamale, Ghana, West Africa. [^]
- University for Development Studies, P. O. Box 1350, Tamale, Ghana, West Africa. [^]
- Department of Entomology, North Carolina State University, Box 7613, Raleigh, NC 27695-7613. [^]
- Department of Crop Science, North Carolina State University, Box 7620, Raleigh, NC 27695-7620. [^]
- Corresponding author's e-mail: david_jordan@ncsu.edu. [^]
Author Affiliations